This paper summarizes results of clinical studies in the period of years 1971–2014 as follows: (1) In persons with type 1 diabetes, effectiveness of conventional therapy with multiple doses of shortacting insulin (MDI) or continuous sucutaneous insulin infusion (CSII) was demonstrated. Dynamic training was shown to enhance insulin sensitivity and plasma HDL cholesterol, and to improve neuropathy, memory and attention. Influence of alcohol and effects of insulin on postprandial alcohol concentrations revealed similar consequences; model experiments were suggested when answering forensic questions. (2) In persons with type 2 diabetes, advantage of complementary therapy with prandial rapid acting insulin was demonstrated. In MDIpersistent hyperglycaemia, insulin pump was shown to reduce HbA1c, to diminish daily insulin dose and to improve quality of life. Benefits of incretin preparations were described. Effects of losartan on renal and cardiovascular outcomes in diabetic nephropathy were documented. (3) Perinatal mortality of newborns of mothers with diabetes decreased due to thorough diabetes and obstetric care. (4) Manual Device for Insulin injections (MADI-pen) was developed and introduced into daily routine. Its accuracy, pharmacological and microbial safety were proved. (5) «Programmed Treatment of persons with diabetes» comprising the principels of Therapeutic Patients Education of the WHO was introduced. (6) Glycaemic Index of foods was calculated using a new method with continuous glucose monitoring. (7) Intensive selfmonitoring. The accuracy and precision of various glucometers were explored. Tenpoint ambulatory glycaemic profile and continuous glucose monitoring system (CGMS) was introduced to practice. In persons on insulin pumps CGMS resulted in reduction of HBA1c.
Introduction
This review comprises practice-related outcomes of clinical studies carried out in the period of years 1971–2014 at the University Diabetes Centre in Olomouc, Czech Republic [1]. The problems were solved in cooperation with other centres in Czech and Slovak Republic and abroad, particulatly with the Institute of Diabetes «G.Katsch», Karlsburg, Diabetiker Sanatorium Bergfried, Saalfeld, Dept. of Endocrinology, University Heinrich Heine, Düsseldorf, Germany; Royal Infirmary Edinburgh, St. Thomas Hospital, London, UK; Institute of Diabetes «N.C.Paulesco», Bucharest, Romania; etc. Over 100 professionals, medical students and nurses were step by step involved into the presented clinical research. Thousands of various biochemical and radioimunological analyses were performed in highly specialized laboratories in Olomouc, Karlsburg and Moravský Beroun.
All studies paid attention to the optimum diagnostic and therapeutic procedures in type 1 and type 2 diabetes mellitus including the development of aids for insulin administration and educational programmes.
The most important achievements were presented at national and/or international conferences (ADA, EASD etc.) and published. In addition, their implementation into daily routine has always been considered.
In this paper the achievments are summarized in seven chapters:
- Treatment of people with type 1 diabetes (PWD1).
- Treatment of people with type 2 diabetes (PWD2).
- Diabetes and
- Technical prerequisites for the intensive conventional insulin
- Education — Programmed treatment of people with
- Glycaemic Index of
- Intensive
- Treatment of people with type 1 diabetes mellitus
Conventional insulin substitution. A randomized cross-over study (1987–1990) was performed in Institute of Diabetes Gerhardt Katsch, Karlsburg, with 36 PWD1 (men) admitted to hospital for a period of 6 weeks. Each tested person was assigned to one of six arms differing only by the sequence of three defined insulin Regimens A, B, C. In every men all three Regimens were tested (two weeks each). At the end of each 14-day test period a 16-point B-glucose (BG) and P-free insulin (FIRI) profile was performed (Fig. 1).
This study revealed that the basal and prandial insulin substitution with only shortacting porcine insulin given 7 times a day (Regimen A) is the most effective kind of conventional insulin therapy [2]. This approach led to the best metabolic control in 21/36 (58 %) tested PWD1. An insulin regimen with similar insulin doses over morning and afternoon hours but different regimens over night — either intermediate insulin preparation BS at 10 p.m. (Regimen B) or longacting insulin Ultratard HM at 5.30 p.m.(Regimen C) led to the best control only in 6/36 (17 %) or in 9/36 (25 %) PWD1, respectively.
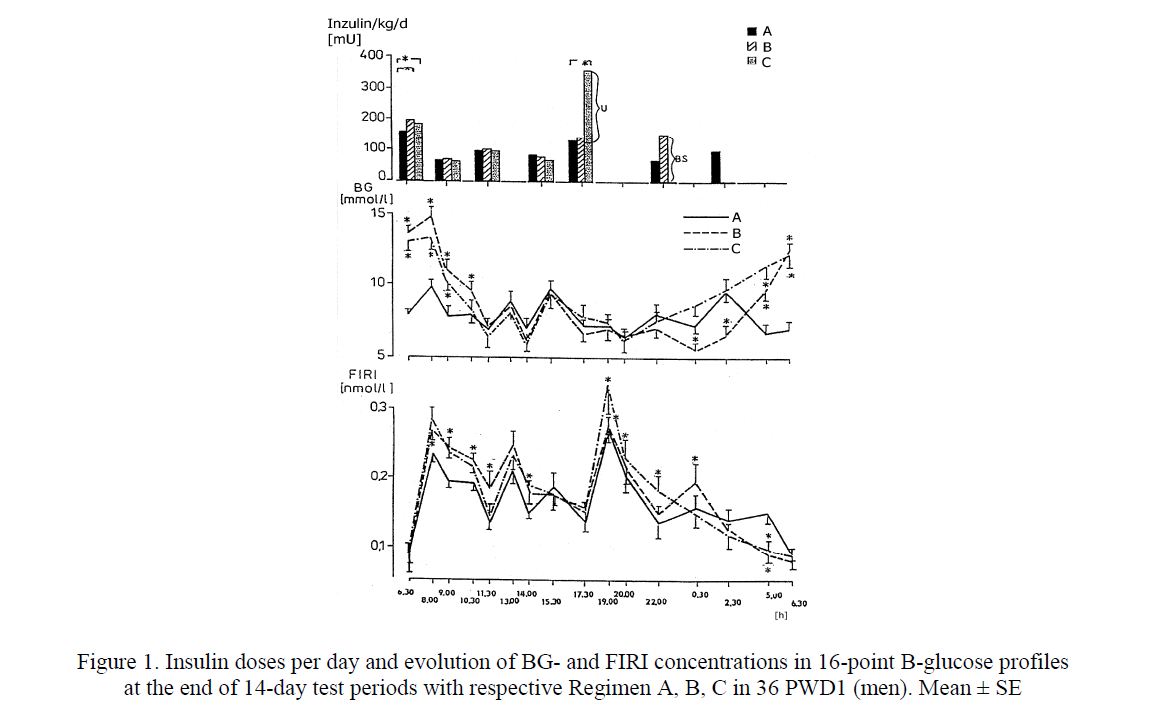
Figure 1. Insulin doses per day and evolution of BGand FIRI concentrations in 16-point B-glucose profiles at the end of 14-day test periods with respective Regimen A, B, C in 36 PWD1 (men). Mean ± SE
In the last two regimens (B or C), the total daily insulin dose was higher than in Regimen A. The group education and a new insulin pen (MADI) proved to motivate the PWD1 to an intensification of insulin therapy including injections of shortacting insulin at 2.30 a.m. in Regimen A. The acceptance of night injections increased from 2 % at the baseline to 42 % at the end of the study. However, despite of such an intensive treatment, a longlasting euglycaemia could not be reached, even though the plasma insulin concentrations were higher than in persons without diabetes (Fig. 2, Fig. 3). There were no differences in metabolic control when using the needle-pen or catheter-pen [3, 4]. The needle pen was preferred in 54 % of all patientdays [5].
