SUMMARY
Amygdala (AMG) comprises a heterogenous group of nuclei located in the medial temporal lobe (MTL) that is essential to acquisition and storage of memory of the conditioning experience and the expression of fear response such as freezing behavior, categorical representation of objects and interpreting facial expressions, emotion regulation; therefore, AMG can be considered as a behavioral modulator. The current memory paradigm states that the synaptic plasticity underlies the molecular basis of memory, so for example, enhanced or weakened synaptic transmission may promote Spinogenesis and synaptic strengthening's and thereby underlying the memory acquisition, consolidation, retrieval and loss, therefore, it may be stated that stress hormones such as glucocorticoids in time dependent fashion from the one hand promote learning and enhance performance but on the other hand lead to memory impairment through the modulation of synaptic plasticity in specific brain areas, which is reflected in U-shape model of memory formation. This review will describe some aspects of current memory formation mechanisms and how stress hormones and different neuropeptides such as neuropeptide S influence those mechanisms thereby leading to memory modulation.
Introduction: Stress is generally defined as any condition that disturbs physiological or psychological homeostasis of an organism which in turn recruits the autonomic nervous system, hypothalamic-pituitary - adrenal axis which in turn elevates the plasma levels of stress associated hormones glucocorticoids (GRs). Being hydro- phobic by nature, GRs can enter the brain were they bind to two subtypes of receptors the mineralocorticoid MRs and glucocorticoid GRs, characterized by a high and a low affinity for glucocorticoids respectively. Glucocorticoids exert their effect though mechanisms such as fast non- genomic and slow transcriptional regulation of responsive genes. The GRs receptors are localized in hippocampus, amygdala (AMG) and prefrontal cortex (PFC) which are believed to be significant for not only memory formation but also for behavior modulation [1]. This short review will describe the effects of stress on the brain regions, particularly, those mentioned above followed by the description of how these may affect the memory mechanisms; then recent research papers will be discussed in the context related to the topic of the review.
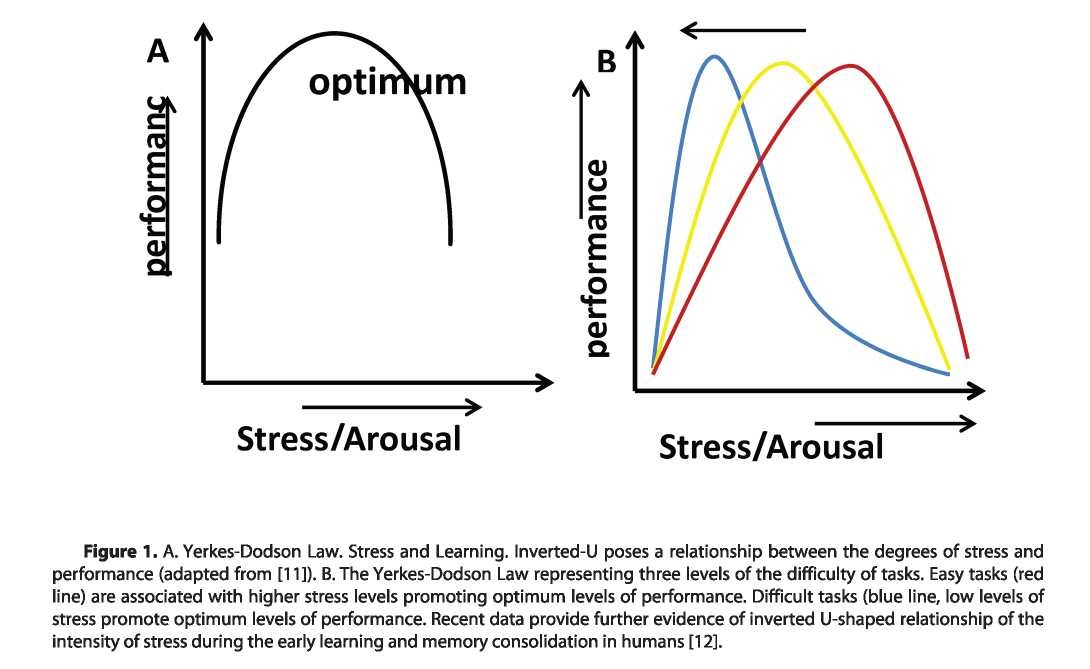
Stress and the Brain. Ample of electrophysiological, human f-MRI and animal behavioral studies [2] showed GRs in time dependent manner influence the excitability of AMG [3], hippocampus [4, 5] and PFC [6] and, so for example, short-term acute GRs elevation promotes the surface delivery of glutamatergic receptor through the recruitment of GRs inducible kinases and modulating protein trafficking [4] but chronic GRs administrations leads to the downregulation of glutamatergic receptors [7, 8]. The current memory paradigm states that the synaptic plasticity underlies the molecular basis of memory, so for example, enhanced or weakened synaptic transmission may promote spinogenesis and synaptic strengthening’s and thereby underlying the memory acquisition, consolidation and retrieval and loss, therefore, it may be stated that GRs in time dependent fashion from the one hand promote learning and enhance performance but on the other hand lead to memory impairment [9] through the modulation of synaptic plasticity in specific brain areas [10]. This was first described by inverted U shape relationship between the amount of stress and performance, described by Yerkes- Dodson law. See Fig.l.
