Abstract
Genotoxicity is usually called the ability of a factor to cause damage to the genetic material of a cell, providing a mutagenic or carcinogenic effect. One of the most common methods for diagnosing genotoxicity is the micronucleus test. Chemical and physical factors of the environment can induce the appearance of a micronucleus at different stages of the cell's life cycle. Usually, a micronucleus is a rounded structure located at the periphery of the cell, and is stained in the color of chromatin. It is known that most often micronuclei are formed from chromosomes that do not disperse in time to the poles of the cell (acentric or dicentric). Nondisjunction can be caused by a defect in the division spindle, damage to the chromosomes themselves, extrusion of chromatin, or cell death. The appearance of a micronucleus in a cell can have both clastogenic and aneugene effects. Genotoxic factors of a chemical nature, possessing nucleophilic properties, bind to a DNA molecule and contribute to the damage to its native structure, which leads to mutations of genes, in particular, those responsible for the formation of tubulin filaments or centromeric proteins. Physical genotoxicants contribute to the appearance of single- and doublestranded breaks in the DNA molecule, disruption of repair processes. Thus, the action of various genotoxic environmental factors increases the likelihood of the appearance of such a structure as a micronucleus in the cell.
About 130 years ago, the first mention of the micronucleus as a cellular structure appeared in the scientific literature. For the first time this was stated in 1890 by Howell [1], who discovered them in the cells of cats in which anemia was induced. According to his observations, such formations were nuclear fragments that were not eliminated from the mature erythrocyte during erythropoiesis, since the process took place in an accelerated mode in order to regenerate cells damaged by anemia. A little later (1899) Schmauch found similar formations in blood cells of both healthy cats and those exposed to toxic substances, but their frequency was very different, prevailing in damaged erythrocytes. In 1902-1908 Schmidt, Morris, and Jolly showed that such bodies can be found in the erythrocytes of embryos, newborns, people suffering from pernicious anemia, as well as cells of the peripheral blood of animals. Papenheim (1907, 1909) succeeded in proving the nature of Howell-Jolly bodies, experimentally showing that they are always stained in the color of chromatin, which allows them to be distinguished from the granularity of erythrocytes. All of the above authors postulated the peripheral location of micronuclei and indicated that such formations do not occur in the center of the cell, in the immediate vicinity of the nucleus, as the cell tries to push them out.
Currently, the micronucleus test is one of the most widespread and widely used methods for analyzing the genotoxic influence of environmental factors. The first steps towards its development as a marker of genotoxicity can be found in 1959, when Evans et al. [2] recorded the presence of micronuclei in bean root cells exposed to gamma radiation. The authors determined that 60% of the chromosomal fragments contributed to the formation of micronuclei.
In animals the method was first used in the 1970s by Boiler and Schmid. They studied the effect of trelimone on the red blood cells and bone marrow cells of a Chinese hamster. They are considered to be the founders of using the micronucleus counting method as an indicator of genotoxicity. Then the method underwent changes, over the years, scientists have used it on various objects. Among the genotoxic environmental factors, chemical agents have always occupied a special place, as one of the most common in the habitat of animals. Thus, in 1979, Cole et al. [3] and King and Wild studied the induction of micronuclei in liver and peripheral blood cells of embryos of mice whose mothers were exposed to chemical genotoxicants. And in 1980, MacGregor et al. [4] proposed to count micronuclei in erythrocytes of mice, suggesting that the use of erythrocytes, in contrast to bone marrow cells, makes it possible to diagnose chronic exposure to a chemical agent. They determined that in the erythrocytes of mice micronuclei are able to exist for a long time, while in human cells these structures are quickly eliminated due to the active work of the spleen. In 1981-1983, Lahdetie et al. and Tates et al. [5-6] used male germ cells as an object of study to study the degree of genotoxic effect of chemical influences of the environment. In 1983, the United States Environmental Protection Agency (US-EPA), as part of a program to study the genotoxicity of chemicals, published the results of a large number of works, including studies using the micronucleus test. In the same year, the International Chemical Safety Program initiated a program to evaluate substances for carcinogenicity. The micronucleus test has shown the ability to most accurately assess these properties of chemical compounds. Gradually, other countries began to develop their own protocols for assessing the safety of chemicals and the micronucleus test was included in a battery of tests to assess their genotoxic effects.