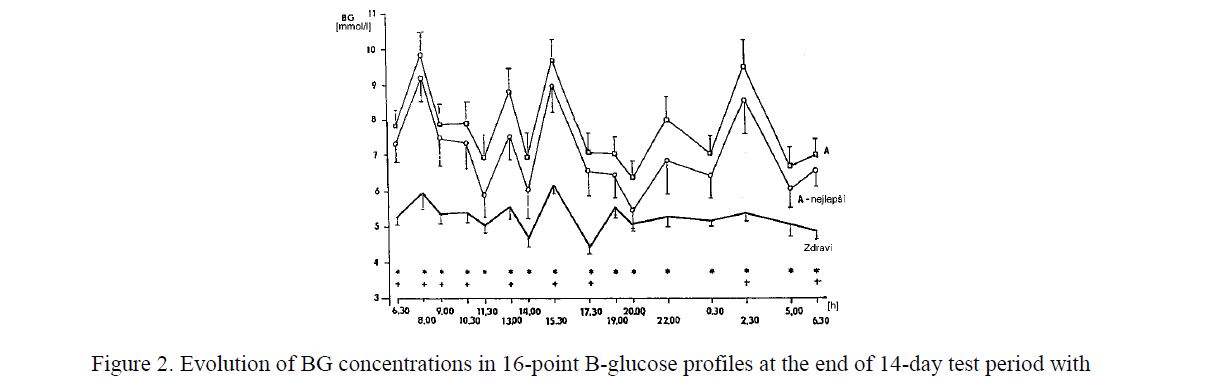
Figure 2. Evolution of BG concentrations in 16-point B-glucose profiles at the end of 14-day test period with Regimen A in all PWD1 (n=36), in the best PWD1 (A — nejlepší, n=21) and in healthy men (Zdraví, n=9). Mean ± SE
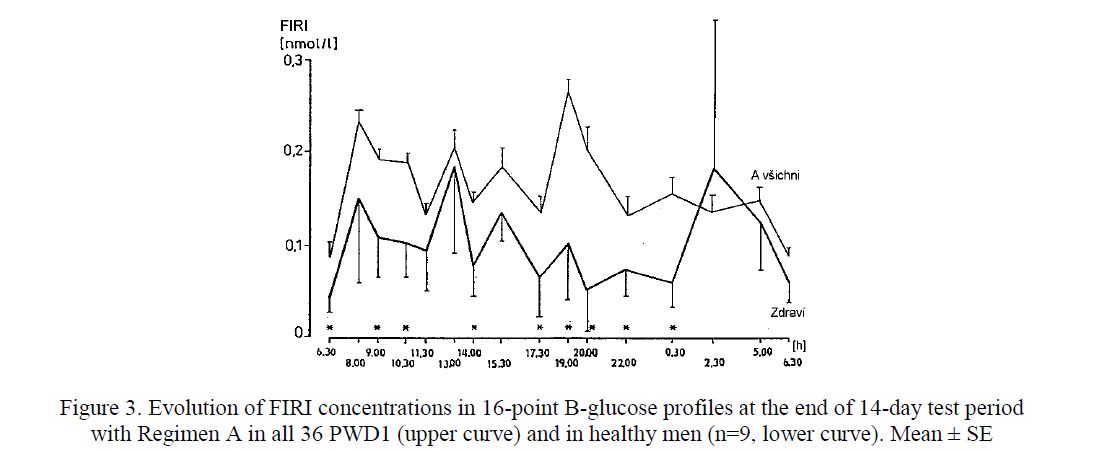
Figure 3. Evolution of FIRI concentrations in 16-point B-glucose profiles at the end of 14-day test period with Regimen A in all 36 PWD1 (upper curve) and in healthy men (n=9, lower curve). Mean ± SE
Continuous subcutaneous insulin infusion (CSII) in PWD1. A retrospective study (1993–1998) with 13 men and women on an insulin pump (Dahedi, H-Tron, Minimed) demonstrated that CSII resulted within 72 days in a decrease of concentrations of HbA1c in blood (9.3±0.46 vs 6±0.28 %, p<0.05, DCCT units), reduction of total serum cholesterol (5.47±0.29 vs 4.85±0.19 mmol/l, p<0.05) and triacylglycerols TG (1.58±0.24 vs 1.13±0.15 mmol/l, p<0.05). The total daily dose of insulin was reduced (47.8±2.75 vs 41.3±2.3 IU/d, p<0.05) and the body mass did not change. An improved metabolic control was also found in a check-up 554 days later. There were no serious complications resulting from handling insulin pump [6]. In the year 2004 we have replaced phosphate buffered insulin (Velosulin) with insulin aspart (Novorapid) for all PWD1 on insulin pumps. Reduction of HBA1c without increased frequency of hypoglycaemias appeared within one year (Fig. 4). The daily insulin dose and body mass did not change [7].
![HbA1c concentration (DCCT) in PWD1 on CSII in the course of treatment with phosphate buffered human insulin (VELOSULIN) and after switching to insulin aspart (NOVORAPID). Mean ± SE [7]](/uploads/data/files/pics8/4_145.jpg)
Figure 4. HbA1c concentration (DCCT) in PWD1 on CSII in the course of treatment with phosphate buffered human insulin (VELOSULIN) and after switching to insulin aspart (NOVORAPID). Mean ± SE [7]
Dynamic physical A prospective study (1978–1982) with 19 men improved insights in submaximum physical training (duration 157±43 days). Here are the effects of training in men with type 1 diabetes:
An improvement of carbohydrate metabolism demonstrated by the increased insulin effectiveness without any change in blood glucose control. An approximate relation between insulin effectiveness Q and physical working capacity (PWC 170) was calculated using formula Q [g /IU = 0,03 PWC 170 [W] – 0,5, (where 90 W < PWC 170 < 295 W). This formula was derived from our observations and needs to be confirmed. The insulin effectiveness Q reached values ranging from 2.5 to 22.4 g Carb./IU. The increase of the PWC depends on the amount of energy expended for the submaximal training. Following the training interruption, the insulin effectiveness drops depending on the decrease of PWC 170 (Fig. 5).
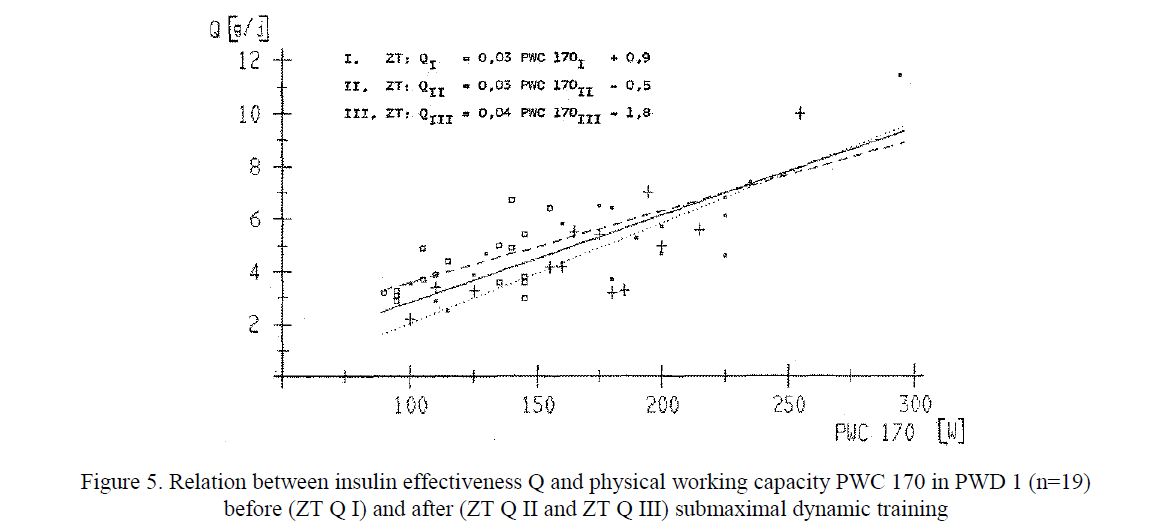
Figure 5. Relation between insulin effectiveness Q and physical working capacity PWC 170 in PWD 1 (n=19) before (ZT Q I) and after (ZT Q II and ZT Q III) submaximal dynamic training
An improvement of lipoprotein metabolism demonstrated by an increase in HDL cholesterol concentration (1.19±0.08 vs 1.86±0.22 mmol/l, p<0.05, Fig. 6) and by a decrease in the index of total cholesterol/HDL cholesterol. These significant changes were also found 7 days after the end of the
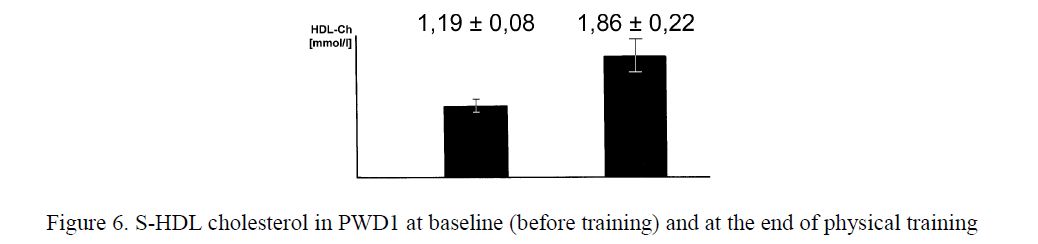
Figure 6. S-HDL cholesterol in PWD1 at baseline (before training) and at the end of physical training
A beneficial influence on some signs of neuropathy, on memory, attention and on the general condition of men with type 1 diabetes was demonstrated as
Based on this study, a submaximal dynamic physical training may be recommended as an additive treatment of persons with type 1 diabetes with no signs of catabolism. At the beginning, the insulin should be reduced or the amount of carbohydrates in food increased, according to the change of insulin effectiveness. Even in patients with high physical working capacity it is not possible to replace insulin by physical exercise. Following the training cessation, the insulin should be increased or the amount of carbohydrates in food decreased according to the decrease of insulin effectiveness [8, 9].
- Ethyl alcohol consumption. A controlled prospective study (1986–1988) on influence of ethyl alcohol consumption on B-glucose concentrations and on the influence of B-glucose on ethyl alcohol concentrations was A group of 15 men with DM1 and a group of 15 healthy men consumed alcohol in order to reach the alcohol concentration in blood of 1 g/kg. Following formula was used to calculate the amount of consumed alcohol:
- Amount of consumed alcohol [g] = 0.7 × body mass [kg] × alcohol concentration in blood [g/kg]. This trial brought the following conclusions [10]:
- Development of alcoholaemia after application of a usual insulin dose and after a fixed breakfast in men with type 1 diabetes is similar to that in healthy males. A reduction of the insulin dose (and omitting of the breakfast) results in a quicker increase of alcoholaemia with a higher maximum
- The maximum alcoholaemia up to 1 g/kg does not influence neither the B-glucose nor the B-acetone within 240 min after drinking the The serum concentration of non-esterified fatty acids (NEFA) in men with diabetes (but not in healthy persons) decreases, the concentrations of TG and lactate does not change. Alcohol had no influence on the concentrations of some amino acids (threonin, glutamic acid, prolin, ornithin, arginin) and ambivalent influence on other amino acids.
- Due to large inter-individual differences in the development of calculated alcoholaemia (Fig. 7) a model experiment appears to be necessary in order to answer some important forensic questions [11].