Therefore, stress may promote survival of the organism by facilitating performance under certain conditions but it has detrimental effects when amount of stress hormones are elevated chronically. For example, chronic GRs elevation leads to the hypertrophy of AMG [13], atrophy Ofhippocampus and PFC [14]. These in turn might be associated with post-traumatic stress disorders, hyper-arousal and over-anxiety.
Memory modulation and related brain circuits.
AMG comprises a heterogenous group of nuclei located in the medial temporal lobe (MTL) that is essential to acquisition and storage of memory of the conditioning experience and the expression of fear response such as freezing behavior, categorical representation of objects [15,16] and interpreting facial expressions [17], emotion regulation [18]; therefore, AMG can be considered as a behavioral modulator. It receives inputs from the thalamus and the sensory cortex and sends outputs to thalamus, hippocampus and PFC. Behavioral studies show that GRs-induced AMG neuronal activity modulates hippocampal dependent memory acquisition and consolidation in time-dependent manner [19, 20]. So, for example, pharmacological studies showed that within a short time window GRs application to AMG enhances hippocampus and PFC based memory consolidation by influencing neuroplasticity in these regions; in contrast, chronic GRs applications leads to the atrophy of PFC which is involved in attention and associative learning [19]; therefore, GRs not only lead to attention deficits, impaired ability to Ieam but also to the impairment of working memory retrieval.
Memories are highly dynamic entities that are built in stages. After initial encoding, the “new” fragile memory trace is consolidated and stored for future retrieval. Hippocampus functions as a temporary store for recent information but permanent storage depends on a broadly distributed anatomically connected cortical networks. According to these models replay across hippocampal-cortical connections allows new memories be consolidated and be transferred into PFC where it is integrated with pre-existing cortical memories through the interaction with other brain regions for future memory retrieval [21]. Stress by means of glucocorticoids modulates AMG-Iiippocampus- PFC or AMG-PFC circuits thereby affecting on not only memory acquisition but also its consolidation. Interestingly, the effect depends on time, so for example, acute GCs may enhance hippocampal synaptic strengthening thereby heightening cognition and memory consolidation at the time of stress but on the other hand, chronic GRs elevation leads the hippocampal atrophy. Hippocampus is involved
in declarative (facts and events) in humans and spatial memory in rodents thus chronic GRs exposure leads to impaired recall of facts and events and navigational problems in animals. Interestingly, elevated GRs in the absence of fear and/or an intact amygdala are not sufficient to produce deficits in hippocampal processing [18] suggesting that AMG is crucial for stress induced memory modulation. Moreover, acute stress engages multiple-memory systems, so it shifts the brain systems in a manner that favor the consolidation of new information at the expense of other memory processes such as retrieval of previously learned events [22]. Stress outcomes are viewed under 2 approaches such as vertical and horizontal ones. The vertical view makes very specific predictions about the mechanism underlying stress effects on memory based on different brain regions connectivity; while the horizontal view, relying on electrophysiology of brain slices, predicts outcomes based on the time and duration of stress exposure rather than connectivity [23]. Thus, stress enhances memory if it is experienced within the context of the learning episode in a short time window but impairs if it is experienced outside the learning context on the chronic base [24]. It is well-established that acute stress may lead to strong painful memories such as PTSD but chronic stress may lead to over-anxious, hyper-arousal and debilitating conditions; therefore, understanding the exact anatomical as well as molecular signaling cascades will help to not only to treat
PTSD, over-anxiety but also improve the quality of life of people highly prone to stress.
