Falsification of drugs and their illegal turnover in the pharmaceutical market is a global problem nowadays. International organizations are making significant efforts in combating illicit trafficking of counterfeit medicines. The Republic of Kazakhstan is no exception. Over the past decade, the Ministry of health of the Republic of Kazakhstan, more than 90 facts of realization in the retail segment of the pharmaceutical market 40 kinds of counterfeit drugs. A key role in the implementation of activities related to the detection and prevention of falsification of medicines is given to pharmaceutical employees.
Introduction.
Quality assurance pharmaceuticals (p) is one of the main goals of the National drug policy, which recommended by the world health organization (who) to build an effective state policy in the health system and pharmaceutical providing of the population [1, 2]. One of the key positions in the quality assurance the pharmaceuticals belongs to the pharmaceutical employees. Active fight against counterfeit pharmaceuticals in the world has more than 25 years. For the first time at the international level about the alarming amount of fraud drugs, the who said in 1987. [1,3,4] Despite the significant efforts of international organizations (who, Interpol, the Association of international pharmaceutical manufacturers, etc.), as well as the competent authorities, functioning in national health systems, the problem of illegal trafficking of counterfeit remains relevant. According to the latest who data, the proportion of counterfeit drugs on the national pharmaceutical market of the various countries is 5 to 7% [2,5,6]. Unfortunately, the Republic of Kazakhstan (RK) is no exception. Over the past 10 years the Ministry of health established more than 90 facts of realization in the retail segment of the pharmaceutical market (PM) 40 kinds of counterfeit items of the pharmaceuticals. The competent authorities had withdrawn about 40 thousand packs of counterfeit items of PL, in which illegal and unlawful method was involved in more than 500 pharmaceutical activities [6,7,8]. It should be noted that the definition of "counterfeit medicines" in the regulatory framework governing the trafficking of drugs in PM as well as measures of responsibility for distribution of counterfeit products in the RK was adopted only in 2006 [7].
In most countries the direct responsibility of pharmacists and pharmacists in the provision of population pharmaceutical care is the organization and carrying out of a complex of measures aimed at preventing the emergence of the PM falsified/counterfeit drugs. Therefore, the analysis of the level of awareness of pharmaceutical workers on the issue of trafficking in counterfeit drugs on the PM of RK has relevance and socioeconomic importance.
The study materials and methods.
To achieve the main goals of the research we developed a questionnaire that consisted of two parts. The first part was presented by closed and open questions on the studied issue, while the second part - the personal data of the respondents, namely gender, age, education, years of practical experience in pharmacy. Previously, before the survey were reported to the confidential terms of use information received. The required sample size of respondents was calculated based on the expected reliability of the obtained results, i.e. the p value <0.05 was considered statistically significant [12]. The survey was conducted in 300 pharmacies in Almaty, Kazakhstan in 20162017. Just was offered for survey 350 questionnaires in the further work were selected 302 questionnaires, which were carefully filled all of its components. Previously, we have conducted assessment of the homogeneity of the entire set of respondents (302 people), as well as the degree of consistency of the answers of experts to questions of the questionnaire. So, by using standardized statistical approaches it was proved that the sample of respondents for indicator variables that were relatively homogeneous, therefore, the evaluation of the survey results was carried out on the total population of the respondents. In addition, an assessment of the consistency of answers of respondents for questions with the option of a multiplicity of answers. For this, we used the Pearson correlation coefficient ( ). In this case, the sample was vector = (where: j - number of the sample (group of respondents); i (i = 1,m) - the number of answer for the questions in the questionnaire; xji is the number of respondents in the group of specialists, who noted th answer. The calculated ratio ( ) was within the boundaries . A significance test of the Pearson correlation coefficient was the criterion ofStudent (tкр) [12].