Over the next 40 years, the method underwent changes that increased its information content, reliability and contributed to a larger and faster analysis of large amounts of data.
New approaches to the analysis of test results have emerged using fluorescent dyes and flow cytometry. In the modern era of computer technology, there are electronic programs (MetaSystem) that allow automating the entire process of analyzing the data obtained, using both different cells and different versions of the method.
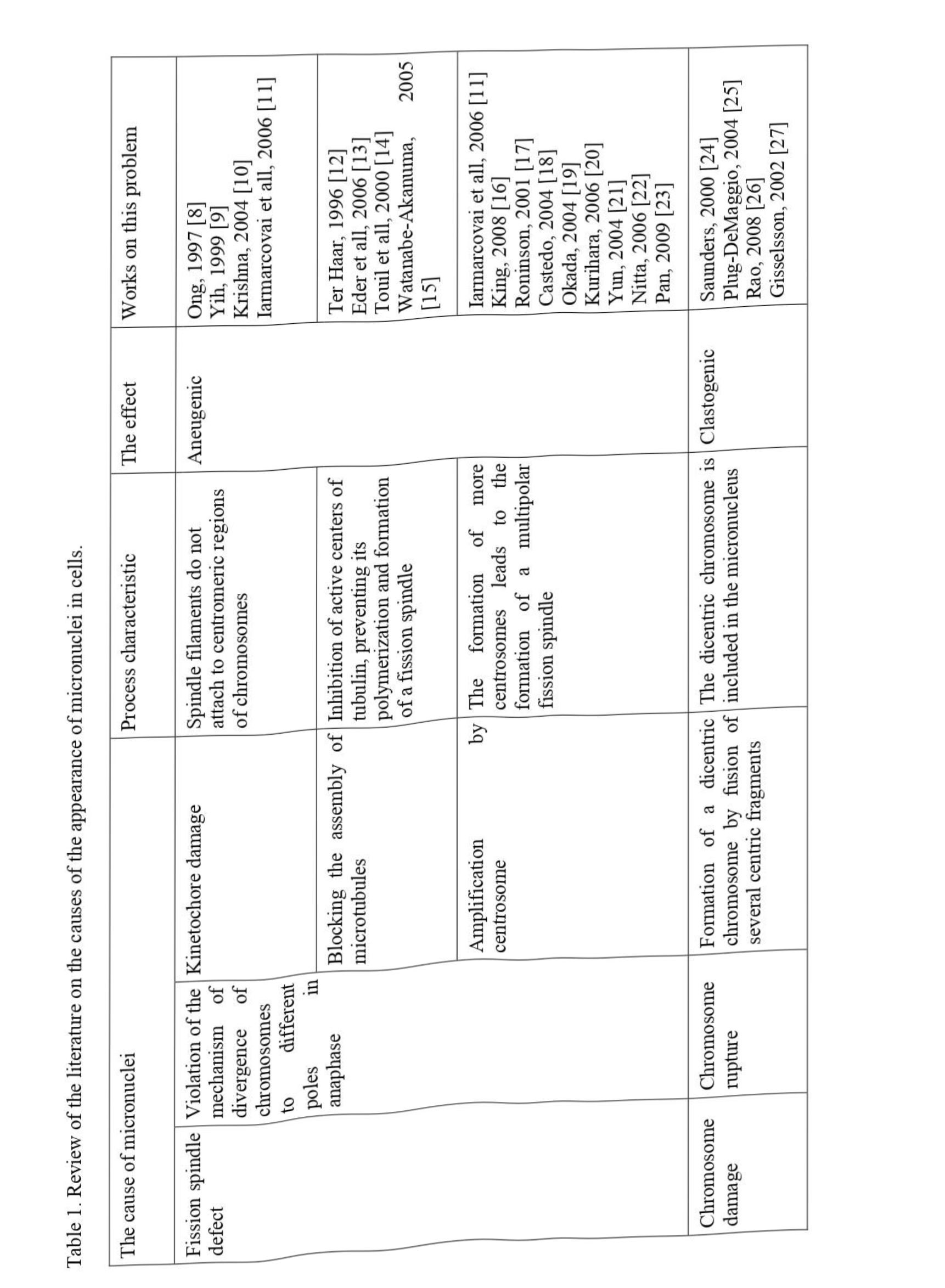
According to the PubMed portal, the number of publications describing the use of the micronucleus test for various purposes is increasing annually, and since 1975 there have been more than 9700 such publications, of which about 3500 concern the effect of chemical agents on a living organism. However, there are very few works devoted to the problem of micronucleogenesis. To date, several possible reasons for the appearance of micronuclei in cells are discussed in the literature; we have summarized these data in Table 1.