Discussion: Recent work identified stress related endogenous neuropeptides, particularly neuropeptide S and its receptors (NPSRs), which are expressed mainly in AMG and PFC. NPS has anxiolytic-like effects and thus being considered as one of the candidates that plays crucial role in stress related response such as anxiety [25], memory consolidation [26], sleep and emotional processing [27]. The activation of NPS∕NPSR signaling cascade results in mobilization of ɛ ɑ and in increase of cellular excitability in hypothalamus [28], in PFC [29] and inhibition in AMG [25]; Interestingly, NPSR polymorphisms may account for different individual susceptibility to anxiety and reactivity to stress [30]. Therefore, NPS and NPSR may represent a good therapeutic target for future treatment of PTSD, outright panic attacks and memory enhancement. The recent paper by Okamura and colleagues showed that NPS and NPSRs may be critical for AMG-Iiippocampus mediated memory consolidation. Rodents’ behavioral studies based on pharmacological and genetic manipulations demonstrated that NPS and NPSR are critically involved in AMG-hippocampal mediated learning and memory. From the one hand, this behavioral study shows that manipulations of NPS∕NPSR had indeed in vivo effects but on the other hand pharmacological manipulations did not exclude off-target effects and not very physiological; moreover, behavior represents an integrated response of the organism based on genetic background, previous experience rather than individual neural circuits response; in addition, as amygdaloidal structure anatomically, neurochemically heterogeneous structure composed of more than 13 nuclei [15] that may have different modulatory functions through targeting different brain regions, so that it is very hard to predict what regions determine the integrated response. The authors used NSPR KO but the experiment could be further improved by KO NPSR particularly in amygdala and hippocampus particularly using the CRE-FLOX-Tecombinase technique. In addition, there is little evidence of the role of NPS∕NPSR in humans; so at the current state regardless being very promising NPS/ NPSR cannot be considered as potential treat any of cognitive disturbances mentioned above. Therefore, though this paper provides a piece of information that NPS∕NPSR plays a significant role in AMG-Wppocampus based memory consolidation, further studies should be conducted to provide direct evidence that NPS∕NPSR recruit AMG/ hippocampus∕cortical structures as well as a direct link between GRS and NPS∕NPSRs. Possible studies may involve optogenetics where specific dissection of amygdalahippocampus or amygdala-PFC circuits may shed some light on stress effects on memory mechanisms.
As it was described above, GRs modulates the engagement of multiple memory systems in the learning however very little is known about exact functional (temporal connection of related brain areas) and effective (direct effect of one neuronal system on to another) connectivity [30, 31]. Recently developed neuromodulation technique such as optogenetics may help to establish causal relationship between stress and behavior. In general, optogenetics is a coupling light and cellular behavior by means of the use of opsins, which are membrane - bound light-driven ion channels, in intact tissues or even in freely behaving animals. One of the main advantages is that specific opsin-expression in the neurons of particular interest allows getting high temporal and spatial precision in contrast to electrophysiological stimulations. Opsins have fast onset kinetics on the order of milliseconds which allows using high frequencies of millisecond-lasting flashes of light which in turn provides reliable evoked action potential and thus reliable fast readouts; however, these are balanced by fast deactivation, channel поп-specificity, difficulties in the expression in particular neurons as well as possible interventions with the normal cellular physiology [32, 33]. It should be noted that little is known about the physiology of opsins, however, recent work by Kato and colleagues showed crystal structures of Channelrhodopsins (ChRs) providing a basic working model of the channels gating. This may have important implications as it may represent future tool to generate optically controlled genetically encoded specific reporters of neuronal activity. So, for example, it was shown that CREB is critically involved in learning [34], so if to use chimeras of light-sensitive part of ChRs and specific proteins this may provide precise biochemical and cellular specificity. Crystal structure of C1C2 (chimeric between CHRl and CHR2 from Chla- mydomonas reinhardtii) at 2.3 Å resolution was obtained from fully dark-adapted C1C2 by multiple anomalous dispersion (MAD) method. MAD is a technique used in X-ray crystallography based on the absorption of X-rays of certain wavelength followed by re-emit after a delay inducing a phase shift [35]. The phase shift determines solution for the phases that are further determined through diffraction data in order to get the structure of the protein of interest. The resolution of ChRs crystallography is 2.3 Å suggesting that some small errors can be present in the suggested structure, though the structure of folds and surface loops can be considered as very close to the natural ones; moreover, at this resolution water molecules and small ligands can be visualized and thus the provided working model of ChRs might be reliable. Therefore, this structure might be further used synthesize chimeric proteins to obtain specific to amygdala reporters, for example, NPSRs. Nevertheless, optical modulations of neuronal activity seems to be very promising however further research on the exact opsin physiology should be done.