![Individual alcohol concentrations in blood reached in PWD 1 after consumption of the calculated amount of alcohol [g] with full dose (100 %) of insulin + full meal, and with half dose of insulin without meal](/uploads/data/files/pics8/7_43.jpg)
Figure 7. Individual alcohol concentrations in blood reached in PWD 1 after consumption of the calculated amount of alcohol [g] with full dose (100 %) of insulin + full meal, and with half dose of insulin without meal
- Treatment of persons with type 2 diabetes mellitus
- Start of Complementary insulin therapy [12]. A prospective study (1991–1994) with 251 men and women with type 2 diabetes (PWD2) aged between 20 and 82 years demonstrated benefits of small supplementary insulin doses (1 to 10 IU) before each meal both in a group of 108 PWD2 previously on diet/oral antidiabetic drugs which started a complementary treatment with total insulin 26 IU/d (Fig. 8), and in a group of 143 PWD2 previously treated with longacting insulin in which on complementary treatment the daily insulin dose was reduced from 47 IU/d to 32 IU/d (Fig. 9) [13].
![Evolution of BG concentrations in 10-point B-glucose profiles without insulin (upper curve) and with complementary prandial doses of 1 to 10 IU of shortacting insulin (lower curve), the mean total of 26 IU/d. n = 108, mean±SE [13]](/uploads/data/files/pics8/8_24.jpg)
Figure 8. Evolution of BG concentrations in 10-point B-glucose profiles without insulin (upper curve) and with complementary prandial doses of 1 to 10 IU of shortacting insulin (lower curve), the mean total of 26 IU/d. n = 108, mean±SE [13]
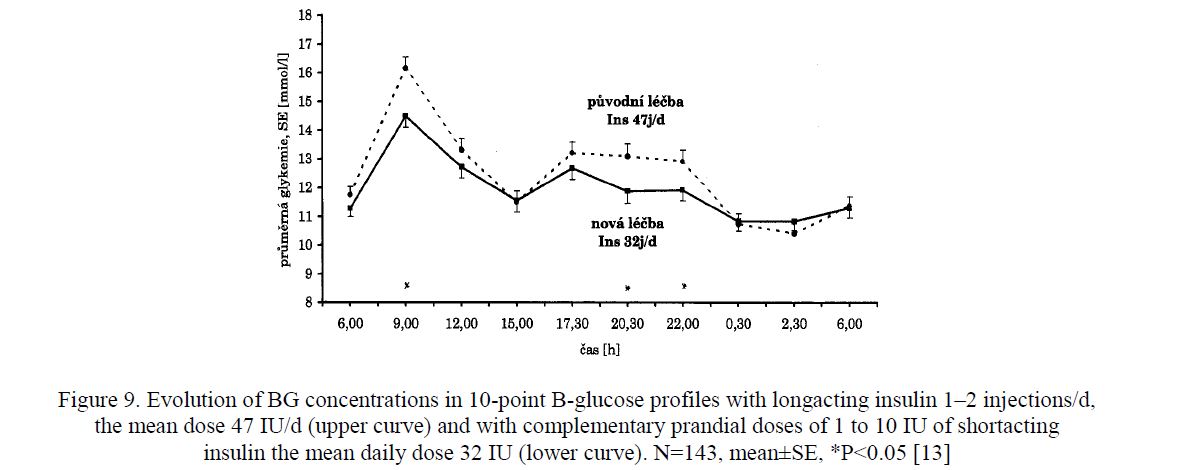
Figure 9. Evolution of BG concentrations in 10-point B-glucose profiles with longacting insulin 1–2 injections/d, the mean dose 47 IU/d (upper curve) and with complementary prandial doses of 1 to 10 IU of shortacting insulin the mean daily dose 32 IU (lower curve). N=143, mean±SE, *P<0.05 [13]
In the course of 10 weeks the BMI decreased from 29.4±0.73 to 28.9±0.73 kg/m2 (p<0.05). In addition, this treatment resulted not only in an improvement of B-glucose profile, mean B-glucose, HbA1c but also in a better spectrum of lipoproteins (increase of LpA, decrease of apo B and Lp(a) — Fig. 10). The patients´ satisfaction with the new treatment was good. The increased number of injections has not been an obstacle: 91 % of PWD2 (when using MADI pen) decided to go on the intensive complementary treatment [14].
![Changes in serum concentrations of Lipoprotein A (upper diagram) and lipoprotein B (lower diagram) in PWD2 between baseline and 10 weeks with complementary treatment [14]](/uploads/data/files/pics8/10_12.jpg)
Figure 10. Changes in serum concentrations of Lipoprotein A (upper diagram) and lipoprotein B (lower diagram) in PWD2 between baseline and 10 weeks with complementary treatment [14]
- Continuation of Complementary (supplementary) insulin therapy over 5 A retrospective analysis of a group of 70 PWD2 in which a complementary insulin treatment was introduced in our centre in the year 1991 or later on (so that this treatment lasted 5.0±0.12 years), and which were consequently treated in three different diabetes centres, brought the following results: the body mass decreased in 56 %, the concentration of HbA1c decreased in 56 % and the daily insulin dose decreased in 57 % of all 70 PWD2 [15]. Based on these observations the complementary insulin treatment may be recommended for a longlasting treatment of both non-obese and obese PWD2.
- Rapid acting insulin analogs in complementary therapy. In our trial (2002–2007) 57 PWD2 treated with human regular insulin for 5.2 ± 0.44 years were investigated [16]. Following two checkups performed in the course of the 364 ± 9-day baseline period, human regular insulin was replaced with aspart in equivalent boluses, and two checkups in the course of 330 ± 11.1-day sequential period were performed (Fig. 11). The control group consisted of 17 PWD2 treated with insulin for 4.2 ± 0.57 years. In the intervention group, following the switch from human regular insulin to aspart, hemoglobin A1c (HbA1c) decreased from 8.4 ± 0.23 % at baseline to 7.9 ± 0.17 % (P = 0.031), and thereafter to 7.5 ± 0.20 % (P < 0.001) daily insulin dose (37.1 ± 1.39 IU/d), BMI (30.5 ± 0.82 kg/m2), and frequency of hypoand hyperglycaemic episodes did not change (P > 0.05).
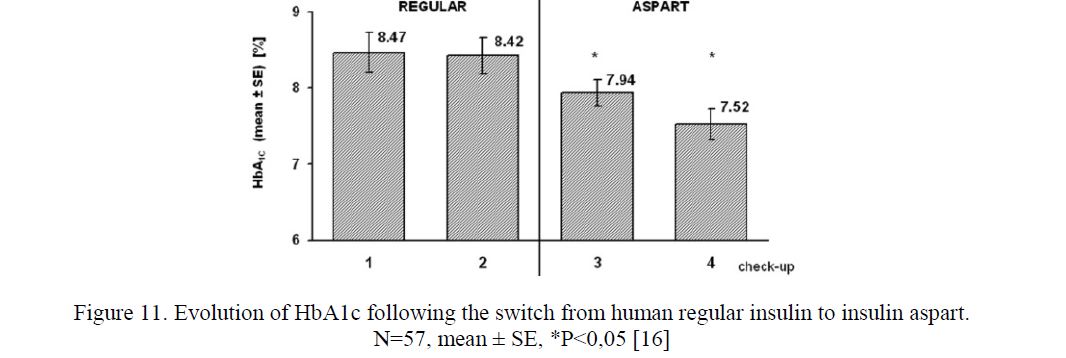
Figure 11. Evolution of HbA1c following the switch from human regular insulin to insulin aspart.
In the control group, no significant change of baseline HbA1c (8.4±0.54 %), insulin dose (33.1±3.17 IU/d), and BMI (32.1±1.12 kg/m2) was found. Aspart appears to be more effective than human regular insulin for supplementary (complementary) insulin treatment in individuals with type 2 diabetes. Our conclusions correspond to other studies [17–19]
- Continuous subcutaneous insulin infusion in Our pilot monocentric uncontrolled trial PARASEN (2006–2010) demonstrated that CSII in trained PWD2 results in reduction of daily insulin dose without change in HbA1c concentration and body mass [20, 21]. This is in accordance to other studies, in particular to the recent prospective randomized multicentre controlled study OpT2mise (2011–2014) [22, 23].
- Inkretin preparations. In 35 obese metformin-treated PWD2 with persistent hyperglycaemia exenatide twice daily or liraglutide once daily was injected. In the course of 3 to 6 months reduction of body mass up to 1 kg/week (Fig. 12) and HbA1c concentrations about 10 % of baseline (Fig. 13) was registered in most of them. However, following the 6-month successful period, these effects were mostly less pronounced or none. Exenatide QW (once per week) appears to be
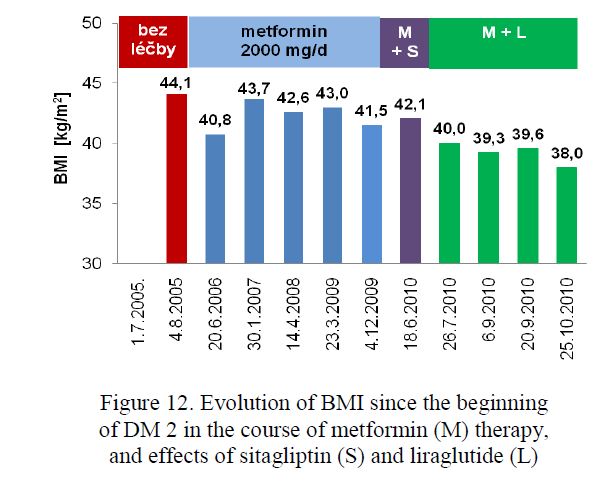
Figure 12. Evolution of BMI since the beginning of DM 2 in the course of metformin (M) therapy, and effects of sitagliptin (S) and liraglutide (L)
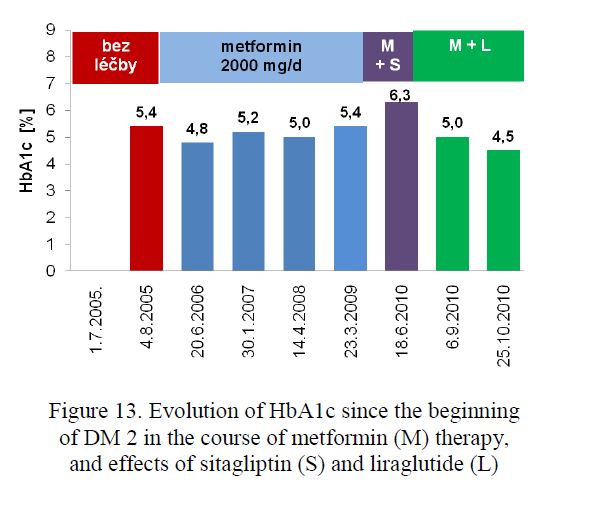
Figure 13. Evolution of HbA1c since the beginning of DM 2 in the course of metformin (M) therapy, and effects of sitagliptin (S) and liraglutide (L)
Study RENAAL (Double-Blind, Randomized, Placebo-Controlled Study to Evaluate the Renal Protective Effects of Losartan in Patients With Noninsulin Dependent Diabetes Mellitus and Nephropathy; 1997–2001, principal coordinator B.M.Brenner, California) [24].