The reasons for the formation of micronucleus can be classified according to several criteria. These include, for example, the place of origin in the life cycle (in interphase or during mitosis), the nature of biochemical abnormalities (disorders in the structure of cellular proteins or DNA), the physiological state of the cell (healthy or tumor). However, all authors agree that chemical agents of the environment can equally provoke all of the listed reasons for the occurrence of micronucleus.
The life cycle of a cell is also called mitotic, however, it consists not only of the process of cell division itself, but also of preparatory stages, such as Gl, S, G2 - periods. The processes occurring during these periods are of no less importance in the vital activity of cells than the divergence of chromosomes in mitosis. The overwhelming number of deviations leading to the formation of micronuclei occur at the stage of mitotic anaphase. However, there are a number of works that indicate the possibility of separating genetic material from the nucleus and at the interphase stage. This phenomenon was named in Russian-language works "chromatin extrusion", in English-language - "nuclear buds" (caudate nucleus). A similar mechanism is found in healthy cells as a way to get rid of excess genetic material that arose in the course of excessive replication of extrachromosomal elements or amplification of oncogenes; there is evidence that the cell is thus rid of homogeneous disomy. When exposed to chemical mutagens, the number of such micronuclei increases significantly.
In the literature, biochemical disorders leading to abnormalities at different stages of the cell cycle are most extensively described. Chemical mutagens that cause disturbances in centromeric regions of DNA block the possibility of attachment of kinetochores and, as a consequence, there is a mismatch of the chromosome segregation apparatus [7].
In the event of mutations in the genes responsible for the synthesis of centromeric proteins, such as CENP-A, CENP-H, CENP-E, a decrease in the amount of these substances is observed in the cell, and their other arrangement. This situation leads to the impossibility of connecting the chromosomes with the filaments of the spindle of division, which entails a lag of the damaged chromosome when it diverges to the cell poles.
It has long been known that substances such as colchicine, colcemide, vinblastine, wikristine, and alkylating agents are capable of inhibiting the assembly of microtubules into the fission spindle by preventing the polymerization of tubulin monomers. The absence of a full-fledged spindle of division or its inferiority, contribute to the emergence of residual chromosomal elements that have not shifted towards the poles.
A similar outcome can occur in the case of the opposite situation, that is, in the presence of an excessive number of centrosomes. In a healthy cell, such disorders are promptly detected by proteins, which provide a check of the usefulness of the genetic material during checkpoints (checkpoints). These proteins include the products of the genes CHKl, CHK2, ATM, ATR, PLK, etc. Chemical mutagens can damage the listed genes, which is reflected in the formation of a multipolar spindle, and deviations in the uniformity of chromosome separation.