To conclude, this review described some of effects of stress on the brain regions that are believed to be critically involved in memory mechanisms. Understanding of exact molecular mechanisms underlining memory formation and consolidation are under extensive research. Several neuropeptides may be playing important roles in this process. Nonetheless, behavior represents a complex of genetic background, experience and other modulatory aspects, which in turn determine the very complex process such as memory.
REFERENCES
- Krugers H.J., Hoogenraad С.C., Groc L. (2010) Stress hormones and AMPA receptors trafficking in synaptic plasticity and memory. Nat Rev Neurosci 11:675-681.
- De Quervain DJF, Roozendaal B., Nitsch R.M., McGaugh J.L., Hock C. (2000) Acute cortisone administration impairs retrieval of long-term declarative memory in humans. Nat Neurosci 3:313-314.
- Roozendaal B., McEwen B.S., Chattarji S. (2009) Stress, memory and the amygdala. Nat Rev Neurosci 10:423- 433.
- Yuen E.Y., Liu W., Karatsoreos I.N., Ren Y, Feng J., McEwen B.S., Yan Z. (2011) Mechanisms for acute stress- induced enhancement of glutamatergic transmission and working memory. Mol Psychiatry 16:156-170.
- Waddell J., Bangasser D.A., Shors T. J. (2008) The Basolateral Nucleus of the Amygdala is Necessary to Induce the Opposing Effects of Stressful Experience on Learning in Males and Females. J Neurosci 28 (20): 5290-5294.
- Liu W, Yuen E.Y., Yan Z. (2010) The Stress Hormone Corticosterone Increases Synaptic α-Amino-3-hydroxy- 5-methyl-4-isoxazolepropionic Acid (AMPA) Receptors via Serum and Glucocorticoid-Inducible Kinase (SGK) Regulation of the GDI-RAB4 Complex. J Biol Chem 285 (9):6101-6108.
- Griggs E.M., Young E.J., Rumbaugh G., Miller C.A. (2013) MicroRNA- 182 Regulates Amygdala - Dependent MemoryFormation. JNeurosci 33 (4): 1734-1740.
- Roozendaal B., Okuda S., de Quervain DJF, McGaugh J.L. (2006) Glucocorticoids interact with emotion-induced noradrenergic activation in influencing different memory functions. Neurosci 138:901-910.
- Korz V., Frey J.U. (2003) Stress-Related Modulation OfHippocampal Long-Term Potentiation in Rats: Involvement OfAdrenal SteroidReceptors. JNeurosci 23 (19):7281-7287.
- Ivy A.S., Rex C.S., Chen Y, Dube C., Maras P.M., Grigoriadis, Gall G.M., Lynch G., Baram T.Z. (2010) Hippocampal Dysfunction and Cognitive Impairments Provoked by Chronic Early-Life Stress Involve Excessive Activation of CRH Receptors. J Neurosci 30 (39):13005-13015.
- Shors T.J. (2004) Learning During Stressful Times. Leam Mem 11:137-144.
- Salehi B., Cordero M.I., Sandi C. (2010) Learning under stress: The inverted-U-shape function revisited. Leam Mem 17:522-530.
- McEwen B.S., Gianaros P.J. (2011) Stress-and Allostasis-Induced Brain Plasticity. Annu Rev Med 62:431-45.
- Silva-Gomez A.B., Aguilar-Salgado Y, Reyes-Hemandez D.O., Flores G. (2013) Dexamethasone induces different morphological changes in the dorsal and ventral hippocampus of rats. J Chem Neuroanatom 47:71-78.
- Kuraoka K, Nakamura K (2012) Categorical representation of objects in the central nucleus of the monkey amygdala. EurJNeurosci 35:1504-1512.
- Domelles A, de Lima MNM, Grazziotin M, Presti-Torres J, Garcia VA, Scalco ES., Roesler R,, Schroder N. (2007) Adrenergic enhancement of consolidation of object recognition memory. Neurobiol Leam Mem 88:137-142.
- Phelps E.A., LeDoux J.E. (2005) Contribution of the Amygdala to Emotion Processing: From Animal Models to Human Behavior. Neuron 48: 175-187.