A total of 1513 patients were enroled in this study at 250 specialized centres in 28 countries, 25 of them in Olomouc. There were a total of 327 patients in losartan group and 359 in placebo group. In conclusion, losartan conferred significant renal benefits in PWD2 and nephropathy and it was generally well tolerated.
- Diabetes mellitus and pregnancy
An open epidemiologic study aiming to the diabetes treatment in pregnancy demonstrated that the intensive management of diabetes carried out at the University Hospital in Olomouc resulted in a marked decrease of the perinatal mortality of newborns of diabetic mothers. This mortality dropped from 66 % in the year 1965 to 6.5 % in the year 1980 and finally reached the same percentage as in non-diabetic mothers (i.e. less than 1 %). To date, the management of a pregnant diabetic woman is based on the intensive treatment with shortacting insulin or insulin aspart (either conventional by means of a pen or as continuous subcutaneous insulin infusion by means of an insulin pump) aiming to the euglycaemia level (P-glucose between 4.0 and 7,0 mmol/l) and on intensive obstetrical care in a specialized centre. Insulin pump and intensive selfmontoring using CGMS is recommended (Fig. 14). Delivery in 38–40 week of pregnancy; in threatening complications any delay of a caesarean section should be avoided [25, 26].
![Evolution of P-glucose concentration at the day of delivery. PWD1, Primipara [26]](/uploads/data/files/pics8/14_2.jpg)
Figure 14. Evolution of P-glucose concentration at the day of delivery. PWD1, Primipara [26]
- Technical prerequisites for the intensive conventional insulin treatment
- The Pen-like case [5]. In a close cooperation with other institutions a proposal of a simple pen-like case for an insulin syringe filled with insulin (Fig. 15) was submitted and the safety of this case was investigated. The pen case was produced from light metals or plastic materials and used several years before the era of insulin

Figure 15. «Disposable» syringe filled with shortacting insulin and the pen-like case
A new insulin pen MADI (Manual Device for Insulin Injection) was proposed, constructed and developed (Fig. 16). The MADI was one of the first insulin pens worldwide [5, 27–32].
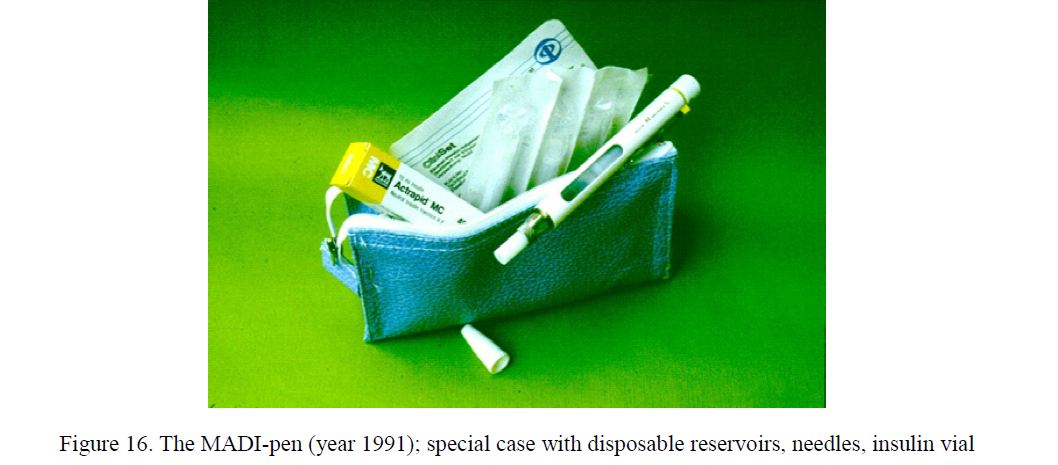
Figure 16. The MADI-pen (year 1991); special case with disposable reservoirs, needles, insulin vial
- The precision of MADI was tested In a technical study. The variability of individual doses is insignificant. In the type MADI 7/2, the average dose equals 1.9 IU and the coefficient of variation ranges between 0 and 4.6 %, exceptionally up to 5.7 %. Neither the speed of application, nor the temperature ranging between 20 and 37oC have any influence on the total dose injected. Even a simulated 5 years usage of the MADI did not lead to any significant change of individual doses.
- Pharmacological Under the standard laboratory conditions (Institute of diabetes Karlsburg 1987–1990) it was found out that within one year there is no significant change in the activity of shortacting porcine insulin when stored in a plastic reservoir under 37oC without mechanical stress (Fig. 17). If a heavy mechanical stress is added, the full insulin activity remains three days at least, and then it begans to decrease.
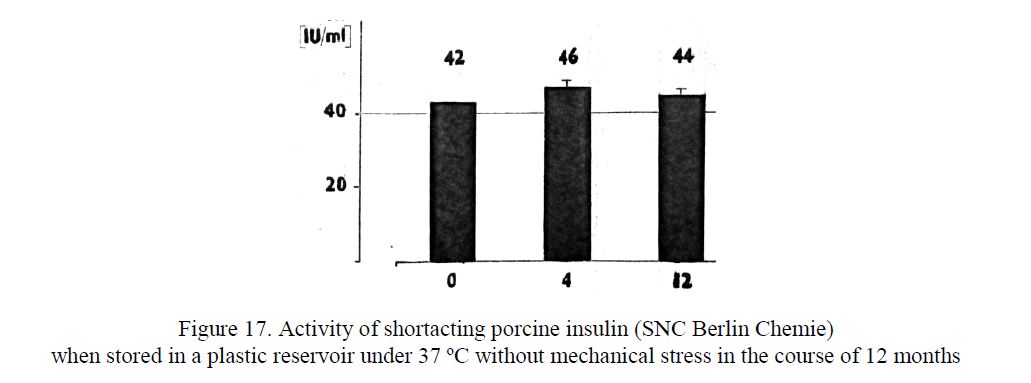
Figure 17. Activity of shortacting porcine insulin (SNC Berlin Chemie) when stored in a plastic reservoir under 37 ºC without mechanical stress in the course of 12 months
- Microbial Thorough microbial investigations and clinical observations of over 500 000 injection sites resulted in a conclusion that the repeated use of disposable syringes, reservoirs and needles in one PWD may be recommended as a convenient and safe approach in insulin administration. If no obvious contamination occurs, the period of usage of a needle seems to be limited by its sharpness [33].
- Clinical study. A prospective multicentre clinical study (1985–1992) confirmed that MADI is a useful aid for the application of all kinds of insulin in all age-groups of PWD1 and PWD2 which are able to handle the pen. No visual control of insulin injection is necessary. The MADI was produced from light metals and plastic materials. The insulin is filled from original vials. The filling and application is performed by twisting the The latest models of MADI from the year 1994 (40–1, 40–2, 100–1, 100–2, 80–1, 80–2) may be distinguished by color. The volume of a reservoir is 3 ml. The needle is protected by a special sliding cover. The MADI was used as a needle-pen or as a catheter-pen. Since 1986, more than 1800 MADI-pens were introduced at the Teaching Hospital Olomouc (Fig. 18).
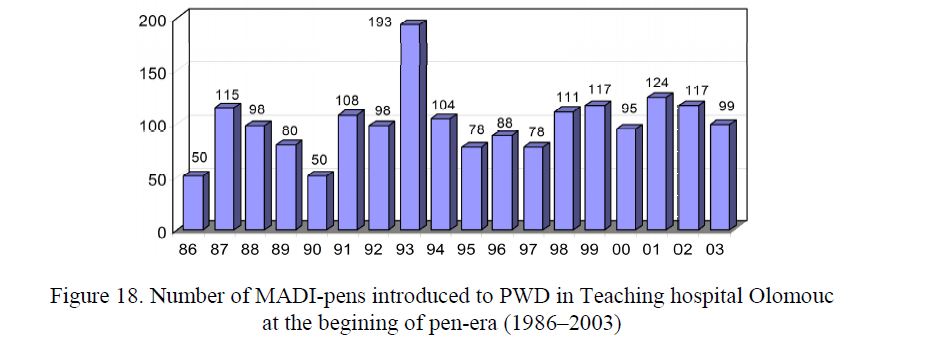
Figure 18. Number of MADI-pens introduced to PWD in Teaching hospital Olomouc at the begining of pen-era (1986–2003)
At present, the MADI production is over. MADI pens have been replaced by disposable flexpens prefilled with insulin aspart or by other pens. Despite of that, the experience and skills with MADI are effectively used on behalf of all PWD.