Thus, the consequences of the action of chemical mutagens described above lead to a violation of chromosome segregation. As a result of this phenomenon, small fragments of genetic material are left out of work. Subsequently, they can be surrounded by a nuclear membrane and then turn into micronuclei, providing aneugenic effect.
Another reason for the appearance of such fragments of chromosomes may be the appearance of double-strand breaks in DNA. In this case, either dicentric or acentric chromosomes are formed. In the first case, breaks occur in two chromosomes, which then join. In metaphase, the spindle filaments attach to both centromeres of the resulting structure, and then, at the anaphase stage, the dicentric chromosome either loses the ability to divide into chromatids and remains entirely in the center of the cell, or forms a chromatid bridge. The fragment remaining after the rupture of both chromosomes does not have a centromeric region, therefore it is called the acentric chromosome.
From the formed structures, micronuclei are then formed, which can include both whole chromosomes, in the case of non-divergence of dicentrics, and individual fragments of chromosomes, in the case of chromatid bridges and acentrics. Such micronuclei have a Clastogenic effect.
Despite the fact that the vast majority of double-strand breaks are induced by physical factors such as radiation, according to the literature, chemical factors are also capable of introducing breaks in DNA. Thus, for example, in mouse cells, using the DNA comet method, data were obtained indicating the ability of some pesticides (chlorpyrifos, penconazole, dimethoate, Iambda-Cyhalothrin, Auopyram, ephion, pendimethalin, mancozeb) to provoke DNA damage, manifested in its rupture.
To be fair, it should be said that DNA breaks that form dicentrics and acrocentrics, with normal formation of the fission spindle, can lead to an imbalance in chromosome segregation, to the formation of aneuploidy, but without the formation of a micronucleus. If such anomalies are combined with the damage to the fission spindle described above, the likelihood of micronucleus formation increases significantly. For example, the formation of a dicentric chromosome, in combination with impaired tubulin polymerization or abnormalities in the development of kinetochores, leads to the fact that fission spindle filaments either do not form or do not attach to centromeres.
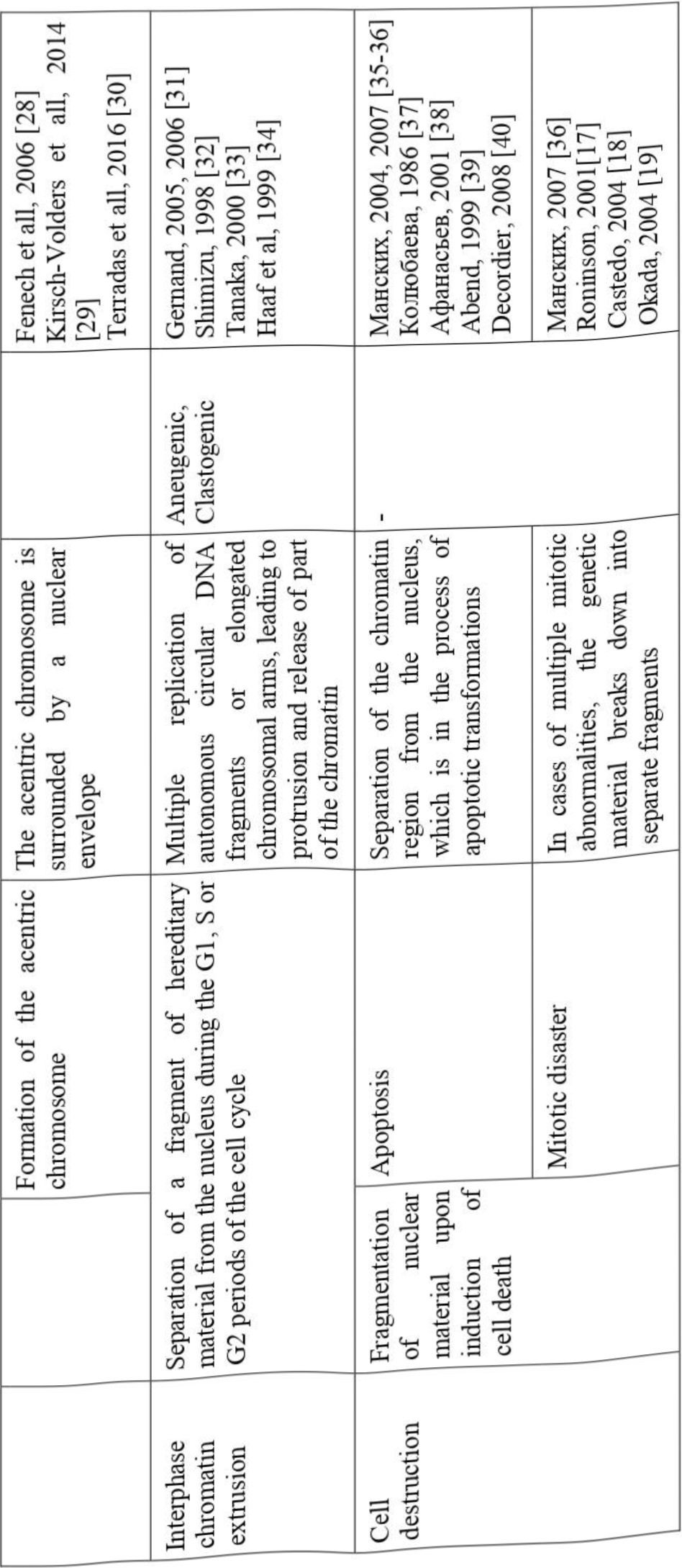
All this contributes to the formation of micronuclei containing a fragment of genetic material surrounded by a nuclear membrane.