Positive, from the point of view of the level of awareness of workers about the falsification of the pharmaceuticals on PM seem to be the results of the responses to the first question of the questionnaire, which was formulated thus: "do You Know what "falsified/counterfeit drug?". 256 people (84,76%) answered "Yes, familiar," 40 people (13,25%) indicated the answer "Difficult to answer" and only 6 (1,99%) of respondents answered "No, not familiar with what a concept". The next closed question "were there any cases of detection of counterfeit pharmaceuticals in Your practical activities?" 222 (73,51%) respondents answered No, while the rest (80 people or 26,49%) of respondents were familiar with the problem of counterfeit drugs. Further, the portion of respondents who met in their practice with counterfeit drugs, were asked to note how frequently they faced this problem. It was proposed to mark such answers: "Once"; "Periodically". Of the 80 interviewed experts who said that they are familiar with counterfeit drugs in their professional activities the vast majority, i.e. 52 people said "Once". As you can see, every fourth Respondent, the expert met in their work with the fact of falsification of the pharmaceuticals, which came in the pharmacy network for sale to the public. It's 17.22% of all the surveyed experts, or 65.0% of the respondents who noted the identification of counterfeit pharmaceuticals in their practice. The remaining 28 people responded that "Occasionally" collide with the fact of falsification of drugs. Thus, 14 (4.64% of all surveyed experts) man further noted the answer "1 year", 11 (3,64% of all respondents) respondents - "1 every 6 months", 1 Respondent (0.32 per cent respectively) - the "1 per quarter" and 2 people said the answer "1 per month".
Interesting is the analysis of the survey results the following question. Pharmaceutical employees who have experience with the forged pharmaceuticals (80 respondents) were asked to answer the question: "what are the signs of the inconsistencies You were an established fact of falsification of the pharmaceuticals that came to the pharmacy?". It was proposed to mark such answers:
- "The data of accompanying documents and the information included on the packaging of the pharmaceuticals";
- "The lack of a complete set of documents received by the pharmacy";
- "Marking on the outer (primary) packaging of the goods";
- "Design and status of the external secondary packaging pharmaceuticals";
- "The absence of instructions to the pharmaceuticals";
- "Failure to follow the procedure and complete reflection of the necessary information in the instructions on the pharmaceuticals";
- "Difficult to answer".
The results of data processing for this question is presented in figure 1.
The survey we have found that the greatest number of respondents indicated the answer "Marking on the outer (primary) packaging of the goods" (28 expert or 35.0% from persons who have had experience in detecting counterfeit drugs). Further, the frequency of reflection by the experts followed by the answer "Design condition and appearance of secondary packaging drugs" (20 experts or 25.0%, respectively). Third place was taken by a "Data accompanying documents and information contained on the packaging of the drugs" (17 experts or 21,25%). "The lack of a complete set of product documentation, received by the pharmacy," said 6 experts (7,5%), "Lack of instructions on the pharmaceuticals" - 4 experts (5,0%) and "failure to follow the procedure and complete reflection of the necessary information in the instructions on the pharmaceuticals " only 2 respondents (2.5 percent). Difficulties with the wording of the answer to this question had 3 respondents, which made up 3,75% of all pharmaceutical workers who had practical experience of the detection of counterfeit pharmaceuticals in pharmacies.
The next set of questions concerned the organization of the complex measures to prevent fraud on PM of pharmaceuticals of RK. These questions were asked to all respondents, regardless of whether they had experience of detection of counterfeit products in pharmacies or not. Thus, in the processing we used the data of
all 302 questionnaires. The following question was formulated thus: "what is the most important in Your opinion the factors that contribute to fraud drugs in Kazakhstan?". By results of processing of questionnaires, the rating factors presented in table 1. As you can see in table 1 the three leaders formed factors that have a direct relationship to the functioning of the state system of ensuring the quality of pharmaceuticals in the Republic of Kazakhstan. Every third interviewed expert noted that the most important factor contributing to the falsification of drugs in RK is "Ineffective organization of quality control of imported to Kazakhstan pharmaceutical products by the competent state authorities". Further, by a considerable margin followed by the answers "Flaw in the organization of work of the competent authorities governing the circulation of pharmaceuticals in Kazakhstan" and "Lack of strict sanctions in case of detection of falsification of pharmaceutical products and its illegal and illegitimate implementation on PM RK". These factors are noted every fourth expert, who participated in the survey. Every sixth Respondent said this important as we consider the factor as a "Low level of coordination of government bodies responsible for the implementation of quality pharmaceutical services to the population of Kazakhstan", and every seventh is the "Low level of efficiency of state regulation on FR in General."
