Alteration and destruction of pancreatic B-cells caused by chemicals are one of numerous causes of developing of diabetes mellitus. Authors demonstrated results of investigation of mechanisms developing of diabetes caused by some of 18 diabetogenic derivatives of 8-oxyquinolin including chemicals formed in Human as result of disturbances of metaboilsm of aminoacids. Authors proposed a few ways for prevention of diabetes developed as result of their action and proposed for possible using of method inhibition of endogene synthesis of diabetogenic metabolites of Tryptophan as more suitable way for prevention developing of diabetes.
Today there are more than 30 chemicals which are able to induce experimental diabetes mellitus by selective destruction of B-cells. More than 20 of their possess chelat diabetogenic properties. 18 from them belong for derivatives of 8-oxyquinolin.
More than 70 years ago Scott and Fischer were separated insulin from the native pancreas as insulin-Zn complex and supposed that the presence of Zn-ions determined physiological activity of insulin [1]. Interest to this problem was increased after reporting by these authors in 1938 that in pancreas of death diabetic patients total amount of Zn is not more than 50 % in compared with non diabetic men [2]. They found 0,07 mg of Zn per 1 g of pancreas tissue of diabetic patients comparatively with 0,14 mg per 1 g pancreas of healthy persons. Analogical result was obtained by Eisenbrandt and coll. [3]. A large amount of Zn were found in human pancreas of healthy men. In 1942–1943 K.Okamoto discovered in pancreatic B-cells a large amount of Zn [4–12]. It is supposed today the important role of Zn-ions in processes of storage of insulin in B-cells [13]. There are proportional dependence between content of Zn-ions in B-cells and in cytoplasm. Decreasing of content of deposited insulin accompanied by decreasing of Zn-ions in B-cells [12, 14]. It is known that Zn-ions reacted in processes of synthesis as in cristallization of insulin [4]. It was showed that pancreas of mammals-animals, birds and in earth-water animals contained a large amount of Zn-ions.
The amount of Zn is evidently decreased in experimental diabetes induced by any causes [9, 10, 14–16]. Zn-ions are able be accumulated in pancreas tissue. Administration of Zn in organism outside accompanied by increasing of total amount in pancreas in 4–20 times [17]. 0,3 % of Zn administrated in organism was accumulated in pancreas of alloxan diabetic rats comparatively with 2,6 % in healthy animals [18]. H.Kawanishi and K.Okamoto confirmed [19, 20] by electron histochemical microscopy that in B-cells Zn-ions are located in B-granules, a deposited form of insulin and that Zn is concentrated in central part of B-granules, in periphery and partly in cover of granules.
Zn-ions contained in cytoplasm of B-cells have the coordinate number (chemical coordinate number) 4 and 6 and interacted with chemicals which formed with Zn-ions chelat salts in which atom of Zn is fixed between a few other atoms [21]. The affinity of Zn-ions to formation of chelats is evidently more high comparatively with other metals of main group.
- Diabetogenic derivatives of 8-oxyquinolin
In 1947 A.Albert reported that 8-oxyquinolin which usually is not toxic substance, is very toxic for cells in the presence of metals and for the first time of Zn-ions. It was showed that this fact determined by ability of 8-oxyquinolin to form with metals the chelat metal-complexes which are toxic for B-cells [21]. Studying of toxicity of 8-oxyquinolin for B-cells Okamoto K. [7, 20, 22] reported that injection of it to animals accompanied by developing of experimental diabetes. Later it was showed that injection of 18 derivatives of 8-oxyquinolin and of 8-oxyquinaldin accompanied by rapid developing of heavy diabetes in animals [7].
It was noted that all these chemicals have in position 8 of quinolin ring OH– group or any other radical contained atom of S or atom of O. Six isomers of 8-oxyquinolin which not contained in position 8 of the active group are not able to form chelat complexes with Zn-ions and not induced experimental diabetes. Experimental diabetes is induced by derivatives: 8-para(toluenesulphonylamino)quinolin /8PTSQ/, 8-para(benzolsulphonylamino)quinolin /8PBSQ/, 8-para(methansulphonylamino)quinolin /8PMSQ/, 5-para(acetaminophenylaso)-8-oxyquinolin /5A8OX/, 8-hydroxyquinaldin, 5-amino-8-hydroxyquinolin and others (Fig. 1). It was demonstrated by Okamoto K. and Kadota I. that injection of these derivatives result strongly selective necrosis of B-cells and developing of diabetes. Injection of these chemicals in doses of 30–100 mg/kg accompanied by developing within a few days of heavy diabetes with marked degenerative changes in islets [23–28].
- 2,4-dimethyl-8-oxyquinolin, 35 mg/kg; b) 5-phenylaso-8-oxyquinolin, 20 mg/kg; c) 5-para(toluene)-8oxyquinolin, 20 mg/kg; d) 5-orto(toluene)-8-oxyquinolin, 40 mg/kg; e) 8-oxyquinolin, 50–60 mg/kg;
- f) 5-para(diethylaminophenylaso)-8-oxyquinolin, 20 mg/kg; g) 5-meta(hydroxyphenylaso)-8-oxyquinolin, 30 mg/kg; h) 5-para(dimethylaminophenylaso)-8-oxyquinolin, 45 mg/kg; i) 5-para(acetylaminophenylaso)-8-oxyquinolin, 50 mg/kg; к) 8-oxyquinaldin, 10 mg/kg; l) 5-para(aminophenylaso)-8-oxyquinolin, 10 mg/kg; m) 5-amino-8-oxyquinolin, 30 mg/kg; n) 5-para(diethylaminophenylaso)-8-oxyquinolin, 40 mg/kg; о) 9-oxy-7-jodoquinolin, 50–60 mg/kg; p) 4,8–dihydroxyquinolin-2-carboxylic acid (xanthurenic acid); r) 8-para(toluenesulphonylamino)quinolin, 30–50 mg/kg; s) 8-para(benzolsulphonylamino)quinolin, 30–100 mg/kg; t) 8-para(metansulphonylamino)quinolin, 40–81 mg/kg; u) diphenylthiocarbazone (dithizon), 45–50 mg/ kg
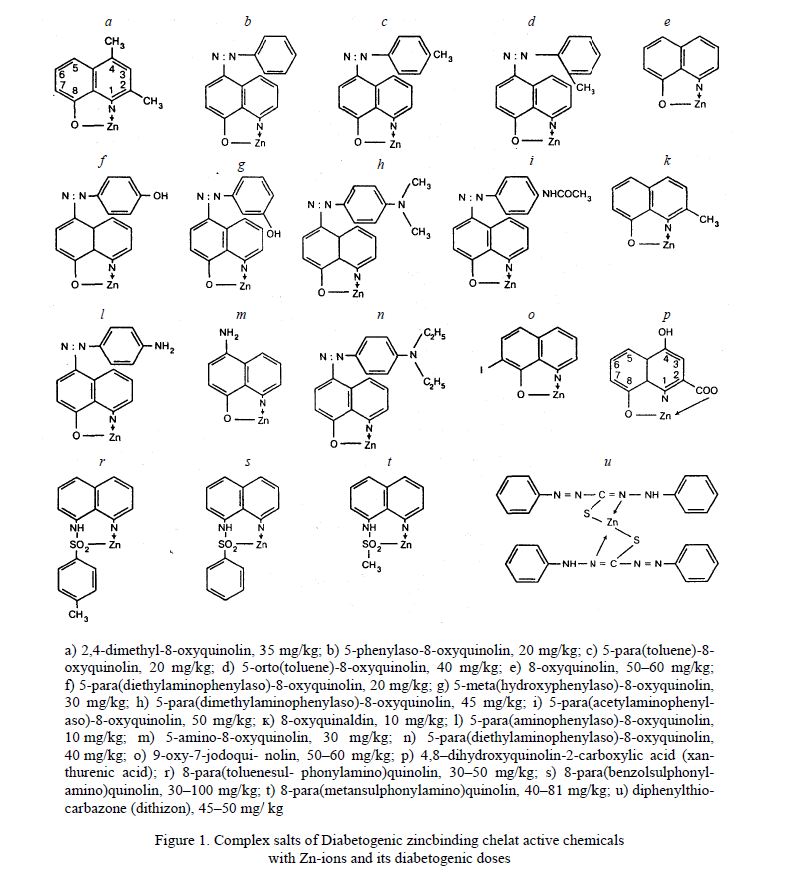
Figure 1. Complex salts of Diabetogenic zincbinding chelat active chemicals with Zn-ions and its diabetogenic doses
It is known that most stable complexes are formed in case if atom of Zn is fixed between 2 atom of N, S and O of molecule of chelator. Later it was reported that only derivatives of 8-oxyquinolin contained in position 8 of quinolin ring of the hydroxyl or other radical contained atoms of S, N or O possess diabetogenic properties. Atom of Zn is fixed between atoms of S and O in position 8 and between atoms of N and O in position 1 or 2.