When a cell is faced with the influence of environmental factors that it is unable to cope with, it enters a cascade of processes leading to its death. It is known that during apoptosis, it is the nucleus that undergoes the greatest changes, during which several fragments of genetic material may appear, which are separated from the main nucleus. Such structures can also be called micronuclei, although their nature is fundamentally different from the above-described micronuclei, which are formed in healthy and undamaged cells. Apoptotic micronuclei are usually multiple, unequal in size, have a different chromatin structure, so it is not difficult to distinguish them even externally, moreover, in a cell undergoing apoptosis, there are other characteristic features that are absent in classical micronucleogenesis. However, research on this kind of micronuclei has led to the discovery of some similarities. For example, some of the participants in apoptosis, such as caspase 3, protein kinase, endonuclease, are also important in the formation of micronuclei by the mechanisms described above [40].
When choosing a micronucleus test as a method for determining the genotoxic effect of an environmental factor, it is necessary to take into account its advantages and disadvantages. The indisputable advantages of this method include:
- A wide range of cell types used as research objects. Red blood cells and bone marrow cells are traditionally used. In modern modifications, there are works on the use of lymphocytes, fibroblasts, buccal epithelium cells, urinary stem cells.
- Availability, ease of implementation. For the basic version of the method from the equipment, glass slides, Giemsa stain and May-Grunwald fixative are sufficient.
- Mobility. The method can be carried out in the field, which significantly increases its efficiency.
- Fast receipt of research results. Conclusions about the genotoxicity of the factor can be made within 24 hours after collecting the biological material.
Among the disadvantages are the following:
- Collection of biological material for this method is invasive (blood, bone marrow, etc.)
- The possibility of fixing only the presence of damage to the genetic material, without specifying its nature and mechanism of occurrence.
However, the second drawback is compensated by the use of additional methods, selected by the researcher depending on the goals.
The analysis of the possible causes of micronucleation and the indicated advantages of the method led us to the conviction of the need to use this method to determine the genotoxic effect of prohibited and unused pesticides on farm animals kept in the territory of Almaty region.