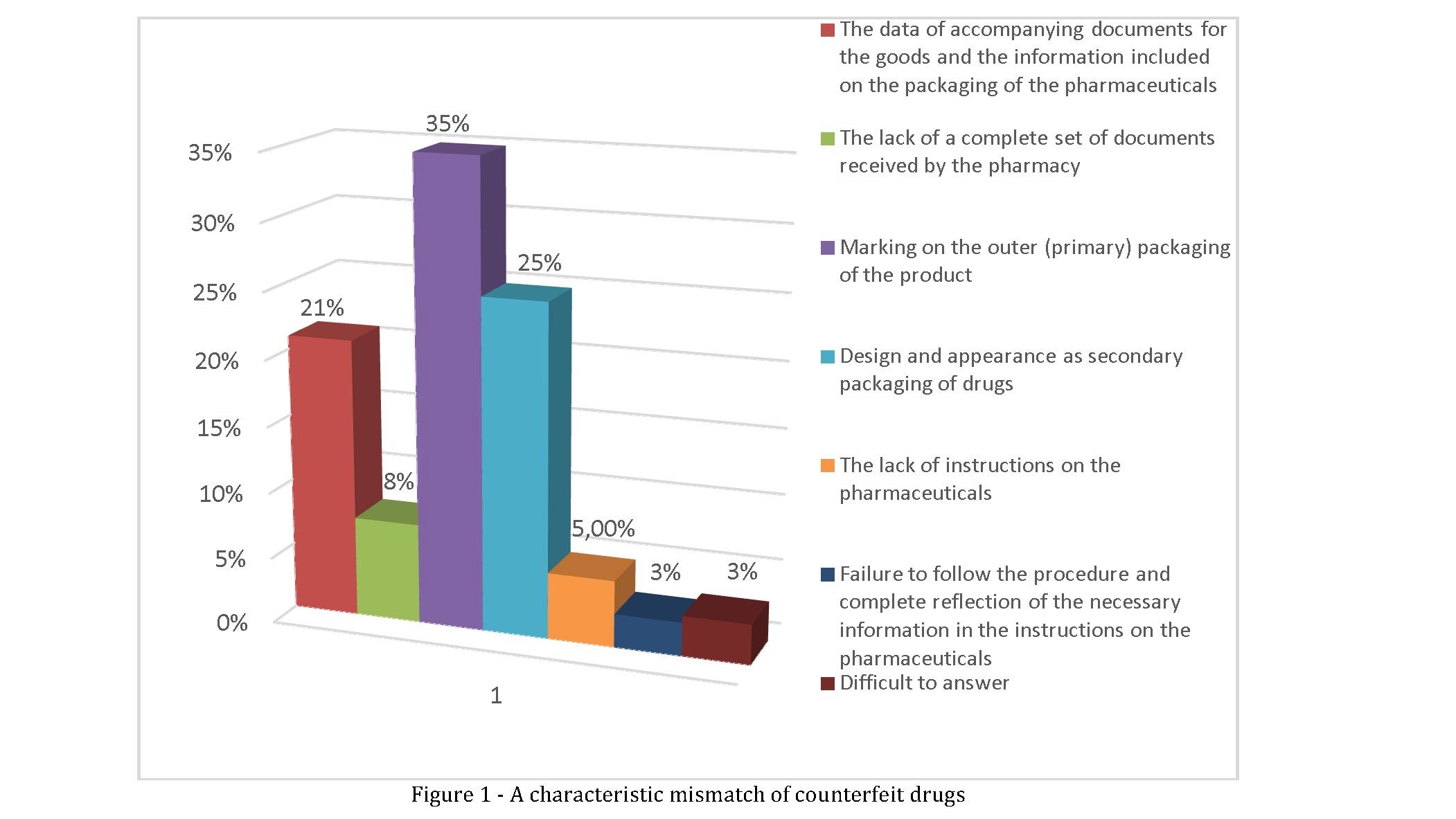
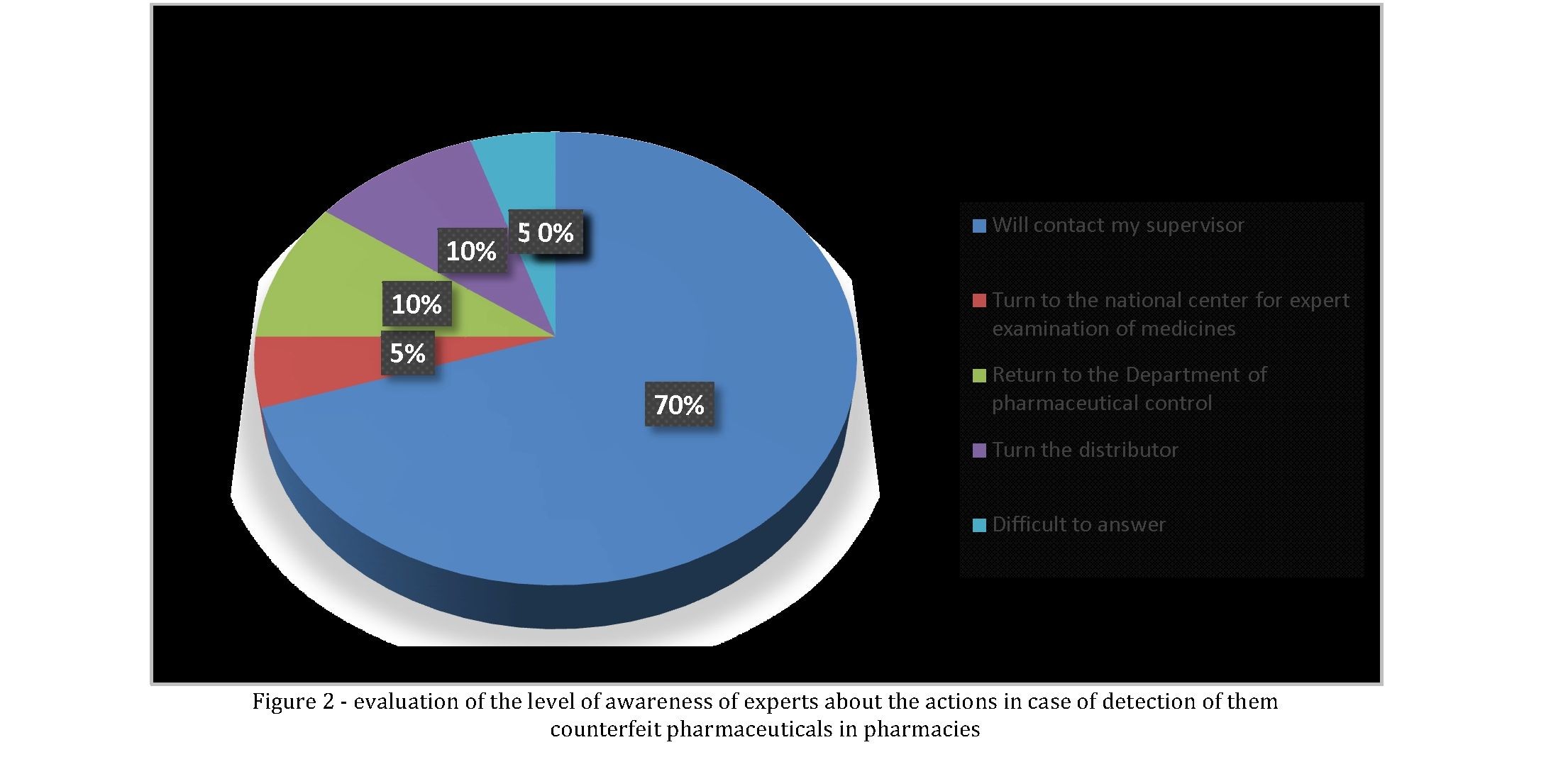 Table 1 - Ranking of contributing factors, according to experts, falsification of pharmaceuticals in Kazakhstan
Table 1 - Ranking of contributing factors, according to experts, falsification of pharmaceuticals in Kazakhstan
Dale by a considerable margin followed such answers as "the Lack of competence of specialists working in the state system of ensuring the quality of the pharmaceuticals coming on PM RK" (every fourteenth Respondent) and "Imperfect legislative and regulatory framework governing the turnover of drugs in the Republic of Kazakhstan" (one seventeenth). Less likely, according to experts affect the falsification of pharmaceuticals such well known factors as their social relevance and commercial appeal. Thus, we can say that the vast majority of experts believe that to the greatest degree of falsification of pharmaceuticals in Kazakhstan contributes to the lack of effective quality control over drugs at all stages of the supply chain in the system of pharmaceutical provision for the population is especially observed in the stage of importation of pharmaceutical products into the country. This is a complex problem requiring to solve a wide range of professionals working not only in the pharmaceutical industry.
