Abstract
An ion chromatographic method for determination of anion composition of natural waters was described. An automated 850 IC Professional, Metrohm system equipped with conductivity detector after chemical and carbon dioxide suppression was used. The main analytical characteristics were estimated for each of studied analytes. The tmeness of the method was estimated by analysis of certified reference material for soft drinking water. Recovery test was performed on spiked drinking water samples. The method was applied for analysis of drinking water before and after chlorination, as well as high mountain lakes.
The selected methods were successfully applied for the analysis of water samples from Iiigh mountain lakes, as well as drinking water before and after chlorination. The results obtained in this study show that there is no indication of contamination with elements or anions in the lake water. There were no deviations found from the legally acceptable values for priority substances, which could present a risk to the aquatic environment
Introduction
The environmental monitoring of pollutants in waste, surface, drinking or natural water is a requirement for achieving a good ecological and chemical status of the water, for protection of human health, the water supply system and natural ecosystems [1, 2]. Particular attention is paid to natural water as a medium for the migration of chemical elements and compounds with natural and anthropogenic origin. Many factors can have an influence - climate, relief, geological structure and rock formation, soil and vegetation cover and anthropogenic factors.
Up to now chemical monitoring of the high mountain lakes in Bulgaria has been carried out in the period 1993 - 1996 in the Rila Mountains. There was no indication for contamination with heavy metals in the considered mountain lakes, the results are below the limit of quantification of the used methods for analysis [3, 4].
Nowadays due to intense development of chemical and biochemical technologies new substances appeared in the environment, which imposes determination of new analytes in common matrices or standard anions in new matrices. The environmental and health authorities, as well as modern industrial technologies impose stringer demands to the water quality and methods for determination at trace levels are highly demanded [5-9]. Ion chromatography appears to be widely studied method for anion determination in different matrices in very precise and accurate manner offering low detection limits, low sample and reagent consumption, as well as short analysis time [7 - 14]. A line of studies on optimization and validation of ion chromatographic methods for anions determination aimed at ensuring quality of the results and their fit for purpose according to the demands of customers have been recently published [7-15].
This paper presents the results from determination of anion composition of treated and untreated drinking water, as well as high mountains lakes by ion chromatographic method with conductivity detection after chemical and carbon dioxide suppression.
Experimental
Instruments and operating conditions
An ion chromatographic, metal - free system (IC - 850 Professional model, Metrohm AG, Switzerland) controlled by Metrodata MagIC Net™ software and equipped with 858 Professional Sample Processor, sample filtration system with a 0.2 pm regenerated cellulose membrane, six channel injection valve, low pulsation high - pressure pump, chemical suppression and CO2 suppression, eluent degasser and conductivity detector was used. The separation was performed on a Metrosep A Supp 7 - 250 column (250 x 4 mm, polyvinyl alcohol with quaternary ammonium groups, 5 pm particale size), Metrosep RP 2 Guard/3.5 (polymer with pore size 0.2 pm). In order to keep the baseline signal as low as 0.99 pS/cm, 0.1 M sulphuric acid solution was used for regenerated chemical suppression. The optimized chromatographic parameters are presented in Table 1. Eluent solution was prepared by sodium carbonate Suprapure ШзСОз (CertiPUR®, Merck, Darmstadt, Germany). Multi ion standard solutions of fluoride, chloride, nitrate and sulphate were prepared by appropriate dilution of their anion standard stock solution (Fluka 1000 mg/L for IC, TraceCERT®, Sigma - Aldrich, Buchs, Switzerland). All solutions were prepared gravimetrically in Milli - Q water, (Mill - Q Direct 8, Merck Millipore; resistivity > 18.2 MQcm, equipped with a Millipack 0.22pm filter). Certified Reference Material for soft drinking water UK - Anions № ERM® - CAO 16a, LGC, Teddington, Middlesex was used to confirm the trueness of the method. Sulphuric acid (96% Suprapur®, Merck KGaA, Darmstadt, Germany) was used in conductivity suppression regeneration system after appropriate dilution. Before measurements the solutions were filtered by MF - Millipore™ membrane filter with pore size of 0.45 pm (Merck KGaA, Darmstadt, Germany).