It was reported, what is more, that extraction of these radicals from position 8 accompanied by complete disappearing of diabetogenic properties of chelators [29]. Formation of chelats by atoms of O and N of chelator result usually forming of pentagonal or hexagonal rings [21] (Fig. 1). Pentagonal rings are more stable. The most stable are quadrangular complexes with atom of S. It is known that derivatives of 8-oxyquinolin formed quadragonal complexes with atom of S often. Electrons of indivisible pair are displaced from donor-atom of N in position 1 to Zn atom.
On the base of data obtained by A.Albert, G.Zentmyer supposed that toxic effect of 8-oxyquinolin is determined by its ability to bind and eliminate ions of metal from B-cells [30]. But later this hypothesis was not confirmed: it was showed that long time prolonged elimination of Zn ions from B-cells result any effect on the state of histostructure and function of B-cells [31]. Finally, S.Rubbo and A.Albert established that toxic effect of 8-oxyquinolin determined by its ability to form in cells toxic complexes with metals [32] that many times was confirmed later. It was showed that presence of chelat a short time in cytoplasm of B-cells accompanied by alteration of cells. In experiences with using derivatives of 8-oxyquinolin — a various isomers of the azaoxyquinolin (azaoxyn) — it was demonstrated dependence: most toxic are isomers formed chelats 1:1 with metal and with logarifm of constant of stability as 7.6 and more high, until 9.4. Meanwhile toxicity of chelats of other isomers of azaoxyn with constant of stability 5.8–6.7 was clearly more less [21]. It was showed that very toxic chelats of derivatives of 8-oxyquinolin with Zn-ions have a more high logarifm of constant of stability as 8.5. Weitzel G. and coll. showed that complex 1:1 contained 1 molecule of 8-oxyquinolin and 1 atom of ion of Zn is most toxic for cells [33].
Stability of formed complexes 2:1 is depended not only of affinity of chelator to metal but in added — of 2 properties of chelator and metal:
1) presence of additional radicals in para-positions molecule of chelator, especially — in zones contacted with part of molecule, reacted with ions of metal conduce to forming of the steric effect; as result, two molecules of the chelator are not able to approach for to put atom of metal in stable ring;
2) size of diameter of atom; in case if atom of metal have a small diameter, ring may be not formed; atom of Zn have radius as 0,74 nm between Berillium (0,31 nm) and Rubidium (1,49 nm). A high stability of the complex Zn-Dithizon is determined by stretch form of molecule of Dithizon and by location of 2 phenol rings on the 2 ends of molecule.
That is why atom of N and S are easy approach to atom of Zn. More over, atom of Zn is fixed between atoms of N and S. Meanwhile it is known that affinity of Zn to N and S is more high comparatively with affinity of Zn to O. In added, complex is formed by two molecule of Dithizon each of two have a great number of double couplings.
Mechanisms of diabetogenic action of derivatives of 8-oxyquinolin and Dithizon were investigated since 1967. For the first it was showed that injection of diabetogenic doses of derivatives of 8-oxyquinolin (D8OX) 1–2 min past injection accompanied by complete binding of all amount of Zn-ions in cytoplasm of B-cells. 1.5–2 h later this complex is dissociated and same amount of Zn-ions in B-cells is reveal as before injection [14, 34–36].
Stability of complexes 1:1 formed by derivatives of 8-oxyquinolin is determined by: 1) great number of double coupling in molecule of chelator; 2) forming of quadragonal ring; 3) derivatives of 8-arensulphonylaminoquinoline formed chelat-complex by aid of atom of S. More high stability of the complex Zn-Xanturenic Acid is determined by additional fixation of the atom of Zn between 2 atom of O.
Later it was showed that 8PTSQ, a derivative of 8-oxyquinolin, formed with Zn ions toxic chelats which in UV-light have specific intensive green fluorescence. This fact was used for elaboration of high specific and very high sensitive fluorescent method of revealing of Zn ions [13, 37, 38].
This amount of Zn is able to form a new chelat-complexes with new portion of diabetogenic substance again. Extraction of complexes Zn-D8OX from B-cells by CHCl3 or by CCl4 result completely negative fluorescent reaction for Zn-ions in B-cells. This complex dissociated within 1–2 h and Zn-ions are able again to react with chelator [14, 35].
- On the mechanisms of Diabetogenic activity of chelat active substances
In 1949 K.Okamoto first induced experimental diabetes by injection of Dithizon [39]. Dithizon are able to form red chelat complexes with 18 metals; Zn-ions only contained in pancreatic B-cells of some animals and human. Dithizon possess very high affinity to Zn-ions and rapidly reacted with forming complex DZ-Zn 2:1. Dithizon is not synthesed in organism as of animals as human. Later Maske [40] proposed vital method of colour detection of Zn-ions in B-cells based on ability of Dithizon to form purple granules of Zn-dithizonat past injection of Dithizon solution. It was noted that diabetes induced by DZ accompanied by formation of red granules of chelat Zn-DZ in B-cells. Diabetes never developed in case if red granules are not formed in cytoplasm of B-cells. By aid of this method Zn-ions were discovered in islets of rabbits, human, rats, pigs, mice, dogs, horses, pigeons, frogs, some sorts of fish and other animals, excluding guinea pig only which not contained Zn in B-cells [22, 41–47]. As it was showed later Dithizon not formed red granules in B-cells of guinea pig and diabetes in this case not developed [34]. Later it was confirmed by spectral analysis that spectrum of absorbance of purple granules formed in B-cells past injection of Dithizon exactly correspond to spectrum of absorbance of pure synthetic Zn-DZ chelat [34].
K.Okamoto supposed that diabetogenic action of Dithizon determined by its ability to form in B-cells of chelat complexes with Zn-ions. He concluded finally: the binding of Zn-ions in B-cells by DZ is main cause of developing of diabetes. This suggestion was confirmed many times later. For the first it was confirmed that diabetes developed past injection of DZ in case if purple granules of Zn-DZ is formed in cytoplasm of B-cells only. Meanwhile according conception of Okamoto K. is not possible to understand what are mechanisms of diabetogenic action of Dithizon on B-cells as of chelator.
There are important question: does this complex Zn-DZ is eliminated from B-cells or this complex is dissociated in B-cells and Zn-ions are preserved in islets and are able to interact again with DZ?
It was established that 30 min past injection the total amount of red granules of Zn-DZ in cytoplasm of B-cells is evidently decreased and 1 h past injection (mices) and 1,5–2 h (rabbits) red granules disappeared completely from islets. In opposite, parallelly at the same time the amount of free Zn-ions in B-cells is increased and 1,5–2 h past injection concentration of Zn-ions in cytoplasm of B-cells is same as before injection of Dithizon [34, 35]. Meanwhile it was showed that injection of diabetogenic dose of Dithizon result a complete binding of all amount of Zn-ions contained in B-cells: past extraction from cytoplasm of B-cells of all amount of complex DZ-Zn by CHCl3 free Zn-ions were not revealed in cytoplasm of B-cells by absolutely and high specifical for Zn-ions fluorescent method. However, 1–1,5 h later concentration of free Zn-ions in cytoplasm of B-cells was maximally high as before injection of Dithizon. Meanwhile it was not possible to have same result in case if Zn-ions were eliminated from cytoplasm of B-cells.