Next, we faced the task to determine the level of awareness of experts about the correctness of their actions in case of detection of them counterfeit pharmaceuticals in pharmacies. To the question "Where or to whom You would appeal if found falsification of pharmaceuticals in the pharmacy?" respondents were asked to note these options: "Turn to your immediate supervisor"; "Appeal to the hotline of the National center of examination of medicines, products of medical purpose and medical equipment" (NCELS) of the Ministry of health of the Republic of Kazakhstan"; "Addressed to Department of Committee of control of medical and pharmaceutical activities of the corresponding territorial subordination"; "Turn to a distributor that has implemented this PL the pharmacy"; "unsure" (figure 2). Optimistic is the fact that only 14 people or 4.64 percent had difficulty answering this question. The vast majority of respondents indicated the answer "Turn to your immediate supervisor" (of 77.48%). Thus, more than three fourth of the respondents consider it necessary to inform the management of the pharmacy in case of detection of counterfeit goods, which seems the correct and logical action. This starts next, a mechanism of phase-state control over the quality of drugs in circulation on PM.
Already in the purview of the management of the pharmacy are such actions as "return to the Department of Committee of control of medical and pharmaceutical activities of the corresponding territorial subordination"". It should be noted that in accordance with the requirements of laws and regulations
governing the circulation of pharmaceuticals in the Republic of Kazakhstan in case of detection of counterfeit pharmaceuticals that is head of the pharmacy institution is obliged to immediately report this fact to the Department of Committee of control of medical and pharmaceutical activities of the corresponding
|
Possibleanswers (factors) |
Results thesurvey |
|
|
Numberofexperts /% |
A ratingfactor |
|
|
"Ineffective organization of quality control of imported to Kazakhstan pharmaceutical products by the competent state bodies" |
119/39,40 |
I |
|
"Defects in the organization of work of the competent authorities governing the circulation of pharmaceuticals in Kazakhstan" |
72/23,84 |
II |
|
""Lack of strict sanctions in case of detection of falsification of pharmaceutical products and its illegal and illegitimate implementation on PM RK" |
64/21,19 |
III |
|
"Low level of harmonization of state bodies responsible for the implementation of quality pharmaceutical services to the population of Kazakhstan" |
51/16,89 |
IV |
|
"The low level of efficiency of state regulation on PM in General" |
43/14,24 |
V |
|
"The lack of competence of specialists working in the state system of ensuring the quality of the pharmaceuticals coming on PM RK" |
21/6,95 |
VI |
|
"An imperfect legal and regulatory framework governing the turnover of pharmaceuticals in Kazakhstan" |
18/5,96 |
VII |
|
"High social value of pharmaceuticals for health and society" |
15/4,97 |
VIII |
|
"The high commercial attractiveness of pharmaceuticals for fraud" |
11/3,64 |
IX |
territorial subordination" Department of pharmaceutical control [7,13].