Table 1 Ion chromatographoperating conditions
|
IC—850 Professional, Metrohm AG |
|
|
Column |
Metrosep A Supp 7- 250/4.0 |
|
Precolumn |
Metrosep RP 2 Guard/3.5 |
|
Eluent |
КагСОз (3.6 mmol/L) |
|
Eluent flow rate |
0.7 mL/min |
|
Elution mode |
Isocratic |
|
Sample volume |
20 pL |
|
Column temperature |
45 0C |
|
Detection |
Suppressed conductivity |
|
Determination mode |
Peak area |
Reagents and Certified Reference Materials
All solutions were prepared gravimetrically with deionized water Milli-Q(18.2MQcm, Millipore, Merck). Multiion standard solutions were prepared by appropriate dilution of their anion standard stock solution 1000 mg/L, TraceCERT®", (Sigma-Aldrich, Buchs, Switzerland). Before measurements, the samples were filtered through MF-Millipore™ membrane filter with pore size of 0.45 pm (Merck KGaA, Darmstadt, Germany).
Results and discussion
A previously developed and validated ion chromatographic method was used [16]. The studied anions are separated by 3.6 mmol/L МагСОз eluent under the optimized parameters presented in the Table 1. All anions were completely separated in a run with total analysis time of 30 min. The developed and validated methods allowed the determination of low concentrations of anions which werebelow the established limits for natural water [17].
Analysis of water from high mountain lakes
The anionic composition is an important indicator of the water quality and ecological lakes status. Different types of minerals, rock mass, acid rain, human and animal activity are major sources of increased concentrations of anions in the water [8]. The results are presented in Table 2 and 3. The measurement uncertainty varies between 7-10% for different anions. Samples were also tested for bromide (Br ), bromate (BrO3 ), nitrite (NO3 ) and phosphate (PO43 ).The results are below the detection limits and no contamination of the water was found.
|
lakes |
F' mg/L |
BrO3 mg/L |
CT mg/L |
NO2 mg/L |
Br mg/L |
NO3 mg/L |
PO43 mg/L |
SO42 mg/L |
|
Frog lake |
0.017 |
<DL |
0.20 |
<DL |
<DL |
0.064 |
<DL |
1.5 |
|
Eye lake |
0.026 |
<DL |
0.31 |
<DL |
<DL |
0.054 |
<DL |
1.3 |
|
Muratovo lake |
0.057 |
<DL |
0.28 |
<DL |
<DL |
0.16 |
<DL |
2.0 |
|
Fish lake |
0.027 |
<DL |
0.26 |
<DL |
<DL |
0.26 |
<DL |
1.7 |
|
Long lake |
0.020 |
<DL |
0.25 |
<DL |
<DL |
0.081 |
<DL |
1.8 |
|
Lake under the Long |
0.043 |
<DL |
0.23 |
<DL |
<DL |
0.047 |
<DL |
2.8 |
Table 2 Anion composition of waters of high mountain lakes from Pirin mountain, Bulgaria
Analysis of drinking water
The validated method was applied for analysis of treated and untreated drinking water [17]. The recovery for standard anions obtained in spiked certified reference material for soft drinking water (UK - Anions № ERM® - CAO 16a) were between 98 and 106%. The results are presented in Figure 1. The obtained concentrations are far below the recommended chemical and indicator values in drinking water [5].