- Kim M.J., Loucks R.A., Palmer A.L., Brown A.C., Solomon K.M., Marchante A.N., Whalen P.J. (2011) The Stmctural and functional connectivity of the amygdala: From normal emotion to pathological anxiety. Behav Brain Res 223:403-410.
- Bouret S., Duvel A., Onat S., Sara S.J. (2003) Phasic Activation of Locus Ceruleus Neurons by the Central Nucleus of the Amygdala. J Neurosci 23(8): 3491-3497.
- Chavez C.M., McGaugh J.L., Weinberger N.M. (2013) Activation of the basolateral amygdala induces long-term enhancement of specific memory representations in the cerebral cortex. Neurobiol Leam Mem 101:8-18.
- Frankland P.W., Bontempi B. (2005) The Organization of recent and remote memories. Nat Rev Neurosci 6:119- 130.
- Schwabe L., Wolf O.T. (2012) Stress Modulates the Engagement of Multiple Memory Systems in Classification Learning. JNeurosci 32 (32): 11042-11049.
- Schwabe L., Joels M., Roozendaal B., Wolf O.T., Oitzl M.S. (2012) Stress effects on memory: An update and integration. Neurosci and Biobehav Rev 36:1740-1749.
- Schwabe L., Wolf O.T., Oitzl M.S. (2010) Memory formation under stress: Quantity and quality. Neurosci and Behav Rev 34: 584-591.
- Domschke K., ReifA., Weber H., Richter J., Hoholf C., Ohrmann P., PedersenA., Bauer J., Suslow T., Kugel H., Heindel W, Baumann C., Klauke B., Jacob C., Maier W, Fritze J., Bandelow B., Krakowitsky P., Rothermundt M., ErhardtA., Binder E.B., HolsboerF., Gerlach A.L., KircherT., Lang T., Alpers G.W., StrohleA., FehmL., Gloster A.T., Wittchen H-U, Arolt V, Pauli P., HammA., Deckert J. (2011) Neuropeptide S receptor gene-converging evidence for a role in panic disorder. Mol Psychiatry 16:938-948.
- Okamura N., Garau C., Duangdao D.M., Clark S.D., Jiingling K., Pape H-C, Reinscheid R.K. (2011) Neuropeptide S Enhances Memory During the Consolidation Phase and Interacts with Noradrenergic Systems in the Brain. Neuropsychopharm 36:744-752.
- Schank J.R., Ryabinin A.E., Giardino W.J., Ciccocioppo R., Heilig M. (2012) Stress-Related Neuropeptides and Addictive Behaviors: Beyond the Usual Suspects. Neuron 76: 192-208.
- Yoshida K., Kim J., Nakajima K., Oomura Y., Wayner M.J., Sasaki K. (2010) Electrophysiological effects of neuropeptide S on rat ventromedial hypothalamic neurons in vitro. Peptides 31:712-719.
- Si W, Aluisio L., Okamura N., Clark S.D., Fraser I., Sutton S.W., Bonaventure P., Reinscheid R.K. (2010) Neuropeptide S stimulates dopaminergic neurotransmission in the medial prefrontal cortex. J Neurochem 115: 475-482.
- Franklin T.B., Saab B.J., Mansuy I.M. (2012) Neural Mechanisms of Stress Resilience and Vulnerability. Neuron 75:747-761.
- Kim J.J., Diamond D.M. (2002) The stressed hippocampus, synaptic plasticity and lost memories. Nat Rev Neurosci 3:453-462.
- Tye K.M., Deisseroth K. (2012) Optogenetic investigation of neural circuits underlying brain disease in animal models. NatRevNeurosci 13: 251-266.
- MeiY., Zhang F. (2012) MolecularTools and Approaches for Optogenetics. Biol Psychiatry 71:1033-1038.
- Middei S., Spalloni A., Longone P., Pittenger C., O’Mara S.M., Marie H., Ammassari-Teule M. (2012) CREB selectively controls learning-induced structural remodeling of neurons. Learning &Memory 19:330-336.
- Kato H.E., Zhang F., Yizhar O., Ramakrishnan Charu, Nishizawa T., Hirata K., Ito J., AitaY, Tsukazaki T., Hayashi S., Hegemann P., Maturana A.D., Ishitani R, Deisseroth K., Nureki O. (2012) Crystal structure of the Channelrhodopsin light-gated cation channel. Nature 482:369-374.