At the end of the questionnaire, respondents were asked to mention the most important, in their opinion focus on the fight against illicit trafficking of counterfeit products on the domestic PM. On the proposed options of answers were obtained the following data:
- "Strengthen the specialized work of the internal Affairs bodies on the ground" (90 experts or 29,80%of the total number of respondents);
- "To toughen criminal liability for fraud and illegal sale of counterfeit pharmaceuticals in Kazakhstan" (121 respondents or 40,06%);
- "The introduction and consolidation of the legislative and normative level, the Institute of "authorized person" responsible for the quality of the drugs coming in and dispensed from the pharmacy LP" (94 respondents or of 31.13%);
- "Implement education and training programs for pharmaceutical professionals to prevent the illegal trafficking of counterfeit pharmaceuticals in the RK" (6 people or 1.99%);
- "To expand the powers of the state bodies, controlling the quality of pharmaceuticals in the country" (45 expert or 14,90%);
- "To create and implement educational programs for people in order to raise awareness among people about fake drugs on FR" (9 respondents of 2.98%);
- "Other" (56 respondents or 18.5%).
Among the answer options "Other" respondents cited such as "Strengthening international cooperation", "Increasing penalties for falsification of drugs", "Deprivation of the license to engage in a particular activity in the field of pharmaceutical security of the population", "Technical re-equipment control and analytical laboratories, Active cooperation with foreign pharmaceutical companies, representing their products in PM RK", etc..
The last question "do You Consider necessary the development of guidelines to identify counterfeit drugs and prevent their illicit trafficking at the wholesale and retail segment of the pharmaceutical market?", more than half of respondents (194 or 64,24%) answered Yes, you need it". 49 (16,23%) of experts answered "No, do not consider it necessary", and 59 (19,54%) experts said the answer "Difficult to answer". Analyzing the preferences of experts on the last two questions of the questionnaire, it is possible to make such a conclusion. The vast majority of experts believe that the most effective way to solve the problem of illegal trafficking of counterfeit drugs is in the tightening of criminal responsibility for fraud and illegal sale of drugs, and also in the legal plane. Until 2015 in Kazakhstan for falsifying PL provided only administrative responsibility. At the moment, article 323. "Treatment of falsified medicines, medical products or medical devices" of the Criminal Code provides imprisonment from 2 to 10 years of imprisonment [14]. It should be noted that at the moment the world's fight against counterfeit products in PM, there is a trend of gradual transition from the concept of increasing the punishment for falsification of the pharmaceuticals to the formation of social active model to prevent the unlawful production and distribution of counterfeit pharmaceuticals on PM [1,2,15]..
Conclusions:
- In the survey of pharmaceutical workers found that the vast majority (84,76%) of respondents are familiar with the concept of "falsified/counterfeit drug", and every fourth expert had experience in detecting counterfeit drugs in pharmacies.
- The experts noted that the most common fraud in PM RK are subjected to antimicrobial and antiviral drugs, the second place - cardiovascular drugs, and the third - antiulcer PL. Drugs of these groups was marked by 44 experts, which accounted for 55.0% of the total number of respondents who had experience of detection of counterfeit pharmaceuticals in its activities.
- Most important, according to experts, detection of counterfeit drugs are inconsistencies for parameters such as "Marking on the outer (primary) packaging of the goods", "Clearance condition and appearance of secondary packaging drugs" and "Data documents accompanying the goods and the information reflected on the packaging of the pharmaceuticals ". They celebrated 65 respondents that 81.25% of the number of experts with experience in detecting counterfeit drugs.
- It is established that the leading positions in the ranking of the factors contributing to illicit trafficking of counterfeit drugs took factors that have a direct relationship to the functioning of the state system of ensuring the quality of medicines in the Republic of Kazakhstan. In this case, the most important is the lack of effective state control over the quality of imported pharmaceutical products (119 expert or 39,40%).
- Positive is the fact that more than three fourth (of 77.48%) of experts in the detection of counterfeits will apply to his direct supervisor that meets the requirements of the current regulatory framework.