Insulin pumps. In the Teaching hospital Olomouc, the first insulin pump (Promedos E 1, Siemens) was introduced in December However, as late as at the end of the last century the CSII became a sophisticated therapeutic approach in several diabetes centres. To date, over 6000 men and women from more than 100 000 insulin treated PWD are profittig from various types of insulin pumps in the Czech Republic [34, 35].
Education — Programmed treatment of people with diabetes
Based on the concommittant clinical studies, principals of education comprising the principels of Therapeutic Patients Education of the WHO was introduced. The proposed educational schedule became a part of the so called «Programmed Treatment of persons with diabetes» which was proposed and checked on hundreds of diabetic patients as a part of the research grant on «The treatment of diabetes by means of an insulin pump and of the pen MADI. The Programmed treatment aims to an introduction of the intensive insulin therapy into the practice and has been composed of 4 parts: (1) diagnostic procedures, (2) introduction of an intensive insulin treatment, (3) patient´s education and supplying him with a pen and a glucose meter, (4) paitent´s self-decision dealing with his forthcoming treatment (acceptance or rejection of intensive insulin therapy). The PWD and the educator are substantial parts of any professional team dealing with diabetes treatment [36, 37].
- Glycaemic Index of foods (GI)
The glycaemic index (GI) is a measure of the food power to raise plasma glucose (PG) concentration after a meal. For its determination, classical methods register the development of glucose concentration in capillary plasma or whole blood in the course of 120 min [38, 39].
There is no standardized protocol for measuring glycemic index (GI) that takes the time-of-day effects into account. Using the CGMS, software CareLink and MS Excel the enhanced data processing software (Degif XL4 and recently Degif XL5) enabeling the GI calculation at breakfast, lunch, afternoon snack and dinner times has been made possible (Fig. 19).
Using the Degif XL5, the glucose concentrations of 20 volunteers are monitored after they consumed either 50 g of glucose or one of six alternative foodstuffs at breakfasts, lunches, snacks and at dinner times. Within the 9-day test period, 7 such meals were monitored in 5 replicates for each volunteer. Specifically, CGMS (monitor Guardian, Sofsensor and transmitter Minilink) was used to monitor plasma glucose levels at 5 minute-intervals for a period of 120 min following ingestion. At the end of day 9, the data from all volunteers are transferred into Carelink, to MS Excel and processed [40].
Any tests that did not fulfill the evaluation criteria (fasting before meal shorter than 210 min, consumption lasted longer than 30 min, incomplete portion or additional food was eaten or sensor failure appeared) were not processed. Next, GIs exceeding three times the interquartile interval were excluded. Such as 312 out of 350 tests (89 %) were analyzed.
The Shapiro-Wilk test showed a non-normal distribution of GI values. Median and 1st and 3rd quartiles were used to express the value of group-related GI. The evolution of plasma glucose in the course of 210 min after the meal start is demonstrated (Fig. 20–22).
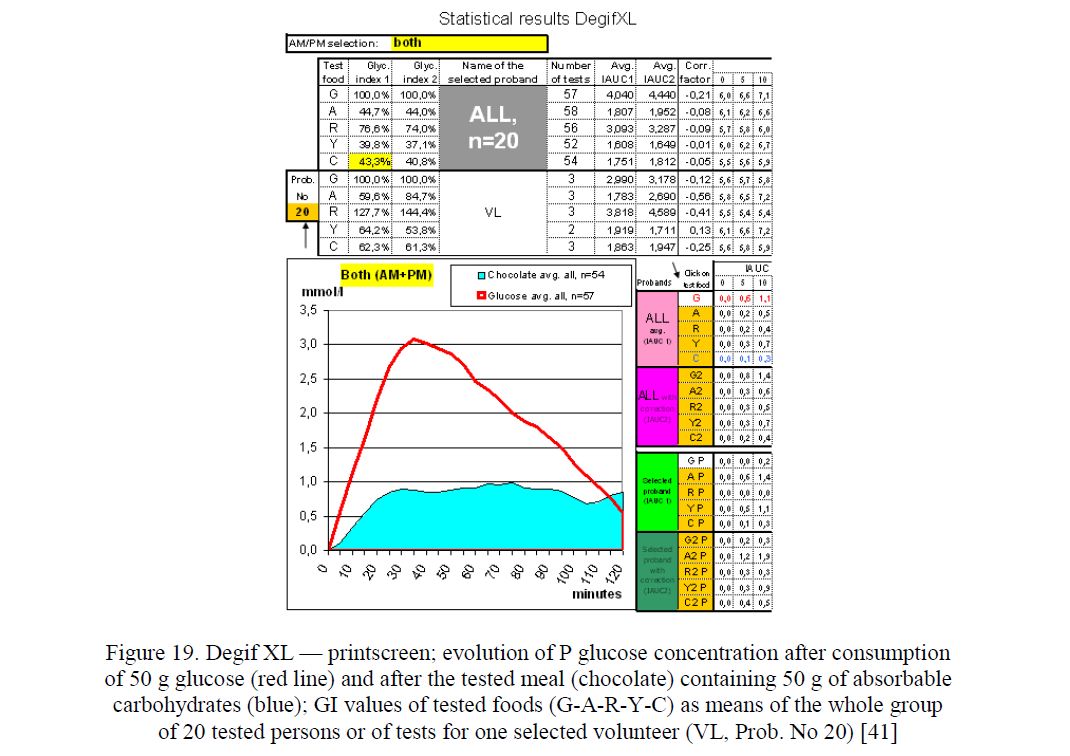
Figure 19. Degif XL — printscreen; evolution of P glucose concentration after consumption of 50 g glucose (red line) and after the tested meal (chocolate) containing 50 g of absorbable carbohydrates (blue); GI values of tested foods (G-A-R-Y-C) as means of the whole group of 20 tested persons or of tests for one selected volunteer (VL, Prob. No 20) [41]
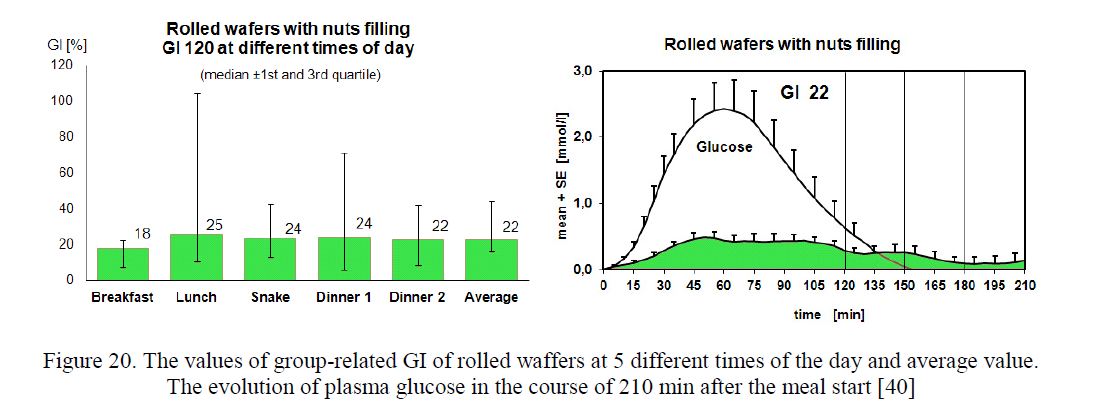
Figure 20. The values of group-related GI of rolled waffers at 5 different times of the day and average value.
The evolution of plasma glucose in the course of 210 min after the meal start [40]
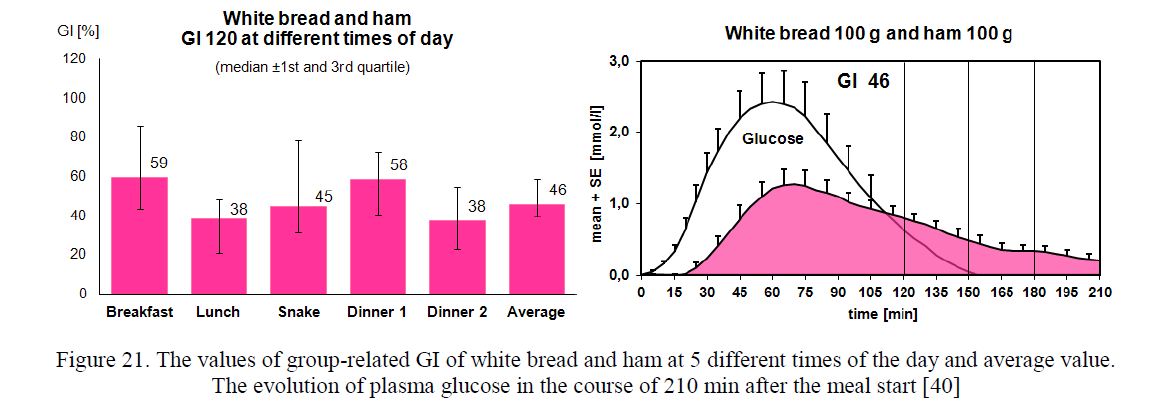
Figure 21. The values of group-related GI of white bread and ham at 5 different times of the day and average value.