Thus, it was confirmed that injection of Dithizon accompanied by forming of chelat Zn-DZ in cytoplasm of B-cells, which is dissociated in cells within 1–2 h past injection of chelator and free Zn-ions are able to form chelat complexes again with chelator in cytoplasm of B-cells.
Later it was showed that in animals at +36 ºC without circulation of blood dissociation of complex DZ-Zn is markely delayed: 3 h past injection of Dithizon 50–60 % of granules contained in cytoplasm of B-cells are not dissociated yet [34, 35].
However later it was supposed that injection of chelator result binding of Zn-ions in Zn-contained enzymes that result inactivation of enzymes and as final — disturbances of metabolism in cells and developing of histological changes. But this conception was not confirmed later. It is known that metal-contained enzymes usually fixed strongly atom of metal in structure of molecule enzyme and chelators are not able to form chelat-metal complexes. According other conception, complete elimination of Zn-ions from B-cells protect cells of formation of toxic complexes Zn-diabetogenic chelator but in this same time result disturbances of processes of synthesis and storage of insulin in B-cells. But this hypothesis was not confirmed too, because it was showed, that complete binding of Zn-ions in B-cells by Zn-diabetogenic chelator not accompanied by elimination of complex. As it was evidently established, this complex is dissociated in cytoplasm of B-cells within 1–2 h and Zn-ions are not eliminated from cytoplasm of B-cells.
In opposite, it was showed, that complete elimination of Zn-ions from cytoplasm of B-cells by Glibenclamide not accompanied by changes of structure and function of B-cells [48]. Injection of Dithizon to animals pretreated by Glibenclamide not accompanied by forming of complex Zn-DZ in cytoplasm of B-cells and diabetes is not developed.
As is known, Zn-ions contained in rethina of eye and injection of DZ to rabbit accompanied by forming of chelat-complex Zn-DZ in retina which result blindness [21, 45]. Meanwhile, displacing of Dithizon from complex Zn-DZ by non-diabetogenic chelators within first 5 min. past forming of complex accompanied by prevention developing of diabetes in 95 % of animals [48] whereas analogical displacing of Dithizon 15–20 min past forming of complex Zn-DZ result developing of diabetes in 95–97 % of animals and diabetes was prevented in 3–5 % animals only.
2.1. Investigation of toxic action of complex «Zn-chelat active metal» on histostructure and ultrastructures of pancreatic islets
It was showed that first changes in cytoplasm of B-cells appeared 5 min past injection of DZ as small zones of destruction of cytoplasm. More detail analysis by aid of transmission electron microscopy showed that process of destruction of B-cells started by destruction of B-granules [49] (Fig. 2.7, 2.8).
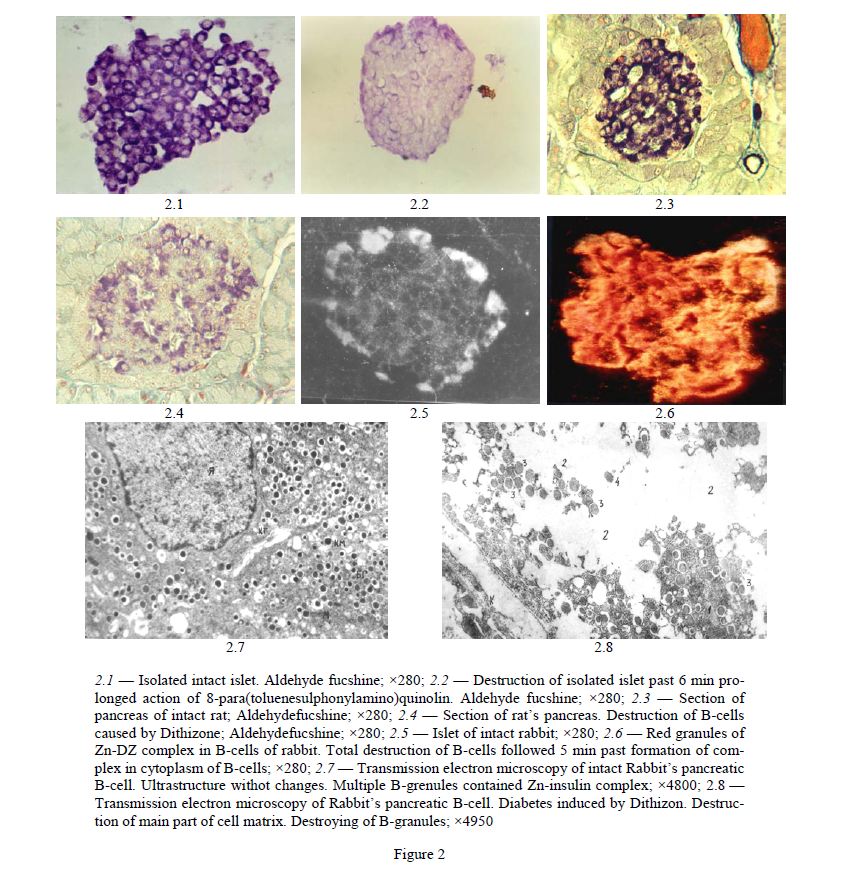
2.1 — Isolated intact islet. Aldehyde fucshine; ×280; 2.2 — Destruction of isolated islet past 6 min prolonged action of 8-para(toluenesulphonylamino)quinolin. Aldehyde fucshine; ×280; 2.3 — Section of pancreas of intact rat; Aldehydefucshine; ×280; 2.4 — Section of rat’s pancreas. Destruction of B-cells caused by Dithizone; Aldehydefucshine; ×280; 2.5 — Islet of intact rabbit; ×280; 2.6 — Red granules of Zn-DZ complex in B-cells of rabbit. Total destruction of B-cells followed 5 min past formation of complex in cytoplasm of B-cells; ×280; 2.7 — Transmission electron microscopy of intact Rabbit’s pancreatic B-cell. Ultrastructure withot changes. Multiple B-grenules contained Zn-insulin complex; ×4800; 2.8 — Transmission electron microscopy of Rabbit’s pancreatic B-cell. Diabetes induced by Dithizon. Destruction of main part of cell matrix. Destroying of B-granules; ×4950
Figure 2
For the first, the 2–4 B-granules are destructed with forming of small zones of destruction of cytoplasm of B-cells [7, 49, 50], not more than 3–5 % of total surface of section of B-cells. 15–20 min later the sizes of these zones rapidly increased until 30–40 % of surface of B-cells and 1–2 h past injection almost all cell’s matrix, 80–90 % of section’s surface, is destroyed completely [7, 49, 50]. We showed that these changes are not visible on light microscopy but very well discovered by transmission electron microscopy. Destructive histological changes developed a few days later — are secondary changes as result of not visible destroying of B-cells within first 15–20 min past forming of chelat complex in cytoplasm of B-cells.
Thus, it was concluded that destruction of B-cells past injection of chelators is determined by destructive action of red complex Zn-DZ on structures, for the first — B-granules, of B-cells within first 15–20 min. past forming of complex in cytoplasm of B-cells (Fig. 2, 3).