References
- Howell W. Observation upon the occurrence structure and function of the giant cells of the marrow// J. Morphol. - 1890. - Vol. 4., Nol. - P. 57-63
- Evans H.J., Neary G.J., Williamson F.S. The relative biological efficiency of single doses of fast neutrones and g-rays on Vicia faba roots and the effects of oxygen. Part II. Cromosome damage: the production of micronuclei // International Journal OfRadiation Biology and Related Studies in Physics, Chemistry and Medicine. - 1959. - Vol.3. - P. 216-229. doi: 10.1080/09553005914550311
- Cole R.J., Taylor N., Cole J., Arlett C.F. Short-term test for transplacentally active carcinogens. 1. Micronucleus formation in fetal and maternal mouse erythroblasts // Mutation ResearchZFundamental and Molecular Mechanisms of Mutagenesis. - 1981- Vol. 80. - P. 141- 157. doi: 10.1016/0027-5107(81)90184-6.
- MacGregor J.T., Wehr C.M., Gould D.H. Clastogen-induced micronuclei in peripheral blood erythrocytes: The basis of an improved micronucleus test // Environmental Mutagenesis. - 1980. - Vol.2. -P. 509-514. doi:10.1002/em.2860020408
- Lahdetie J, Parvinen M. Meiotic micronuclei induced by X-rays in early spermatids of the rat // Mutation ResearchZFundamental and Molecular Mechanisms of Mutagenesis. - 1981. - Vol.81, NoL -P. 103-115. doi: 10.1016/0027-5107(81)90091-9.
- Tates A.D., Dietrich A.J.J, de Vogel N., Newteboom I., Bos A. A micronucleus method for detection of meiotic micronuclei in male germ cells of mammals // Mutation Research Letters. - 1983. - Vol.121, Nol31. - P. 131-138. doi: 10.1016/0165- 7992(83)90111-2.
- Ильинских H.H., Ксенц A.C., Ильинских E.H., Манских В.Н., Ильинких И.Н. Микроядерный анализ в оценке цитогенетической нестабильности. - Томск: Издательство ТГПУ, 2011. - 312 с.
- Ong T., Liu Y., Zhong В.-Z., Jones W. G., Whong W.-Z. Induction of micronucleated and multinucleated cells by man-made fibers in vitro in mammalian cells // Journal of toxicology and environmental health. - 1997. Vpl.50, No4. - P. 409-414. doi: 10.1080/009841097160447
- Yih L.H, Lee T.C. Effects of exposure protocols on induction of kinetochore- plus and -minus micronuclei by arsenite in diploid human fibroblasts // Mutation Research Letters. - 1999. - Vol.440, NoL - P. 75-82. doi:10.1016/s!383-5718(99)00008-x
- Krishna G., Fiedler R., Theiss J.C. Simultaneous analysis of chromosome damage and aneuploidy in cytokinesis-blocked V79 Chinese hamster lung cells using an antikinetochore antibody/ZMutation Research Letters. - 1992. - Vol. 282, No2. - P. 79-88.
- Iarmarcovai G, Botta A, Orsiere T. Number of centromeric signals in micronuclei and mechanisms of aneuploidy//Toxicology Letters. - 2006. - Vol. 166(1), NoL -P.10. doi:10.1016/j.toxlet.2006.05.015
- Ernst ter Haar, Billy W. Day, Herbert S. Rosenkranz. Direct tubulin polymerization perturbation contributes significantly to the induction of micronuclei in vivo//Mutation ResearchZFundamental and Molecular Mechanisms of Mutagenesis. - 1996. - Vol. 350, No.2. -P. 331-337. doi:10.1 Ol6/0027-5107(95)00175-1
- Erwin Eder, Wolfgang Kutt, Christoph Deininger. In vitro induction of micronuclei by monofunctional methanesulphonic acid esters: Possible role of alkylation mechanisms//Chemico-Biological Interactions. - 2006. - Vol. 164, No. 1-2. - P. 76-84. doi: 10.1016/j.cbi.2006.08.020