- Most experts believe that the most effective direction for addressing the problem of illicit trafficking of counterfeit pharmaceuticals in RK must be criminal and legal plane. Thus, in European countries is considered more efficient implementation of the complex preventive measures aimed at preventing the illegal production and distribution of counterfeit pharmaceutical products.
- Systematizing the results of the survey we can say that the most promising research in this direction will be the analysis of the factors that reduce the effective functioning of state mechanisms for quality control of pharmaceuticals in circulation. In addition, the majority of experts expressed the need to develop practical guidelines for detection of counterfeit drugs and prevention of illicit trafficking in the chain of distribution of drugs and in pharmacies.
We also believe that the most important direction for future research will be the analysis of various factors influence on the effective functioning of state mechanisms of control over the quality of drugs in circulation. In addition, it is necessary to develop practical guidelines for detection of counterfeit drugs and prevention of illicit trafficking in the pharmaceutical market of the Republic of Kazakhstan.
REFERENCES
- Ю.В. Подпружников, А.А. Ишмухаметов, А.С. Немченко и др. Хрестоматия фармацевтического качества. - М.: Группа Ремедиум, 2015. — 432 c.
- Eric Przyswa. Сounterfeit medicines and criminal organizations-Режим доступа к https://www.researchgate.net/profile/Eric_Przyswa
- Юмашева И. П. Фармацевтический рынок: проблема фальсификации лекарственных средств // Вестник ТГУ - 2011. - №3. - С. 897-901.
- Jessica Krüger Anti-Counter feitingin Global Pharma covigilance // Dessirt. - 2015. - №1. - P. 5-6.
- Резвых Ю. А. Мероприятия по предотвращения поступления недоброкачественных и фальсифицированных лекарственных средств в медицинские организации // Менеджмент в здравоохранении. - 2014. - №6. - С. 31-36.
- Почкун А. Предотвращение оборота фальсифицированных лекарственных средств в странах Европы // Закон и жизнь - 2013. - № 8(4). - С. 204-208.
- О мерах по предотвращению обращения фальсифицированной / контрафактной медицинской продукции в Казахстане - Режим доступа к https://www.zakon.kz/4640337-o-merakh-po-predotvrashheniju.html
- Зординова К. А. Проблема фальсифицированных и субстандартных лекарственных средств в мире и Казахстане // Весник КазНМУ. - 2015. - №1. - С. 459-465.
- Бегалиев Е. Н. К проблеме фальсифицированных лекарственных препаратов в Республике Казахстан // Известия ВУЗОВ Кыргызстана. - 2014. - №2. - С. 32-35.
- Бердимуратова Г. Д. Международный опыт борьбы с распространением фальсифицированной медицинской продукции / Г. Д. Бердимуратова, Р.С. Кузденбаева // Фармация Казахстана - 2011. - №5(120). - С.10-11.
- Серикбаева Э. А. Фальсификация лекарственных средств :методы борьбы и пути решения // Вестник КазНМУ. - 2017. - № 2. - С. 288-290.
- Елисеева И. И. Общая теория статистики: Учебник. — 5-е изд., перераб. и доп. — М.: Финансы и статистика, 2004. - 656 с.
- Закон Республики Казахстан от 6.04.2015 года № 299-V ЗРК «О внесении изменений и дополнений в некоторые законодательные акты Республики Казахстан по вопросам здравоохранения» - Режим доступа к https://pharm.reviews/dokumenty/item/121-zakon-respubliki-kazakhstan-ot-6-aprelya-2015-goda-299-v-zrk
- Уголовный кодекс Республики Казахстан (сизменениями и дополнениямипо состоянию на 01.01.2018 г.)- Режим доступа к https://online.zakon.kz/Document/?doc_id=31575252
- 2017 global life sciences outlook.Thriving in today's uncertain market -Режимдоступакhttps://www2.deloitte.com/content/dam/Deloitte/cn/Documents/life-sciences-health-care/deloitte-cn-lshc-2017- life-sciences-outlook-en-170524.pdf