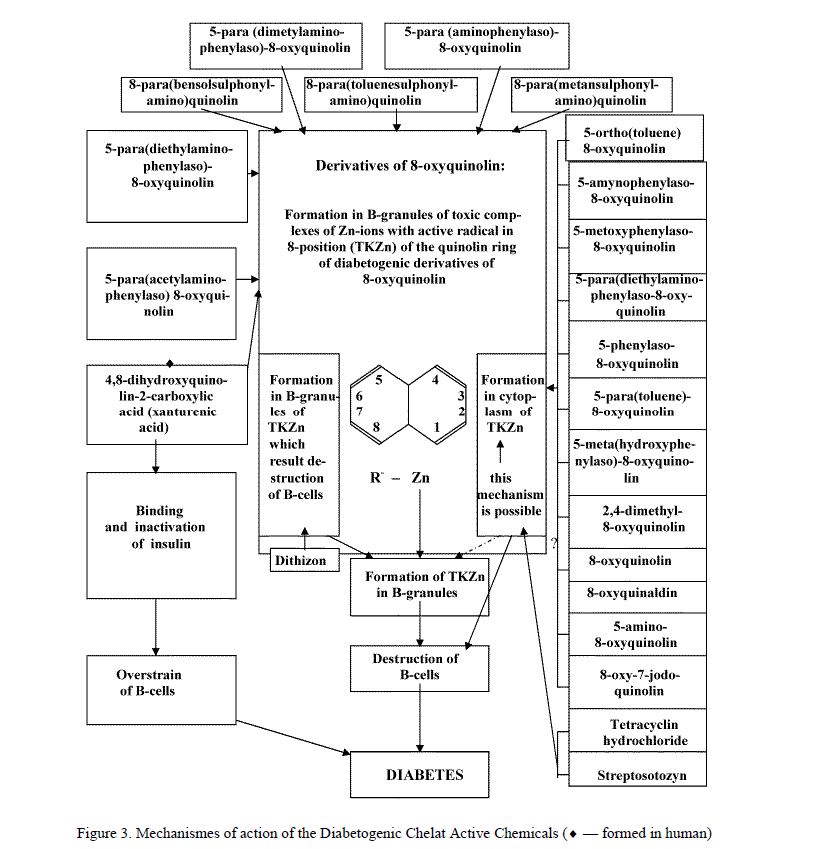
Figure 3. Mechanismes of action of the Diabetogenic Chelat Active Chemicals (¨ — formed in human)
Using of transmission electron microscopy method we showed that 2 h past injection of Dithizon a strongly marked destruction of B-cells was developed: total devastation of cytoplasm of cell’s matrix, destruction of mitochondria, endoplasmatic reticulum and B-granules were discovered in the most parts of cells with remained matrix [49, 50]. Same results were obtained 1 h past injection. Meanwhile, 15 min past injection in the contrary to 2 h cell’s matrix was remained on 60–70 % of B-cell’s surface but 30–40 % appeared as zone free of matrix or zone of complete destruction of ultrastructures of B-cells [49]. Minimal alterations were revealed 5 min past injection: sometimes a few destructed B-granules formed very small zones of destruction of cells. Detail analysis showed that process of destruction of B-cells was begun by destruction of B-granules and forming of zones of destruction of cytoplasm [49]. Investigation of state of ultrastructure of B-cells 5 min past injection have important role for revealing first initial changes in B-cells which are important for understanding mechanismes of diabetogenic action of substance. Then it was confirmed that start destruction of B-cells by destruction of B-granules is determined by forming toxic chelat complexes of diabetogenic chelat chemical with Zn-ions located in B-granules. From the granules process of destruction is spread on other parts of cells. Meanwhile, it is reason to note, that in B-granules is locaited all amount of Zn-ions contained in B-cells.
- Dabetes induced by Xanturenic Acid, a abnormal metabolit of Tryptophan
L.Musajo in 1935 reported about fact of synthesis of Xanturenic Acid (XA). This substance was separated from the urine of experimental animals and confirmed as 4,8-dihydroxyquinolin-2-carboxylic acid [50], C10H7NO4.
S.Lepkovsky and coll. interested by this substance [51]. There are intensification of synthesis of XA in organism when abundance of fat acids was accumulated in organism in combination with vitamin B6 deficiency. These changes were accompanied by developing of symptoms of diabetes [52–57].
XA is product of disturbance of tryptophan metabolism. Usually tryptophan is metabolised by serotonin or kynurenine metabolic ways which completed by forming of 5-oxyindolacetic acid and NADF correspondly. The deficiency of Pyridoxal-5-Phosphate (P-5-P) induced by deficiency of vitamin B6 result inhibition of 5-oxytryptophandecarboxylase and kynureninase which result inhibition of both metabolic ways. As result 4 substances are formed: XA and 8-oxyquinaldine from 3-oxykynurenine and kynurenic acid and oxykynurenic acid from kynurenine (Fig. 4). Meanwhile it was showed that vitamin B6 (Pyridoxin) possess ability to inhibite endogene synthesis of the XA [58–62].
The synthesis of XA determined by main enzymes kynureninaminotransferase (KAT) and oxytryptophandecarboxylase (OTD) with co-enzym as P-5-P [60, 63]. XA is formed from the 3-oxykynurenine by action of KAT. The deficiency of P-5-P result inhibition of synthesis of serotonin and, in the contrary, synthesis of XA and kynurenine are increased [32, 51]. But here we have contradiction: why deficiency of P-5-P accompanied by inhibition of serotonin way and by stimulation synthesis of XA? On the one hand this is determined by fact that pyridoxal enzymes reacted differently for the P-5-P deficiency: activity of kynureninase decreased for 83 % and of KAT — for 42 % only [64]. On the other hand as it was established, KAT are localized as in mitochondries as in soluble part of cells while kynureninase — in soluble part of cells only. Deficiency of P-5-P accompanied by decreasing of content of both enzymes in soluble part of cells and content of KAT-mitochondrial not decreased [65]. This is because elimination of XA with urine is increased. For first time increasing of elimination of XA was discovered in urine of rats contained on diet enriched by tryptophan in combination of vitamin B6 deficiency. XA is disappeared from the urine when vitamin B6 was is added in diet [58, 61, 66]. But aggravation B6 deficiency accompanied by decreasing activity of KAT that result increasing of elimination of XA by urine [32]. More later XA was discovered in the urine of rats, dogs, guinea pigs and human [52, 59, 67–71]. The high concentrations of XA discovered in urine of diabetic patients in middle and old age [72] as more high concentrations of kynurenic acid. Additional administration of Pyridoxine accompanied by decreasing of XA in urine but without complete normalization [72] especially in organism of old persons. XA is eliminated from organism by kidneys. The middle concentrations of XA in the urine of healthy persons in 24 h portion of urine are equalled 2.1–8.9 mg [61].
Deficiency of P-5-P in organism is result of deficiency of vitamin B6 in diet or related with disturbances of synthesis of P-5-P from vitamin B6. Synthesis of XA is intensified by diet enriched by fat acids and casein. It is known 2 enzym systems which determined byosynthesis of P-5-P: pyridoxinphosphateoxydase (PPO) and pyridoxinkynase (PK). Enriched of diet by fat acids result inhibition of activity of PPO in the liver [73] and may be restored by administration of vitamin B2, a co-enzym of PPO. It was showed that in neonatal period — first 3 days — derivatives of kynurenine metabolic ways are not discovered in the urine [74]. In period between 5th and 20th days minimal concentrations of XA are present in urine of babies contained on milk of mother [75, 76]. Addition of α-Tryptophan accompanied by increasing concentration of XA in the urine of babies taked off mother milk as in children in age 4–6 years. In elderly human in age 70 and more synthesis of kynurenine is active. Addition of 100 mg per 1 kg of α-tryptophan in diet accompanied by intensive elimination of XA with urine from organism of old men in age of 70 and more [77–79]. It is possible to normalyse elimination of XA by administration of Pyridoxin [80]. Administration of Pyridoxin accompanied by normalization [21, 80–86] of concentration of xanturenic acid in the urine excluding patients with strong deficiency of vit. B6 [62, 83] due to reduction activity of kynureninaminotransferase. Administration of 100 mg per 1 kg of α-tryptophan in organism of pregnant women accompanied by abnormally high elimination of XA during all time of pregnancy as of kynurenic acid for 3–4 months [82]. It was showed deficiency of vit. B6 in diabetic patients with high concentration of xanturenic acid in the urine [87, 88]. Usually disturbances of tryptophan metabolism accompanied by forming of abundance of XA as of other abnormal products in result of deficiency of P-5-P in organism [47].