- Touil N, Elhajouji A, Thierens H, Kirsch-Volders M. Analysis of chromosome loss and chromosome segregation in cytokinesis-blocked human lymphocytes: nondisjunction is the prevalent mistake in chromosome segregation produced by low dose exposure to ionizing radiation//Mutagenesis. - 2000. - Vol. 15, No.l. - P. 1-7. doi: 10.1093/mutage/l 5.1.1
- Watanabe-Akanuma M, Ohta T, Sasaki YF. A novel genotoxic aspect of thiabendazole as a photomutagen in bacteria and cultured human cells// Toxicology Letters. - 2005. - Vol.158, No3. - P. 213-219. doi:10.1016/j.toxlet.2005.03.013
- King RW. When 2+2=5: the origins and fates of aneuploid and tetrapioid cells//Biochimica et Biophysica Acta. - 2008. - Vol. 1786, No.l. - P. 4-14. doi: 10.1016/j.bbcan.2008.07.007
- Roninson I.B, Broude E.V, Chang B.D. If not apoptosis, then what? Treatment- induced senescence and mitotic catastrophe in tumor cells//Drug Resistance Updates. - 2001. - Vol.4, No.5. -P. 303-313. doi:10.1054/drup.2001.0213
- Castedo M., Perfettini J.L, Roumier T., Andreau K., Medema R., Kroemer G. Cell death by mitotic catastrophe: a molecular definition// Oncogene. - 2004. - Vol.23, No.16. -P. 2825-2837. doi: 10.1038/sj.one. 1207528
- Okada H, Mak T.W. Pathways of apoptotic and non-apoptotic death in tumour cells// Nature reviews. Cancer. - 2004. - Vol.4, No.8. - P. 592-603. doi:10.1038/nrcl412
- Kurihara D, Matsunaga S, Kawabe A, et al. Aurora kinase is required for chromosome segregation in tobacco BY-2 cells//The Plant Journal. - 2006. - Vol.48, No.4. -P. 572-580. doi :10.1 lll/j,1365-313X.2006.02893.x
- Yun C, Cho H, Kim S.J, et al. Mitotic aberration coupled with centrosome amplification is induced by hepatitis B virus X oncoprotein via the Ras-mitogen-activated protein/extracellular signal-regulated kinase-mitogen-activated protein pathway// MolecularCancerResearch. -2004. - Vol.2, No.3. - P. 159-169.
- Nitta T, Kanai M, Sugihara E, et al. Centrosome amplification in adult T-cell leukemia and human T-cell leukemia virus type 1 Tax-induced human T cells// Cancer Science. -2006. - Vol.97, No9. -P. 836-841. doi:10.UU/j.l349-7006.2006.00254.x
- Pan S.H, Tai C.C, Lin C.S, et al. Epstein-Barr virus nuclear antigen 2 disrupts mitotic checkpoint and causes chromosomal instability//Carcinogenesis. - 2009. - Vol.30, No.2. - P. 366-375. doi: 10.1093/carcin/bgn291
- Saunders W.S, Shuster M, Huang X, et al. Chromosomal instability and Cytoskeletal defects in oral cancer cells// Proceedings of the National Academy of Sciences. - 2000. - Vol.97, No.l. - P. 303-308. doi:10.1073/pnas.97.1.303
- Plug-DeMaggio A.W, Sundsvold T, Wurscher M.A, Koop J.I, Klingelhutz A.J, McDougall J.K. Telomere erosion and chromosomal instability in cells expressing the HPV oncogene 16E6// Oncogene. - 2004. - Vol.23, No.20. - P. 3561-3571. doi :10.1038/sj.one. 12073 88
- Xiaotang Rao, Yingyin Zhanga, Qiyi Yi, Heli Hou, Bo Xu, Liang Chu, Yun Huang, Wenrui Zhang, Michael Fenech, Qinghua Shi. Multiple origins of spontaneously arising micronuclei in HeLa cells: direct evidence from long-term live cell imaging// Mutation ResearchZFundamental and Molecular Mechanisms of Mutagenesis. - 2008. - Vol. 646, No. 1-2. - P. 41-49.