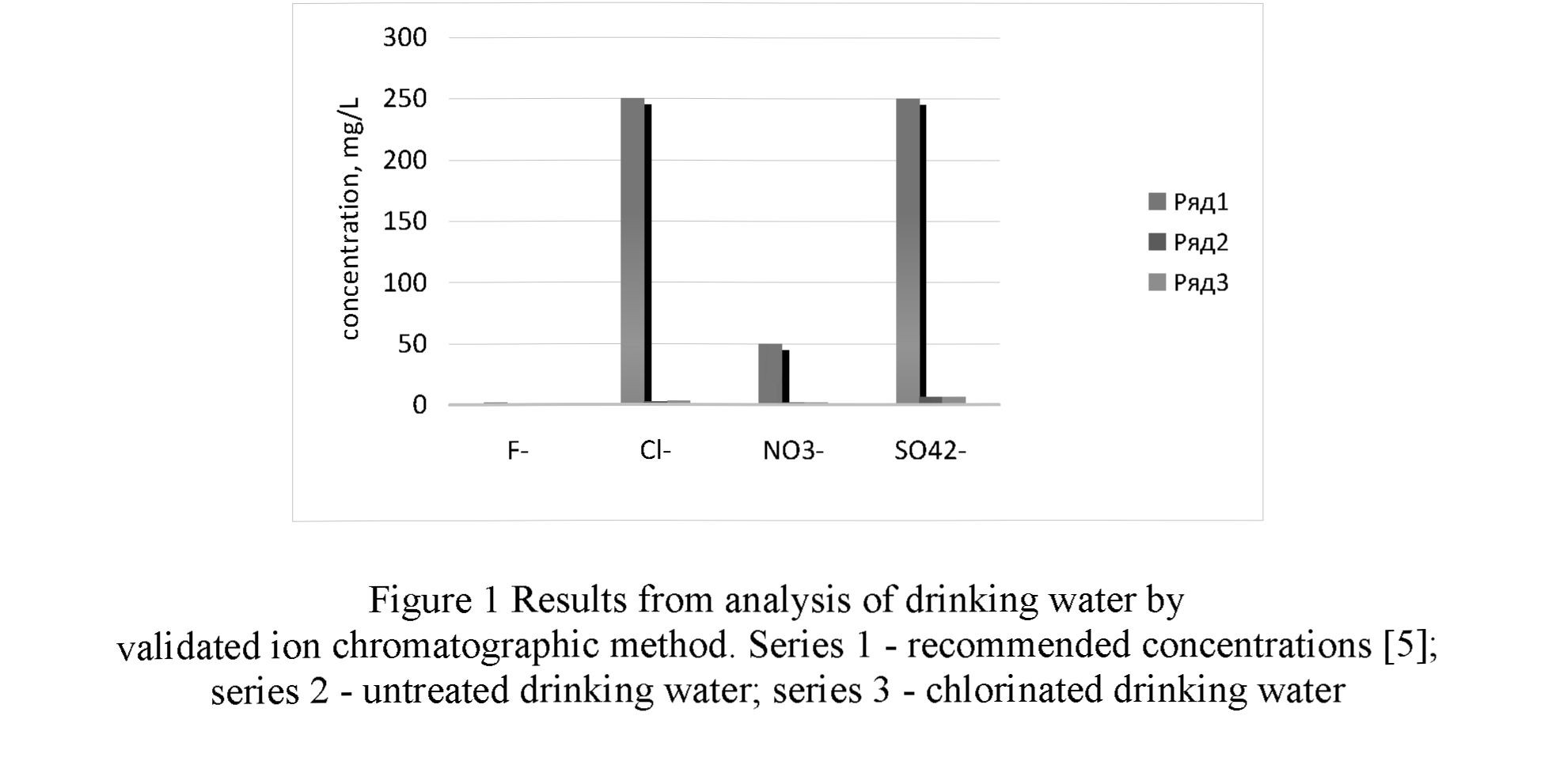
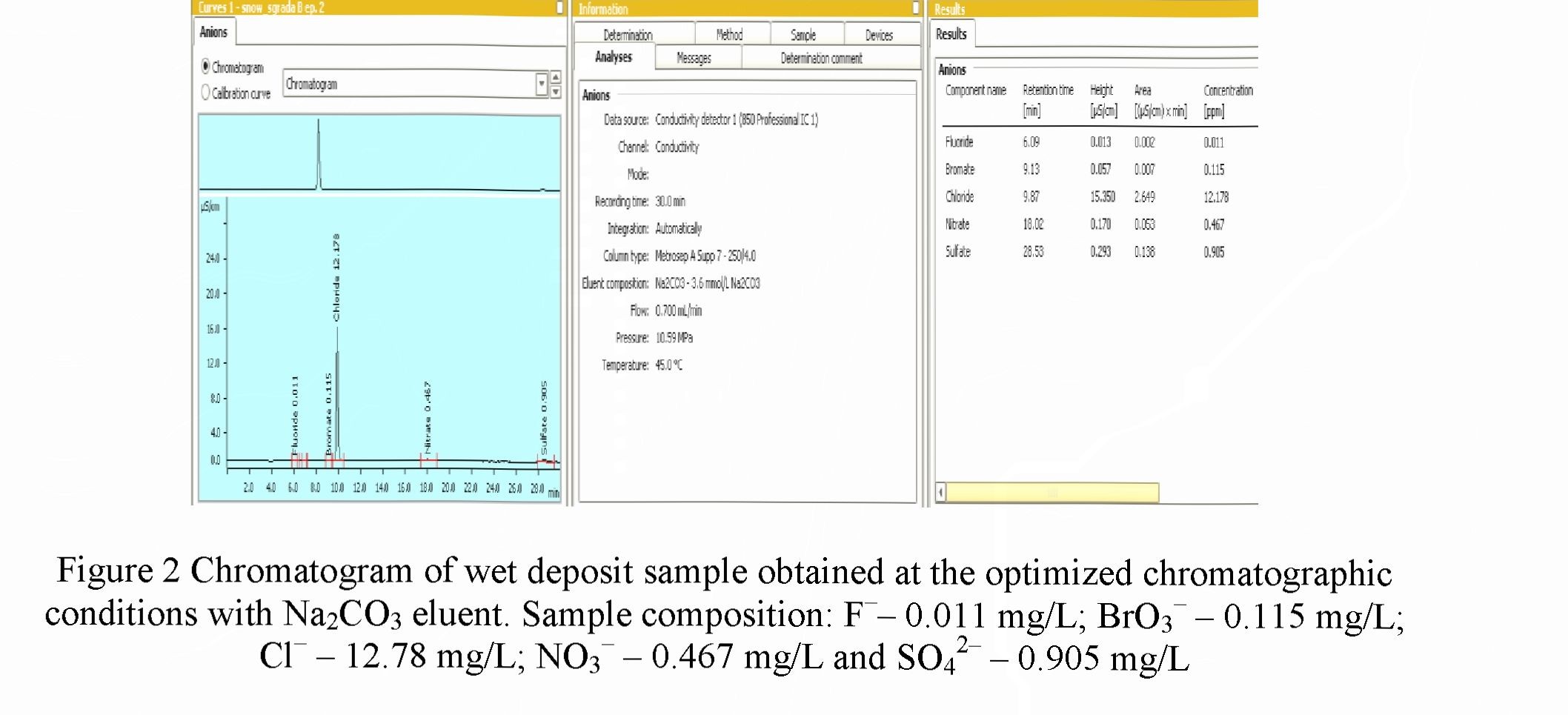
Analysis of wet deposits
Wet deposit from region of Sofia, Bulgaria were analyzed by the validated method. The chromatogram is presented on Figure 2. No matrix interference was observed. The retention times of studied anionscoincided with the standards within 2%. The studied wet deposits contained fluoride, chloride, nitrate and sulfate ions at different concentrations depending of the sampling site. Bromate, nitrite and phosphate anions were found occasionally.
The selected methods were successfully applied for the analysis of water samples from high mountain lakes, as well as drinking water before and after chlorination. The results obtained in this study show that there is no indication of contamination with elements or anions in the lake water. There were no deviations found from the legally acceptable values for priority substances, which could present a risk to the aquatic environment.