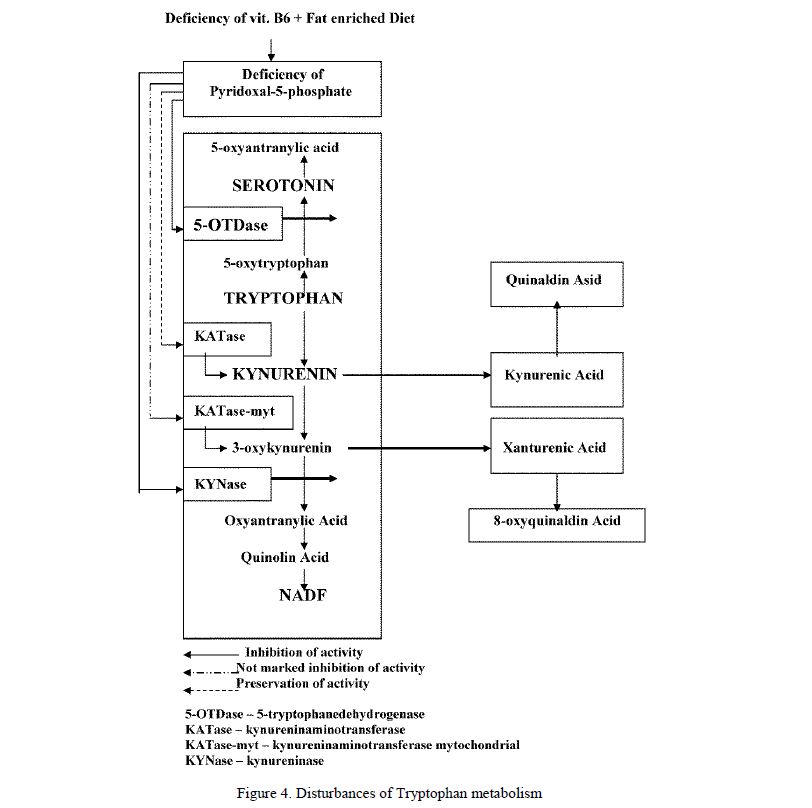
Figure 4. Disturbances of Tryptophan metabolism
Y.Kotake and coll. in 1957 investigated processes of forming and elimination of XA from organism. He used various Na-salts of fat acids and tryptophan which were administrated intraperitoneally in organism of rats. The most large amount of XA was formed and eliminated (elimination — 10,49 mg of XA per 24 h) by urine after using of mixture «tryptophan+oleic acid» and the most less effect — 1,6 mg per 24 h, after administration of tryptophan only. Effects of other mixtures: «tryptophan+acetic acid» — 5,37 mg of XA per 24 h, «tryptophan+propyonic acid» — 8,79 mg of XA per 24 h, «tryptophan+oleic acid» — 9,87 mg of XA per 24 h, «tryptophan+valeryanic acid» — 9,64 mg of XA per 24 h, «tryptophan+palmytin acid» — 9,61 mg of XA per 24 h and «tryptophan+stearynic acid» — 8,57 mg of XA per 24 h. On the base of its experiences Y.Kotake recommended to include in diet a followed products for stimulation of synthesis of XA: casein, salt mixture of McColumn, agar-agar, sugar, saturated oleum, yeast and starch. It was showed that biosynthesis of P-5-P is depended of containing of fat or fat acids in diet. It was concluded: fats and fat acids stimulated decreasing activity of pyridoxinaminotransferase in the liver of rats [73]. It was reported that stress stimulated acceleration accumulation of diabetogenic metabolits of tryptophan [88, 89].
Meanwhile injection of 10,0 mg of vitamin B6 in experimental conditions accompanied by decreasing of elimination of XA till 2,03 mg per 24 h [14] in compared with 8,42 mg per 24 h in control. Y.Kotake in 1968 established that fat acids stimulated inhibition of synthesis of P-5-P from the vitamin B6. As result — increasing of forming of XA. Intraperitoneal injection of 200 mg per 1 kg of endogene synthezed XA to mouse accompanied by developing of diabetes [89] and by temporary hyperglycemia in rabbits [90]. But injection of synthetic XA in dose of 200 mg per 1 kg not accompanied by diabetes after administration to dogs and to rabbits [33]. Combination of injection of synthetic XA and using of diet enriched by large amount of fat accompanied, in the contrary, by hyperglicemia and developing of histological changes in islets typical for diabetes [60, 91–94]. Meanwhile diabetes was not developed in conditions: injection of synthetic XA+diet free of vitamin B6 [95].
Using of diet «10 mg/kg tryptophan+deficiency of vitamin B2» accompanied by hyperglycemia and xanturenuria [53]. Same effect was obtained after using of diet «tryptophan+deficiency of vitamin B6». Using of diet enriched by fats by Y.Kotake accompanied in added by increasing of weight of animals on average from 140 g till 220–260 g and by developing of obesity [94]. The excretion of XA was equalled to 2–3 mg per 24 h [96]. Using of histology it was showed developing of evident changes in B-cells: degranulation of B-cells, vacuolisation of cytoplasm, destruction of cells, developing of hydropic degeneration and changes in the nuclei [29, 53, 54, 97–101]. Not only XA but kynurenic acid induced hyperglicemia. It is transformed in quinaldic acid. XA is metabolized in final product — 8-oxyquinaldic acid which possessed diabetogenic properties.
Theoretically this substance may be transformed in diabetogenic agent 8-oxyquinaldin but we have not found information about forming 8-oxyquinaldin in organism of animals or in human organism as result of disturbances of tryptophan metabolism. It is known that diabetogenic effect of 8-oxyquinaldin is not so intensive as effect of other derivatives of 8-oxyquinolin described above. XA as quinaldic acid and kynurenic acid possess insulin releasing activity [66, 102] and stimulated intensive insulin releasing from isolated pancreatic islets in the first 30 min after start of incubation. Later activity is decreased. The quinaldic acid inhibite completely 2nd phase of insulin releasing [103, 104] and is more active comparatively with 8-oxyquinaldic acid [105]. Incubation of insulin and XA accompanied by forming of stable complex XA-insulin separated on the Sephadex [96, 106]. It was showed by using fluorimetric method that 2 moles of XA are binded with 1 mole of insulin. Activity of this complex is as 49 % of activity of pure insulin.
Not only XA but kynurenic acid induced hyperglicemia too [107]. XA is transformed in quinaldic acid [113] and later is metabolized in final product — 8-oxyquinaldic acid [66] which possessed diabetogenic properties. Activity of native insulin [96, 108] is increased after administration of Zn-ions in incubation medium [106, 109, 110]. It was showed that Zn-ions force out molecule of insulin from the complex XA-insulin and formed new complex XA-Zn. Meanwhile the role of this chelat complex was not investigated.
E.Murakami reported [97, 111–116] that incubation of XA with insulin result forming of 2 sorts of complexes which were separated and purified: complex XA-insulin 1:1 and complex XA-insulin 1:1,5. Activity of both complexes is as 50 % of activity of native insulin only [66, 108]. He supposed that same complex may be formed in human organism. In the blood XA is easy binding with insulin and not transformed chemical structure of it. This complex is stable [96] and chemical connection is formed via atom of Zn and imidazol group of histydin in molecule of insulin [96, 98]. XA possess especial affinity to Zn-ions [114]. Addition of Zn-ions to the serum of blood contained complex XA-insulin stimulated restoration activity of insulin [115]. Not only deficiency of vitamin B6 induced active synthesis of XA in organism as of other abnormal metabolits of tryptophan. The biosynthesis of P-5-P in human organism is inhibited by some drugs for treating of patients with tuberculosis due to its ability to block both enzyme systems of P-5-P [48]. Treatment by Hydraside of Isonicotine acid accompanied by deficiency of P-5-P [116, 117] and as result by xanturenuria. Isoniasid is antagonist of P-5-P [118] and of kynureninase [119]. Treatement by Isoniasid accompanied by xanturenuria and kynurenuria [76]. It was established in added that tuberculosis accompanied by deficiency of vitamin B6 in organism which stimulated aggravation of deficiency of P-5-P.