The evolution of plasma glucose in the course of 210 min after the meal start [40]
![The average values of group-related GIs of 6 different foods [40]](/uploads/data/files/pics8/22.jpg)
Figure 22. The average values of group-related GIs of 6 different foods [40]
Our findings suggest that tests performed at different times of day using CGMS are an acceptable approach to GI determination. This approach appears to be worthy of consideration as an alternative to present methods according to the standard ISO 26642. This method appears to be useful to the assessment of therapeutic effectiveness of oral antidiabetic drugs etc. [42].
- Intensive selfmonitoring
The accuracy and precision of various glucometers was estimated [43–48]. Ten-point ambulatory glycaemic profile and continuous glucose monitoring (CGMS) was introduced to practice [49]. In persons on insulin pumps CGMS resulted in reduction of HBA1c [50].
Accuracy and precision of glucometer-strips systems was evaluated in several studies using different Within the course of 15 years we have tested the glucometer systems Card (Medisense), Optium (Abbott), Advance (Hypoguard, GB) and Linus (Agamatrix, USA) at our diabetes centre considering their accuracy when used in real life. The purpose of our recent trial was to assess the accuracy and precision of the electrochemistry-based glucometers CONTOURLINK, Bayer, Germany, using FAD glucose dehydrogenase strips, and CALLA, Welion, Austria, as well as LINUS, Agamatrix, USA, both using glucose oxidase strips. The tests performed with these glucometers resulted in acceptable results (Fig. 23) [49].
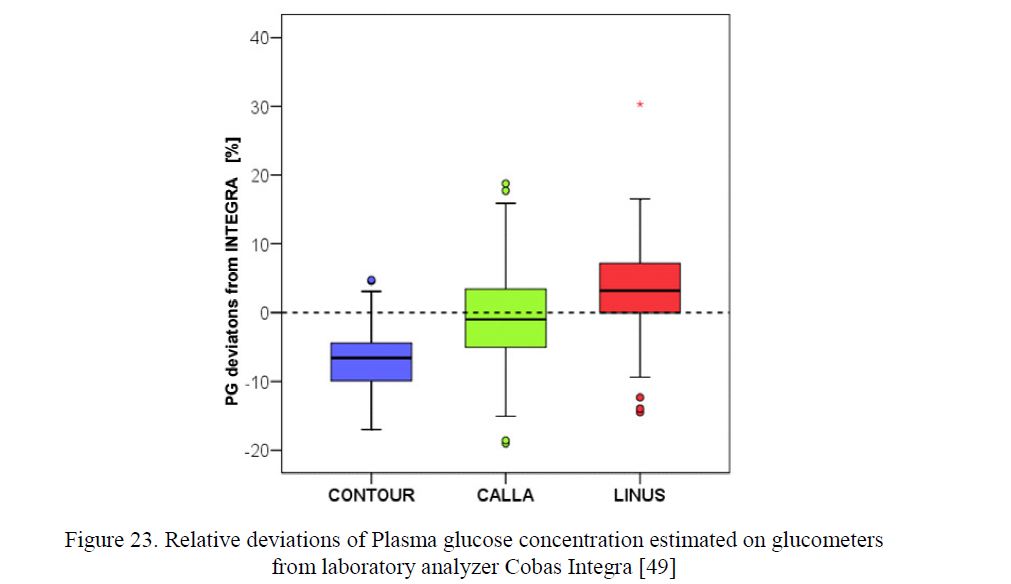
Figure 23. Relative deviations of Plasma glucose concentration estimated on glucometers from laboratory analyzer Cobas Integra [49]
- Ambulatory glycaemic We also paid attention to the assessment of diabetes control in real life using Ambulatory Glycaemic Profiles (AGP) as markers of therapeutic effectiveness (Fig. 24). In our centre, the ten-points ambulatory glycaemic profiles are performed as a substantial part of regular diabetes check-ups. The PWD´s are trained in AGP including corrections of timing of fingerpricks, meals and insulin application exceeding ± 15 min of the times printed on the AGP sheet. On the evaluation of an AGP is especially recommended:
- to compare the fasting PG values at 6,00 h at the beginning and at the end of the AGP to assess the stability of diabetes control;
- to pay attention to the evolution of PG between midnight and 6,00 h a.m.; the increase ≥ 0 mmol/L indicates a dawn phenomenon;
- to explain the postprandial PG variations over the day;
- to identify hypoglycaemias;
- to discuss all items dealing with insulin dosage, meals and exercise;
- to suggest
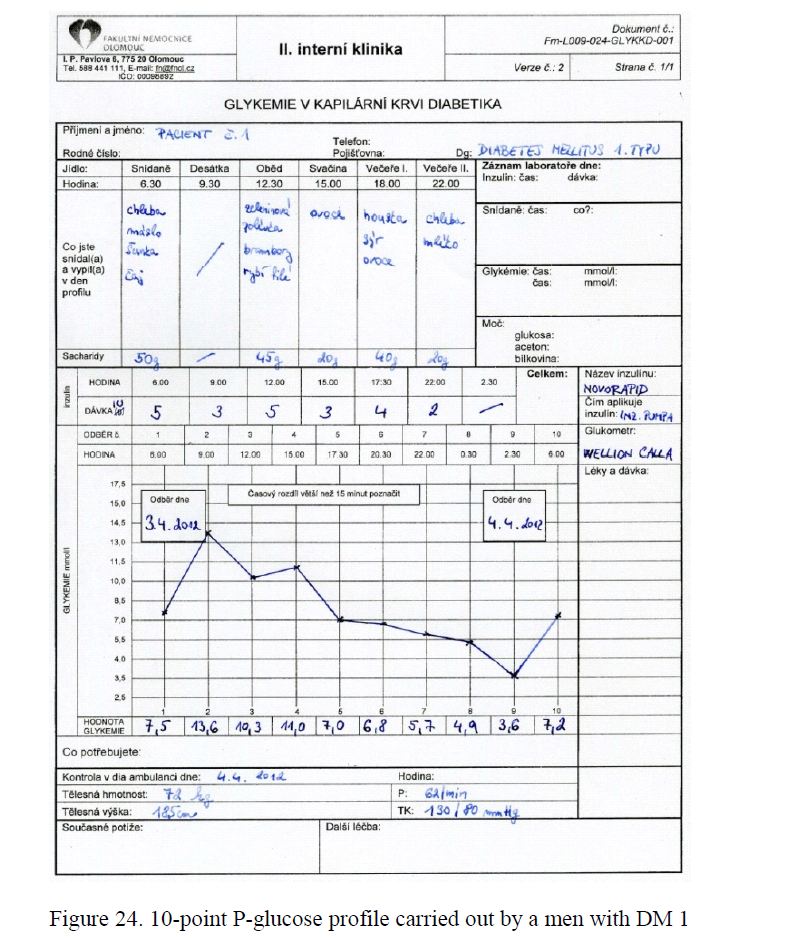
Figure 24. 10-point P-glucose profile carried out by a men with DM 1
Continuous glucose In the course of our study PARASEN (sensor augmented CSII using insulin pump Paradigm x22 or Paradigm x54) following observations were made: Since 1993, insulin pump treatment was started in 167 PWD1 or PWD2. Long-term Continuous Glucose Monitoring (Fig. 25) was shown to reduce HbA1c (Fig. 26). The purpose of a prospective study was to assess the real patient´s interest in routine use of transcutaneous sensors related to the hypothetical optimum,, always on CGM». In the course of 7 years (2006 to 2012) the sensor-augmentation of Continuous Subcutaneous Insulin Infusion (CSII) was repeatedly offered free of charge to all PWD on pumps (n=123) attending the regular check-ups supported by Carelink Personal software. The CGM was accepted for variable number of days by 63 (51 %) of them. Even after offering sensors and CGM education to all PWD free of charge, there seems to be lack of interest/motivation in PWD to try CGM [51].
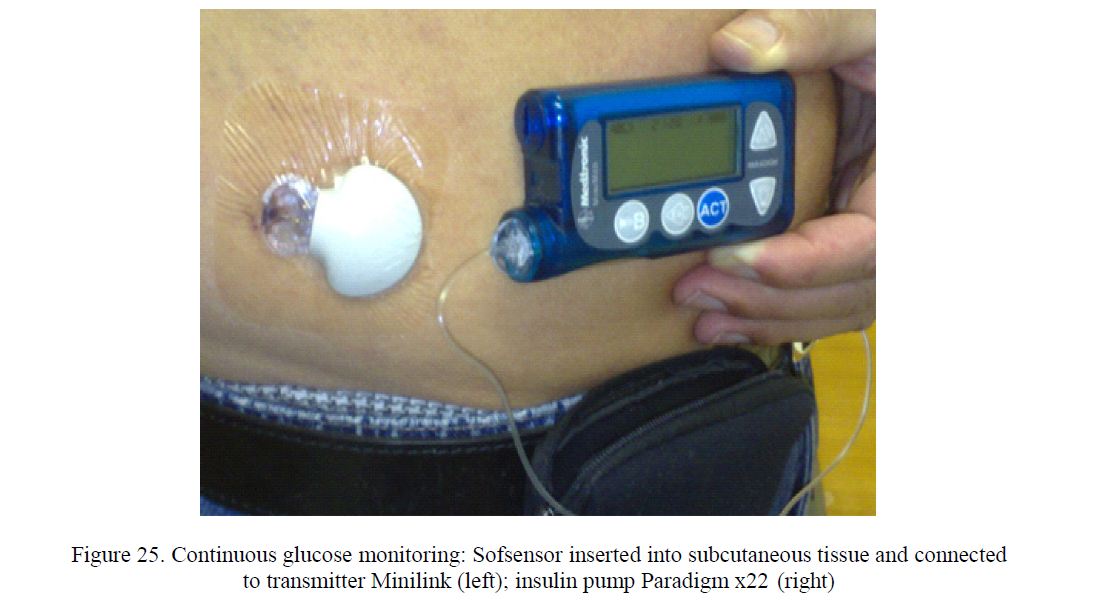
Figure 25. Continuous glucose monitoring: Sofsensor inserted into subcutaneous tissue and connected to transmitter Minilink (left); insulin pump Paradigm x22 (right)
¨![Evolution of HbA1c in PWD1 on CSII (insulin pump Paradigm x22, Medtronic Minimed, Northridge, CA, USA) when performing selfmonitoring of plasma glucose (SMPG) using glucometer-strips system Advance (Hypoguard, GB) and during the 3-month period with continuous glucose monitoring (CGM) using Sofsensor and Minilink (Medtronic Minimed) [50]](/uploads/data/files/pics8/26.jpg)
Figure 26. Evolution of HbA1c in PWD1 on CSII (insulin pump Paradigm x22, Medtronic Minimed, Northridge, CA, USA) when performing selfmonitoring of plasma glucose (SMPG) using glucometer-strips system Advance (Hypoguard, GB) and during the 3-month period with continuous glucose monitoring (CGM) using Sofsensor and Minilink (Medtronic Minimed) [50]
The positiv influence of CGMS on glycaemic variability and evolution of HbA1c [52–57], and on the other hand, a week acceptance (around 50 %) of this approach from men and women with diabetes may challenge both professionals in CGMS technology and professionals in patients´education to improve the outcomes of their endeavour. Occurrence of adverse events is very rare [58].