Acknowledgement The financial support of the Science and Research program of the University of Chemical Technology and Metallurgy, Bulgaria through the project 11802/2018 is gratefully acknowledged.
References:
- B. Lopez-Ruiz. Advances in the determination of inorganic anions by ion chromatography; J. Cliromatography A. 881 (2000) 607 -627
- E. Rodriguez, S. Poynter, M. Curran. P. Haddad, R. Shellie, N. Nesterenko, B. Pauli. Capillary ion Cliromatography with on-column focusing for ultra-trace analysis of methanesulfonate and inorganic anions in limited volume Antarctic ice core samples; Journal of Chromatography A. 1409 (2015) 182— 188
- J.N. Stamenov. B.I. Vachev. Monitoring and management of mountain environment. Journal of Enviromnental Radioactivity 72 (2004) 121-128
- G. Pekov. P. Mandjukov. S. Tsakovski. V. Simeonov. Chemical and multivariate statistical analysis of waters from the Rila mountain lake. Observatoire de mounagne de Moussala. OM2. 1998 Sofia 253- 258 p.
- Emopean Coimnision 2014 Cormcil Directive 98/83/EC of 3 November 1998 on the quality of water intended for human consumption. Off. J. Em. Coimnun. 2014 L221, 1-101.
- Emopean Coimnission 2002 Off. J. Em. Conunun. L221 8-36
- D. Zivojinovic, L. Rajakovic, Application and validation of ion chromatography for the analysis of power plants water: Analysis of corrosive anions in conditioned water-steam cycles, 2011 Desalination 275 17-25
- M. Cengiz, A. Bilgin, Determination of major sodium iodide Symporter (NIS) inhibitors in drinking waters using ion chromatography with conductivity detector,2016 J. Pharm. Biomed. Analysis 120 190-197
- D. Verrey, M. Louyer, O. Thomas, E. Baures, Direct determination of trace-level haloacetic acids in drinking water by two-dimensional ion chromatography with suppressed conductivity, 2013 Microchemical J. 110 608-613.
- E. Gilchrist. D. Healy, V. Morris, J. Glennon, A review of oxyhalide disinfection by-products determination inwaterby ion chromatography and ion chromatography-mass spectrometiy,2016 Anal. Chim. Acta 942 12-22
- H. Abudiak, C. Robinson. M. Duggal, S. Strafford. K. Toumba, Effect of fluoride sustained slow- releasing device on fluoride, phosphate and calcium levels in plaque biofilms over time measured using ion chromatography. 2012 J. Dentistry 40 632-638
- . Bajenaru, I. Minca, C. Guran, A. Josceanu, F. Bacalum, Validation of an Ion Chromatographic Method for Determinationof Anions in Wet Depositions, 2013 Rev. Cliim.(Bucharest) 64 122913
- C. Lopez-Moreno, I. Viera, A. Urbano, Validation of an ion chromatographic method for the quantification ofanions in water, 2010 Desalination 26 111-116
- A. Krata, V. Kontozova-Deutsch, L. Benes, F. Deutsch, R. Van Grieken, Single-run ion chromatographic separation of inorganic and low-molecular-massorganic anions under isocratic elution: Applicationto environmental samples, 2009 Talanta 79 16- 21.
- Eurachem Guide: The Fitness for Purpose of Analytical Methods - A Laboratory’ Guide to Method.
- V. Ivanova. A. Surleva, B. Koleva, Validation of Ion Chromatographic Method for Determination of Standard Inorganic Anions in Treated and Untreated Drinking Water, IOP Conf. Ser.: Mater. Sci. Eng. 374 (1). 2018.012053.
- V. Ivanova. B. Koleva, A. Surleva, Determination of pollutants in high mountain lakes by ion Cliromatography and inductively coupled plasma mass spectrometry, Metrology’ and metrological assurance 2018, 10-14 September 2018, Sozopol, Bulgaria, pp 264-267.