Other state — pregnancy — accompanied by xanturenuria too. Intensity of xanturenuria is decreased by administration of vitamin B6 [50]. Total amount of XA eliminated with urine from organism of patients with pregnancy aggravated by toxycosis and reached 190 mg per day in compared with 0,3–13 mg of XA per day in control group without pregnancy and toxycosis [50]. It is showed that frequency of diabetes is depended of number of pregnancy during woman’s life: 2,7 % among women with 1 pregnancy, 5,2 % — 2 pregnancies, 7 % — 3 and more pregnancies [120]. Now are not cleared causes of xanturenuria in pregnancy. There are suppositions that pregnancy induced acceleration of desintegration of tryptophan by activation of tryptophanoxygenase [21] but it is evidently that more high frequency of diabetes in patients with tuberculosis and pregnancy have relation with endogene synthesis of large amount of XA in organism.
Thus, noted above data about diabetogenic properties of XA are especially interested due to fact that in the contrary to many other diabetogenic chemicals, including all investigated previously diabetogenic derivatives of 8-oxyquinolin, XA only may be formed in organism of animals and human in result of simple disturbances of diet accompanied by deficiency of vitamin B6.
It is necessary to turn attention on 4 coincidences: 1. Xanturenuria is often discovered in the urine of diabetic patiens. 2. Xanturenuria is often discovered in organism of old men. Meanwhile it is known that diabetes of 2nd type is developed more often among elderly persons. 3. Very often deficiency of vitamin B6 is discovered in the group of elderly and old men. 4. XA as chemical is belong to derivatives of 8-oxyquinolin which have in position 8 active group as all other diabetogenic derivatives of 8-oxyquinolin which diabetogenic properties were investigated previously. 5. Extraction of active group from position 8 of molecule of XA accompanied by complete disappearing of its diabetogenic properties as of some other diabetogenic derivatives of 8-oxyquinolin. Returning back of this group in position 8 accommpanied by restoration of diabetogenic properties of XA. 6. Contrary to all other 18 diabetogenic derivatives of 8-oxyquinolin XA is formed in human organism.
- On the mechanisms of diabetogenic action of tryptophan’s metabolits
More than 40 years ago Y.Kotake was fixed attention on fact that chemical structure of XA is very similar with structures of other diabetogenic derivatives of 8-oxyquinolin. He supposed that its diabetogenic properties determined by the presence of active OH-group in position 8 of quinolin ring [121, 122]. In 1957 Y.Kotake and M.Kato were confirmed fact that XA may to induce diabetes only in case if in position 8 is fixed OH-group. Extraction of this group from molecule accompanied by complete disappearing of diabetogenic properties of XA [29, 122].
G.Weitzel and coll. and S.Ikeda and coll. [33, 114] confirmed that XA formed with Zn-ions complex 1:1 and atom of Zn is fixed between hydroxyl and carboxyl groups of quinolin ring. As it is known this sort of complex of 8-oxyquinolin derivatives (1:1) is most toxic for cells. E.Murakami and Y.Kotake were investigated interaction between insulin and XA. They confirmed that xanturenic acid in vitro formed complex XA-insulin [112].
On the base of obtained data Kotake Y. and Ueda T. were proposed a followed point of view on the understanding of mechanismes of diabetogenic action of XA [108, 112, 115] (Fig. 5, left part).
Meanwhile authors in passing showed that after dissociation of complex XA-insulin, xanturenic acid again formed new complex with Zn-ions as XA-Zn. But it was make not attention to this fact and this chelatcomplex was not investigated. Howerer, more later it was showed in vitro that XA binded Zn-ions in B-cells [123–131] and that presence of this complex in cytoplasm of B-cells within short time result alteration and destruction of cells [132–139].
Deficiency of vitamin B6 stimulated forming not only of XA but additionally kynurenic acid and quinaldic acid. These acids stimulated releasing of insulin from the isolated islets [102–104]. On the other side these metabolits are inhibite forming of B-granules [104] in result of blocking of Zn-ions in cells. 8-oxyquinaldic acid inhibite in added synthesis of proinsulin [122]. More over XA inhibite synthesis of insulin by bindng of insulin with Zn-ions [124].
As result of disturbances of Tryptophan metabolism the 8-oxyquinaldin may be accumulated. Meanwhile 8-oxyquinaldin, a derivative of 8-oxyquinolin, is diabetogenic substance which are able to induce hyperglycemia and degenerative changes in B-cells [33]. However XA is eliminated from the organism with urine and now there are not reported facts that XA is transformed in 8-oxyquinaldin in organism. Nevertheless, we cannot to exclude this possibility.
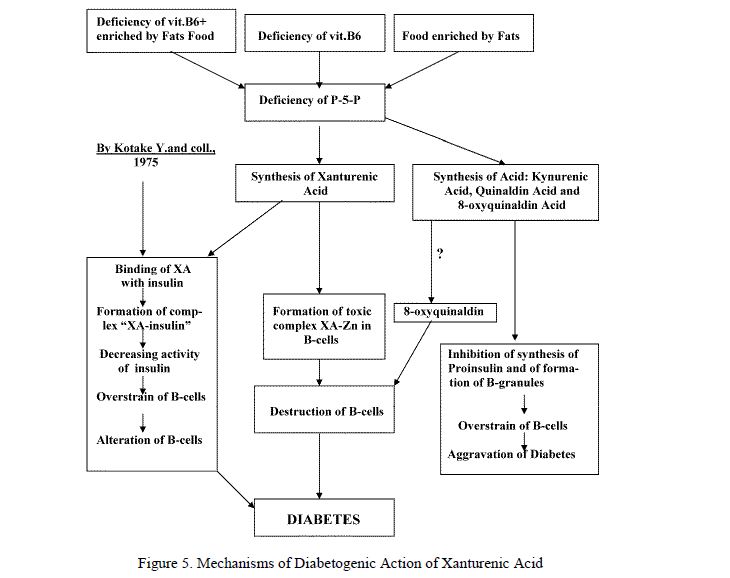
Figure 5. Mechanisms of Diabetogenic Action of Xanturenic Acid
Diabetes induced by derivatives of 8-oxyquinolin may be prevented in experimental conditions by preliminary preventive binding of Zn-ions in B-cells by not diabetogenic chelat active substances as Dietyldithiocarbamate of sodium which are able to protect B-cells in 95–100 % animals for 24 h [48] of destruction. The other, 2nd way: preventive almost complete elimination of Zn-ions from B-cells before administration of diabetogenic chelat active chemicals. And 3rd way: preventive concurent interseption of diabetogenic chelator in nutria media by Zn-ions delivered in solution outside as ZnSO4. But this way is valid for experimental defence of B-cells of isolated pancreatic islets. The advantage of this method: reaction passed not into cytoplasm of B-cells, but outside of cells.
These methods protection of B-cells which we have used in process of investigation of mechanisms of developing of diabetes induced by chelat active chemicals, not have perspective of practical using because it is not possible and not expediently to keep Zn-ions in B-cells permanently connected with not diabetogenic substances or to eliminate permanently Zn-ions from the cytoplasm of B-cells and to keep cells free of Zn-ions permanently.
Thus, despite of fact that by aid of both methods it is possible to prevent developing of experimental diabetes in 95–100 %, these methods are not suitable for prevention developing of XA-diabetes on aspect of human diabetes.
However it is known that synthesis of XA in organism may be prevented by administration of vitamin B6. This way of preventinion of xanturenic diabetes is, as we think, more perspective. Besides this method not need additional investigations and scientifical reasons concerning elaboration related with practical using of vitamin B6.
Injection of other diabetogenic derivatives of 8-oxyquinolin result 2–3 days later developing of heavy diabetes 1 type. Diabetes induced by XA in the contrary look like diabetes of 2 type. It is explayned probably by a followed circumstances. Other diabetogenic derivatives of 8-oxyquinolin were used as one injection of 100 % diabetogenic doses of substance. More less amount of XA is formed in organism more slowly day by day permanently in result of changes of diet or changes of tryptophan metabolism especially in old organism. Interest to diabetes induced by XA is increased due to followed factors: 1. XA in the contrary to other diabetogenic derivatives of 8-oxyquinolin is formed in human organism as result of simple changes of diet.
A large amount of XA is discovered in the urine not only of diabetic patients in middle or old age, but in the urine of persons in same age without diagnosis of diabetes. 3. Deficiency of vitamin B6 is discovered in organism of old persons with registrated diagnosis of diabetes or without it.