References
- Chlup R. The Diabetes Centre at Olomouc University in the Czech Republic // Practical Diabetes International. — 1997. — Vol. 14, No. 2. — P.
- Chlup , Menzel R., Keilacker H., Heinke P., Jutzi E. Night injections of regular insulin improve diabetes control in C-peptide negative patients // Diabetologia. — 1997. — No. 40 (Suppl). — P. 1292.
- Menzel , Chlup R., Felsing W., Witte K., Alt E., Jutzi E. Insulininjection devices connected to the body by subcutaneous catheter — a cheap alternative to highly sophisticated insulin pumps? // Diabetologia. — 1988. — No. 31. — P. 520A.
- Menzel R., Chlup R., Jutzi E., Hildmann W. «CatheterPens» an alternative to insulin pump treatment? // Exp. Endocrinol. — 1990. — Vol. 95. — P. 15764.
- Chlup , Janu K., Venhacova J., Bartek J. Six models of a new insulin pen (MADI): Description and first clinical trial // Practical Diabetes International. — 1995. — Vol. 12, No. 1. — P. 32–35.
- Chlup , Možíšová L. Léčba diabetiků pomocí inzulinové pumpy — teorie a praxe (Treatment of people with diabetes by means of an insulin pump — theory and practice) // Diabetologie Metabolismus Endokrinologie Výživa. — 2000. — Vol. 3, No. 1. — P. 3–17.
- Chlup , Zapletalová J., Seckar P., Malá E., Doubravová B., Táncosová S., Chlupová L., Pukowietz L., Zatloukal P. Benefits of complementary therapy with insulin aspart versus human regular insulin in persons with type 2 diabetes mellitus // Diabetes Technol. Ther. — 2007. — Vol. 9, No. 3. — P. 223–231.
- Zander E., Bruns W., Wulfert P., Besch W., Lubs D., Chlup R., Schulz B. Muscular Exercise in Type 1-Diabetics // Exp. Endocrinol. — 1983. — Vol. 82, No. 1. — P. 78–90.
- Zander , Schulz B., Chlup R., Woltansky P., Lubs D. Muscular exercise in Type-1-Diabetics: II. Hormonal and metabolic responses to moderate exercise // Exp. Clin. Endocrinol. — 1985. — Vol. 85, No. 1. — P. 95–104.
- Neoral L., Chlup R., Loyková V., Franková M., Zedníková K., Bartek J., Mazochová J. Vliv insulinu na metabolismus etylakoholu a vliv etylalkoholu na glykemii u diabetiků Typu (Influence of insulin on ethylalcohol metabolism and influence of ethylalcohol on glycaemia in type 1 diabetics) // Protialkoholický obzor. — 1991. — Vol. 26, No. 6. — P. 329–337.
- Chlup R., Neoral L. Methods for retrospective assessment of the unusual behavior of a person with diabetes: ebrietas alcoholica and/or hypoglycaemia? // Biomed. Papers. — 2004. — Vol. 148, No. 1. — P. 39–43.
- Bruns W., Fiedler H., Altman B., Lundershausen R., Menzel R., Worms F., Kriegstein E. Insulintherapie bei Typ 2 Diabetes Pathophysiologisch begründete Therapie mit insulin unter besonderer Berücksichtigung der Insulinresistenz und des — Auflage-Bremen, Germany: UNIMED Verlag AG, 2010.
- Chlup , Vaverkova H., Bartek J. Complementary insulin therapy improves blood glucose and serum lipid parameters in type 2 (non-insulin-dependent) diabetic patients. I. Effects on blood glucose control // Exp. Clin. Endocrinol. Diabetes. — 1997. — No. 105 (Suppl. 2). — P. 70–73.
- Vaverková H., Chlup R., Ficker L., Novotný D., Bartek J. Complementary insulin therapy improves blood glucose and serum lipid parameters in type 2 (non-insulin-dependent) diabetic patients: II. Effects on serum lipids, lipoproteins and apoproteins // Clin. Endocrinol. Diabetes. — 1997. — No. 105 (Suppl. 2). — P. 74–77.
- Chlup , Rancová A., Both P., Axmannová M., Vaverková H., Bartek J. Intensive komplementäre Insulintherapie bei adipösen Typ-2-Diabetikern hat meistens eine Gewichtsreduktion zur Folge // Diabetes und Stoffwechsel. — 1999. — No. 8 (Suppl). — P. 17.
- Chlup , Zapletalová J., Seckar P., Chlupová L., Táncosová S., Reznícková M. Benefits of insulin aspart vs phosphatebuffered human regular insulin in persons with type 1 Diabetes treated by means of an insulin pump // Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. — 2004. — Vol. 148, No. 1. — P. 27–32.
- Home D. The pharmacokinetics and pharmacodynamics of rapid acting insulin analogues and their therapeutical consequences // Diabetes Obesity and Metabolism. — 2012. — Vol. 14. — P. 780–788.
- Heller S., Bode B., Kozlovski P., Svendsen A.L. Meta-analysis of insulin aspart versus regular human insulin used in a basalbolus regimen for the treatment of diabetes mellitus // Journal of Diabetes. — 2013. — No. 5. — P. 482–491.
- Chlup R., Vaverková H., Bartek J. Long-Term Benefits of Complementary Insulin Therapy on Body Mass and Hemoglobin A1c in Obese Type 2 Diabetic Patients // Diabetes Research and Clinical — 2000. — No. 50 (Suppl. 1). — P. S66–S67.
- Chlup R., Peterson K., Zapletalová J., Kudlová P., Matušková V., Ďurajková E., Langová K. Impact of continuous subsutanous insulin infusion on metabolic parameters and well-being in persons with type 2 diabetes // Diabetes. — 2010. — No. 59 (Suppl. 1). — P.
- Chlup R. Benefits of Intensive Selfmonitoring and Continuous Subcutaneous Insulin Infusion in Persons with Type 2 Diabetes Mellitus // BITs 1st Annual Worlds Congress of Endobolism: Abstract. — Xiamen, China, 2011. — P.
- Aronson , Cohen O., Conget I., Runzis S., Castaneda J., de Portu S., Lee S., Reznik Y. For the OpT2mise Study Group: OpT2mise: a randomized controlled trial to compare insulin pump therapy with multiple daily injections in the treatment of type 2 diabetes — research design and methods // Diabetes Technology & Therapeutics. — 2014. — Vol. 16, No. 7. — P. 414–420.
- Reznik Y., Cohen O., Aronson R., Conget I., Runzis S., Castaneda J., Lee S.W. Insulin pump treatment compared with multiple daily injections for treatment of type 2 diabetes (OpT2mise): a randomised open-label controlled trial // The Lancet. — 2014. — Vol. 384, No. 9950. — P. 1265–1272.
- Brenner M., Cooper M.E., de Zeeuw D., Keane W.F., Mitch W.E., Parving H.H., Remuzzi G., Snapinn S.M., Zhang Y., Shahinfar S. For the RENAAL Study Investigators (... Chlup R,...) Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy // New Engl. J. Med. 2001. — Vol. 345, No. 12. — P. 861–869.
- Chlup R., Gazárek F., Krikal Z., Flasarová B. Diabetes mellitus a tehotenstvi [Diabetes mellitus and gravidity (author's transl)] [Article in Czech] // Cesk Gynekol. — 1976. — Apr; Vol. 41, No. 2. — P. 113–115. PMID:
- Poljakova I., Elsikova E., Chlup R., Kalabus S., Hasala P., Zapletalova J. Glucose sensing module — Is it time to integrate it into real-time perioperative monitoring? An observational pilot study with subcutaneous sensors // Biomedical Pap. Med. Fac. Palacky Olomouc Czech Repub. — 2013. — Vol. 157, No. 4. — P. 346–357.
- Chlup R., Menzel R., Kolesár P., Rybka J., Svoboda Z., Škarpová O., Venháčová J., Zander E., Abel P., Rosenfeld R., Hildebrandt R., Janů K., Jutzi E. Erste Erfahrungen mit dem MADI — multizentrische technische und klinische Studie // VIII Internationales Donau-Symposium über Diabetes mellitus. — Bratislava, 18–21 Juni 1985. — Wien: Robidruck, 1985, Abstract P, (nestr).