- Gisselsson D, Jonson T, Yu C, et al. Centrosomal abnormalities, multipolar mitoses, and chromosomal instability in head and neck tumours with dysfunctional telomeres// British Journal of Cancer. - 2002. - Vol.87, No.2. - P. 202-207. doi:10.1038/sj.bjc.6600438
- Fenech M. Cytokinesis-block micronucleus assay evolves into a "cytome" assay of chromosomal instability, mitotic dysfunction and cell death// Mutation ResearchZFundamental and Molecular Mechanisms of Mutagenesis. - 2006. - Vol. 600, No.1-2. - P. 58-66. doi:10.1016/j.mrfmmm.2006.05.028
- Kirsch-Volders M, Bonassi S, Knasmueller S, Holland N, Bolognesi C, Fenech MF. Commentary: critical questions, misconceptions and a road map for improving the use of the lymphocyte cytokinesis-block micronucleus assay for in vivo biomonitoring of human exposure to genotoxic chemicals-a HUMN project perspective// Mutation ResearchZRevi ews in Mutation Research. - 2014. - Vol.759. - P. 49-58. doi:10.1016/j.mrrev.2013.12.001
- Terradas M, Martin M, Genesca A. Impaired nuclear functions in micronuclei results in genome instability and Chromothripsis// Archives of Toxicology volume. - 2016. -Vol.90, No.ll. - P. 2657-2667. doi:10.1007/s00204-016-1818-4
- Gemand D, Rutten T, Varshney A, et al. Uniparental chromosome elimination at mitosis and interphase in wheat and pearl millet crosses involves micronucleus formation, progressive heterochromatinization, and DNA fragmentation//Plant Cell. - 2005. Vol.17, No.9. - P. 2431-2438. doi: 10.1105/tpc. 105.034249
- Shimizu N, Itoh N, Utiyama H, Wahl GM. Selective entrapment of extrachromosomally amplified DNA by nuclear budding and micronucleation during S phase// The Journal of Cell Biology. - 1998. - Vol. 140, No. 6. - P. 1307-1320. doi:10.1083/jcb.140.6.1307
- Tanaka T, Shimizu N. Induced detachment of acentric chromatin from mitotic chromosomes leads to their cytoplasmic localization at G(I) and the micronucleation by lamin reorganization at S phase// Journal of Cell Science. - 2000. - Vol.113, No.4. - P. 697-707
- Thomas Haaf, Elke Raderschall, Gurucharan Reddy, David C. Ward, Charles M. Radding, and Efim I. Golub. Sequestration of mammalian Rad51-recombination protein into micronuclei// The Journal of Cell Biology. - 1999. - Vol. 144, No.l. - P. 11-20.
- Манских B. H. Морфологические методы верификации и количественной оценки апоптоза // Бюллетень сибирской медицины. — 2004. — Т. 3, № 1. — С. 63- 70.
- Манских В. Н. Пути гибели клетки и их биологическое значение // Цитология. — 2007. — Т. 49, № 11. — С. 909-915.
- Колюбаева С.Н. К механизму образования микроядер в ФГА- стимулированных лимфоцитах периферической крови человека / С.Н. Колюбаева, Л.В. Щедрина, Р.П. Степанов [и др.] // Цитология. - 1986. - Т. 28, № 26. - С. 227-231.
- Афанасьев Ю.И., Юрина Н.А. Гистология. M.: Медицина. - 2001 - 672 с.
- Abend M, Frombeck S, Van Beuningen D. Indications for an active process underlying spontaneous and radiation-induced micronucleation in L929 cells// International Journal OfRadiation Biology. - 1999. - Vol.75, No.12. - P. 1567-1578. doi: 10.1080/095530099139179
- Decordier, E. Cundari, L. and M. Kirsch-Volders, Influence of caspase activity on micronuclei detection: A possible role for caspase-3 in micronucleation// Mutagenesis. -2005. - Vol.20, No.3. -P.173-179.