Previous our investigations of mechanismes of diabetogenic action of derivatives of 8-oxyquinolin, which cannot be synthezed in organism or to come into organism outside, have theoretical significance only. However data obtained during these experiences let us to understand more profoundly mechanisms of diabetogenic action of XA. XA due to noted above data make us to call our attention on this substance which may to have some significance in pathogenesis of human diabetes.
On the base of data obtained by other investigators and by us I propose a followed point of view on the mechanisms of diabetes induced by XA (Fig. 5).
Thus, noted above data showed a potential role of diabetogenic metabolits of tryptophan in pathogenesis of human diabetes. From the presented data it is possible to conclude that main role among a few metabolits — XA, kynurenic acid, oxyquinaldic acid, 8-oxyquinaldic acid and 8-oxyquinaldin — are belong to XA. Kynurenic acid and oxyquinaldic acid not contained, in the contrary to XA, in position 8 of quinolin ring of active chemical group and not induced diabetes. Both these chemicals activate insulin releasing from B-cells.
Thus, now it is possible to suppose that main role is belong to XA as to diabetogenic chemical. Other metabolits are able to aggravate diabetes induced by XA.
- On the possible ways for prevention developing of diabetes caused by chelators
What is minimal period of the presence of chelat complexes in B-cells for to induce diabetes? As it was showed previously this period is 15–20 min. For to confirm this fact the experiences with non diabetogenic chelator-Na salt of Diethyldithiocarbamic acid (NaDDC) were conducted. NaDDC have more high affinity to Zn-ions and able to displace Dithizon from chelat complex Zn-DZ as of derivatives of 8-oxyquinolin from its chelats with Zn-ions. Analogical properties possess Dimethyldithiocarbamic acid and its derivatives. EDTA as chelator possess more high affinity to Zn and constant of stability of its chelats with Zn is 13.1 meanwhile with ions of Mg+2, Ca+2 and Fe+3 correspondly 5.4, 7.3, and 10.9 [15]. It was showed that EDTA prevent diabetogenic action of streptosotozin by binding of Zn ions [131]. More detail investigation of processes of interaction of Zn-ions contained in B-cells with NaDDC showed that injection of 250 mg/kg of NaDDC result binding of part amount of Zn-ions inlocated in cytoplasm of B-cells, and of 500 and 1000 mg/kg — a complete binding of all amount of Zn-ions. Dissociation of complex is passed more slowly: 10–12 h past injection dissociated less part of complex and 24–48 h past injection — all amount of Zn-ions are free [34, 35, 49]. Injection of NaDDC 5 min — 6 h before diabetogenic chelator result complete prevention of diabetes in 100 % of animals for 8–10 h. Injection of DZ within this period not accompanied by formation of red stained granules of Zn-DZ complex in cytoplasm of B-cells and diabetes is not developed [41, 48]. Injection of NaDDC 15 min past injection of DZ accompanied by complete displacing of DZ from complex Zn-DZ and by formation of not diabetogenic complex Zn-NaDDC but diabetes developed in 95– 96 % of animals and was prevented in 5 % of animals only. Injection of NaDDC 5 min past DZ accompanied also by complete displacing of DZ from complex Zn-DZ but diabetes was prevented completely in 95 % of animals and was developed in 5 % of animals only (49). Injection of NaDDC 2 h past injection of DZ accompanied by developing of diabetes in 100 % of animals [48]. Thus, by these experiences it was confirmed that the presence of toxic chelat complexes of DZ and diabetogenic derivatives of 8-oxyquinolin in B-cells within first 15 min after its forming result not visible for the first a few hours incorrigible destructive changes in B-cells. Formed more later degenerative histological changes in islets is result of action of chelators in the first 15 min.
The aminoacids Cystein and Glutathion formed not toxic chelats with atoms of heavy metals due to sulfhydril radicals which have high affinity to ions of Zn+2, Pb+2, Cd+2 and Hg+2. It is suggested that by these radicals aminoacids formed not toxic chelats with Zn-ions. The constant of stability of complex Zn-Cystein is very high — 17.1–18.2 [48].
Aminoacid Hystidin formed with Zn-ions high stable complex 2:1 which logarifm is 12. Contrary to other aminoacids chelat activity of Hystidin is determined by the presence in molecule of the imidazol ring [21].
Injection of Cystein 1000 mg/kg prevent formation in B-cells of toxic chelat Zn-DZ an completely protect of diabetes all animals within 6 h; 12 h past injection diabetes was prevented in 6 animals from 8 and 24 h past injection of Cystein — in 2 animals from 4. Cystein protect B-cells of destruction caused by diabetogenic derivatives of 8-oxyquinolin. Aminoacid Serin, which contained hydroxyl radical in molecule instead of sulfhydril radical in molecule of Cystein, not possess diabetogenic properties.
Diabetes is prevented by Restored Gluthation. Preventive injection of it protect B-cells of destruction and of developing of diabetes in all animals: normoglycemia and B-cells — without changes. Oxydation of Restored Gluthation result: two molecules of Restored Gluthation formed one molecule with forming of disulfide connection. Injection to animals of 1000 mg/kg of the Gluthation — oxyd not accompanied by prevention of diabetes in all experimental animals.
Thus, inactivation or change of Sulfhydril radicals in molecules of Cystein and Gluthation result complete disappearing of diabetogenic properties of these both substances [28]. Injection to animals of 1000 mg/kg of the Hystidin Hydrochloride (HH) result complete prevention of diabetes past injection of Dithizon followed 5 min past injection of HH and — in half of total number of animals injected of Dithizon 0,5–1 h past injection of HH [28].
Prevention of binding of Zn-ions in pancreatic islets by diabetogenic chelators may be realized by other way — preliminary complete elimination off B-cells zinc-insulin complex by derivatives of Sulphonurea [31]. Injection of Dithizon or derivatives of 8-oxyquinolin past maximal complete elimination of Znions from B-cells not result forming of toxic copmlexes «Zn-chelator» in cytoplasm of B-cells and diabetes not developed in 100 % of animals. This method of prevention of diabetes is effective and suitable in cases when administration of diabetogenic dose of chelator is expected. In opposite, this method it is not suitable when low doses of diabetogenic chelators are formed in organism during long period.
Dehydroascorbic Acid (DA) which is formed in organism as result of metabolisation of Ascorbic Acid, possess diabetogenic properties and result of direct alterative effect on B-cells [120]. Concentration of DA in organism of diabetic patients is evidently increased in opposite to decreasing concentration of Ascorbic Acid [123].
It is known that Streptozotocin possess chelat properties and have high affinity to Zn-ions. Alterative action of Streptozotocyn may be prevented or reduced by preventive action of EDTA [137].
Investigation of diabetogenic properties of Dithizon and derivatives of 8-oxyquinolin have theoretical significance because these chemicals are not formed in human and really not delivered in human organism outside. In added, peroral administration of its is not effective because they are not soluble and not absorbed in intestinum. Parenteral injection of diabetogenic chelators result developing of diabetes only. Meanwhile solutions of all these chelators are not stable and only injection of the fresh prepared solutions (ex tempore) result diabetogenic effect.
Among 18 diabetogenic derivatives of 8-oxyquinolin the Xanturenic Acid (XA) only is formed in elderly humans. It is known that XA is accumulated in organism of old human as result of disturbances of Tryptophan metabolism. Low doses of the XA accumulated in human gradually. May be that is why diabetes caused by XA developed gradually as type 2 contrary to type 1 diabetes caused by injection of diabetogenic doses of other chelators. Mechanisms of diabetogenic activity of chelat active chemicals and way of prevention development of diabetes caused by them were studied by us before [140–194].
The most perspective way for prevention of diabetes caused by XA is, of course, prevention of endogene synthesis of XA in human in disturbances of Tryptophan metabolism.