- Chlup R, Janů K, Menzel R, Bruns W, Venháčová Léčba diabetu pomocí ručního dávkovače inzulinu MADI (Diabetes treatment by means of manual device for insulin MADI) // Ed. Univerzita Palackého v Olomouci, 1985.
- Felsing , Menzel R., Alt P., Chlup R., Bruns W. Erste Erfahrungen mit dem MADI in der DDR // W Bruns (ed) Lehrgang über aktuelle Probleme des Diabetes mellitus. — Berlin 24–28.11.1986 (Kongressberichte) Edn Akademie für ärztliche Fortbildung der DDR. — P. 60.
- Bibergeil (Ed). Diabetes mellitus — ein Nachschlagewerk für die diabetologische Praxis. — Edition 3. — VEB Gustav Fischer Verlag Jena, 1988.
- Berger , Jörgens V., Chlup R. Léčba inzulinem v každodenním životě. — Ed. 1. — Praha: Victoria Publishing, 1995.
- Janů K., Chlup R. Konstrukční řešení ručního dávkovače inzulínu MADI (Construction of manual device for insulin MADI) // Jemná mechanika a optika. — 1992. — Vol. 37, No. 1. — P. 14–15.
- Chlup , Maršálek E., Bruns W. Prospective study of the hazards of multiple use of disposable syringes and needles in intensified insulin therapy // Diabetic Medicine. — 1990. — No. 7. — P. 624–627.
- Jankovec , Hahn M., Grunder S., Lacigova S., Cechurova D., Krcma M., Zourek M., Haladova I., Rusavy Z. Analysis of continuous patient data from the Czech National Register of patients with type 1 and type 2 diabetes using insulin pump therapy // Diabetes Research and Clinical Practice. — 2010. — Vol. 87, No. 2. — P. 219–223.
- Chlup R., Tomcalova J., Kudlova P., Zapletalova J. Pump away: patients' rejection of continuous subcutaneous insulin infusion, physician's decision to switching to other treatments and death rates // Diabetes Technology & Therapeutics. — 2013. — No. 5 (Suppl. 1). — P.
- Chlup , Navrátilová L., Řehořová J., Vaverková H., Venháčová J., Bartek J., Ficker L., Slezáková L. Programová léčba diabetu (Programme therapy of diabetes). — Ed. 1. — Praha: Galén, 1996.
- Chlup R., Holešinská P., Chlupová L., Komenda S. Assessment of postgraduate education of therapeutic educators (PETE) at a university diabetes centre // Diabetes Metab. — 2003. — Vol. 29, No. 4. — P.
- Chlup , Jelenova D., Kudlova P., Chlupova K., Bartek J., Zapletalova J., Langova K., Chlupova L. Continuous glucose monitoring — a novel approach to the determination of the glycaemic index of foods (DEGIF 1) // Exp. Clin. Endocrinol. Diabetes.
- — 2006. — Vol. 114. — P. 68–74.
- Fajkusova Z., Jadviscokova T., Pallayova M., Matuskova V., Luza J., Kuzmina G. Glycaemic index of selected foodstuffs in healthy persons // Biomed Med. Fac. Univ. Palacky. Olomouc Czech Repub. — 2007. — Vol. 151, No. 2. — P. 257–261.
- Chlup , Peterson K., Kudlová P., Zapletalová J., Langová K., Sečkař P., Milata V. Determination of glycemic indexes of foods at different times of day in healthy persons using continuous glucose monitoring // Diabetes. — 2011. — No. 60 (Suppl. 1). — P. 212–213.
- Pribylova H., Pallayova M., Hucikova J., Luza J. Evaluation of the new software program DEGIFXL4 in the determination of the glycaemic indices of foodstuffs // Biomed Pap. Med. Fac. Univ. Palacky. Olomouc Czech Repub. — 2008. — Vol. 152, No. — P. 65–71.
- Peterson , Chlup R., Zapletalova J., Kohnert K.D., Kudlova P., Bartek J., Nakladalova M., Doubravova B., Seckar P. Influence of oral antidiabetic drugs on hyperglycemic response to foods in persons with type 2 diabetes mellitus as assessed by continuous glucose monitoring system: a pilot study // J. Diabetes Sci. Technol. — 2010. — Vol. 4, No. 4. — P. 983–992.
- Chlup , Payne M., Zapletalová J., Komenda S., Doubravová B., Reznícková M., Chlupová L., Seckar P. Results of selfmonitoring on glucometer systems Advance and Optium in daily routine // Biomed Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. — 2005. — Vol. 149, No. 1. — P. 127–139.
- Chlup , Bartek J., Malá E., Doubravová B., Pukowietz L., Zatloukal P., Chlupová L., Zapletalová J. Uživatelská studie o správnosti a přesnosti měření glukometrů Advance, Card a Optium (User oriented study on accuracy and precision of glucometer systems Advance, Card and Optium) // Klin. Bioch. Metabol. — 2004. — Vol. 12. — No. 3. — P. 171–178.
- Chlup , Bartek J., Zapletalová J., Sečkař P., Pukowietz L., Řezníčková M., Havrdová M., Rulfová L. Výsledky měření P-glukosy na glukometrech Advance a Optium v laboratorních podmínkách (Results of P-glucose estimations by means of glucometers Advance and Optium under laboratory conditions) // Klin. Bioch. Met. — 2004.— Vol. 12, No. 3. — P. 160–170.
- Chlup , Doubravova B., Peterson K., Zapletalova J., Bartek J. Wavesense technology glucometer Linus for routine selfmonitoring and clinical practice // Acta Diabetol. — 2011. — Vol. 48, No. 1. — P. 35–40.
- Chlup R., Doubravova B., Zapletalova J., Langova K., Bartek J. Glucometers (CALLA) appear to be more convenient means for routine P-glucose monitoring in patients admitted to hospital than a laboratory analyzer (COBAS INTEGRA) // Diabetes Technol. Ther. — 2011. — Vol. 13, No. 2. — P. 214–215.
- Chlup R., Doubravova B., Zapletalova J., Peterson K., Bartek J. Plasma glucose concentrations on new glucometer CALLA strongly correlate with laboratory analyser COBAS INTEGRA 400 PLUS under various clinical conditions // Diabetes Ther. — 2011. — Vol. 13, No. 2. — P. 213–214.
- Chlup , Doubravova B., Bartek J., Zapletalova J., Krystynik O., Prochazka V. Effective Assessment of Diabetes Control Using Personal Glucometers (CONTOURLINK, Bayer, Germany; CALLA, Wellion, Austria; LINUS, Agamatrix, USA) // Disease Markers. — 2013. — Vol. 35, No. 6. — P. 895–905.
- Chlup R., Přibylová H., Peterson K., Langová K., Matušková V., Kudlová P., Táncosová S., Luža J. Demands for continuous glucose monitoring (CGMS) in persons with diabetes on insulin pumps Paradigm X22 // Diabetes. — 2008. — No. 57 (Suppl. 1). — P. 548.
- Peterson K., Zapletalova J., Kudlova P., Matuskova V., Bartek J., Novotny D., Chlup R. Benefits of three-month continuous glucose monitoring for persons with diabetes using insulin pumps and sensors // Biomed Med. Fac. Univ. Palacky Olomouc Czech Repub. — 2009. — Vol. 153, No. 1. — P. 47–52.
- Mlcak , Fialová J., Trnková K., Chlup R. A continuous glucose monitoring system (CGMS) — a promising approach for improving metabolic control in persons with type 1 diabetes mellitus treated by insulin pumps // Biomed Pap. Med. Fac. Univ. Palacky Olomouc, Czech Repub. — 2004. — Vol. 148. — P. 33–38.
- Kohnert K.-D., Heinke P., Vogt L., Meyramov G.G., Salzsieder E. Optimizing diabetes management: comprehensive analysis of glucose monitoring data and use of better metrics for glycemic control // of the Karaganda University. — 2013. — No. 4(72). — P. 6–15.
- Kohnert D., Heinke P., Vogt L., Salzsieder E. Utility of different glycemic control metrics for optimizing management of diabetes // World J. Diabetes. — 2015. — Vol. 6, No. 1. — P. 17–29; Available from: URL: http://www.wjgnet.com/1948– 9358/full/v6/i1/17.htm. DOI: http://dx.doi.org/10.4239/wjd.v6.i1.17.
- Kohnert K.D., Augstein P., Heinke P., Zander E., Peterson K., Freyse E.J., Salzsieder E. Chronic hyperglycemia but not glucose variability determines HbA1c levels in wellcontrolled patients with type 2 diabetes // Diabetes Research and Clinical — 2007. — Vol. 77. — P. 420–426.
- Kohnert D., Augstein P., Zander E., Heinke P., Peterson K., Freyse E.J., Hovorka R., Salzsieder E. Glycemic variability correlates strongly with postprandial β-cell dysfunction in a segment of type 2 diabetic patients using oral hypoglycemic agents // Diabetes Care. — 2009. —Vol. 32, No. 6. — P. 1058–1062.
- Cohen , Körner A., Chlup R., Zoupas C.S., Ragozin A.K., Wudi K., Bartaskova D., Pappas A., Niederland T., Taybani Z., Barák L., Vazeou A. Improved glycemic control through continuous glucose sensor-augmented insulin pump therapy: Prospective results from a community and academic practice patient registry // Journal of Diabetes Science and Technology. — 2009. — Vol. 3, No. 4. — P. 804–811.
- Jadviscokova , Fajkusova Z., Pallayova M., Luza J., Kuzmina G. Occurence of adverse events due to continuous glucose monitoring // Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. — 2007. — Vol. 151, No. 2. — P. 263–266.