Last decades the number of diabetogenic chelators human have contacts is very increased. As example, Tetracycline hydrochloride is active chelator which have high affinity to Zn-ions and formed with it complex 1:1 and 2:1 with high constant of stability as 9,0 [21]. Direct action on B-cells of high doses of tetracycline result hyperplasia and degeneration of cells. Isoniazid, a drug for treatment of tuberculosis, formed pentagonal stable chelats with Zn-ions. May be more high frequency of diabetes among patients treated by Isoniazid determined by this fact? This interest is increased taking into consideration fact that in this case concentration of the Xanturenic acid in urine is high because Isoniazid in antagonist of Pyridoxal-5-Phosphate [63].
Diuretics as derivatives of Benzothiodiazine possess chelating properties and treatment during long time accompanied by developing of diabetes sometimes. Treatment by Chlorthiazid accompanied by hyperglycemia and glucosuria.
It is known that chelators which formed with Zn-ions tetragonal or pentagonal rings possess diabetogenic properties. Chelators contained in molecule as least 4 or 5 double chemical connections possess diabetogenic properties also in opposite to chelators contained 1–2 or not contained its which not possess analogical properties. As example — derivatives of Diethyldithiocarbamic Acid of Dimethyldithiocarbamic acid, aminoacids Cystein, Gluthation and Hystidin. Complexes formed by noted above protectors not contained in molecule tetragonal or pentagonal rings and not contained or contained minimal number (1–2) of double connections. Administration of large amount of these chelators not result destruction of B-cells and protect, in opposite, B-cells of destruction caused by diabetogenic chelators.
Noted above data put us to look on these chemicals as on one possible factor in ethiology of human diabetes. The significance of this possibility is increased taking consideration fact that human pancreas contained a large amount of Zn-ions possess to form chelat complexes with diabetogenic chelators.
On the base of presented above our experimental data as data obtained by other investigators we propose a followed point of view on the role of Zn+2-ions in the pathogenesis of diabetes mellitus caused by diabetogenic derivatives of 8-oxyquinolin as followed main steps:
- Diabetes induced by chelators is determined by formation in cytoplasm of B-cells of the chelat complexes «Zn-chelator» only and developed as Any mechanisms prevention synthesis of this chelatscomplexes in cytoplasm of B-cells protect completely cells of destruction and of developing of diabetes.
- Destruction of B-cells caused by direct action of chelat complexes on histoctructures of B-cells — is main mechanism of developing of these models of Inevitable destructive changes in cytoplasm of B-cells developed within first 15–20 min past forming of chelats in cells. Changes developed within first 4–5 min are not significant and displacing of chelator from complex «Zn-chelator» by not diabetogenic chelators that result prevention of destruction of B-cells and developing of diabetes. Incorrigible histological changes in B-cells developed a few days later, are as result of noted above changes developed within first 15–20 min. past formation of toxic chelat complexes in B-cells.
- Diabetes induced by chelators may be prevented by prevention formation of complexes «Zn-chelator» by 4 ways: a) preliminary complete elimination of Zn-ions off cytoplasm of B-cells; b) by preliminary binding of Zn-ions in B-cells by not diabetogenic chelators have more high affinity to Zn-ions comparatively with diabetogenic chelators; 3) by concurent interseption of chelator in liquid media by Zn-ions delivered outside with Zn-contained salts; 4) by prevention of the synthesis in organism (including human) of diabetogenic chelators.
- Inhibition of endogene synthesis in cytoplasm of B-cells of diabetogenic metabolits of tryptopan using of Pyridoxine is most preferable and perspective way for prevention developing of diabetes in animals and
Next years be investigated possibilities for prevention developing of diabetes caused by 4,8-dihydroxyquinolin-2-carboxylic acid (XA) by partial or almost complete inhibition of endogenous synthesis in animals and human of the XA, a product of abnormal tryptophan metaboilsm.
Acknowlegement
This work proceeding 41 years was supported since 1977 by reagents, research and travel grants from Prof. K.-D.Kohnert, Institute of Diabetes «Gerhardt Katsch» (Karlsburg, Germany); by H.-A.Langisch, J.Watzek «Boehringer Mannheim» (Germany), H.Niedderer, E.Horn «SERVA» (Germany), H.Ritzel «HOECHST» (Germany), G.Putlik «SARTORIUS» (Germany), W.Manneck «MERCK» (Germany), «BACHEM» (Germany), O.E.Grundemann «FERAK» (Germany), B.Alimbekova «SERVIER» (France), Ch.Studer «CALBIOCHEM» (Switzerland), R.Thunberg «Pharmacia Fine Chemicals» (Sweden), «Yanssen Chimica» (Belgium), P.Felch, R.Schilly «Boehringer Mannheim Wien» (Austria), V.Riedl (Austria) by research and travel grants from vice-president of International Diabetes Federation, Prof. John R.Turtle (Sydney, Australia), by vice-president of International Diabetes Federation Dr. Lorna Mellor (Sydney, Australia), by President of the American Diabetes Association, Prof. H.Rifkin (USA), Prof. B.Tuch (Sydney, Australia), Prof. Y.A.Lasaris and Prof. I.A.Serebrovska, Karaganda by Prof. F.Wohlrab (Germany) and Prof. E.Murakami (Japan). Scientific project and main financial supporting of work — by families of Prof. G.A.Meyramov and Prof. G.G.Meyramov since 1972.
References
- Scott D.A., Fischer A.M. Pharm. Exp. Therap., 1935, 55, p. 206–221.
- Scott D.A., Fischer A.M. Clin. Invest., 1938, 17, p. 725–728.
- Eisebrandt J., Sienz M., Wegel F. Medizin und Chemie, 1942, 8, p. 259–296.
- Emdin S., Dodson G., Cutfield J., Cutfield S. Diabetologia, 1980, 19, p. 174–182.
- Voinar A. The Biological Role of Microelements in the Human and Animals, Moscow, 1960, p. 311–365.
- Galabova R., Petkov P., Kolev J. Acta Histochem., 1971, 2, p. 335–342.
- Okamoto K. Diabetes Mellitus: Theory and Practice, New York, 1970, p. 236–255. 8 Okamoto K. Liver, 1942, 32, p. 99–105.
- Lapin V.I., Meyramov G.G., Korchin V.I., Satosin V.A. Physiol & Exper. Therapy, Moscow, 1973, 4, p. 36–39.
- Schmidt R., Rautschke R. Acta Histochem., 1964, 17, p. 302–313.
- Lapin V.I. Content of Zn-ions in pancreas tissue of the normal and diabetic animals, Karaganda,
- Voigt G.E. Virchov Arch. Path. Anat., 1959, 4, p. 295–323.
- Andersson T., Betgren P., Flatt P. Metab. Res., 1980, 12, p. 275–276.
- Lasaris Y.A., Bavelsky Z.E. of Exper. Biol & Med., Moscow, 1970, 2, p. 44–48.
- Maske H., Weingas K. Exp. Pathol. Pharmacol., 1957, 230, p. 406–420.
- Voigt G. Acta Path. Microbiol. Scand., 1957, 41, p. 81–88.
- Miller W., Kincaid R., Neathery M. et al. Trace Elem. Metab. Man Anim., Friesing, 1978, p. 175–178.
- Lowry P., Baldwin R., Harrington S. Science, 1954, 119, p. 219–220.
- Kawanishi H. Jap., 1966, 13, 4, Р. 384–408.
- Okamoto K., Kawanishi H. Jap., 13, 3, p. 305–318.
- Albert A. Selective Toxicity, London, 1968, p.
- Okamoto , Fujiwara T., Sukenary K., Fukutomi H. Trans. Soc. Pathol. Jap., 1951, 40, p. 150–153. 23 Kadota I. J. Lab. Clin. Med., 1950, 358, p. 568–591.
- Kadota I., Abe T. J. Lab. Clin. Med., 1954, 43, p. 375–385.
- Fujiwara T. Kobe Med. Coll., 1954, 4, p. 1091–1131.
- Uemura T. Kobe Med. Coll., 1956, 7, S. 1, p. 1–25. 27 Ichioka T. Kobe J. Med. Sci., 1958, 4, 2, p. 63–80.
- Lasaris Y.A., Lasaris A.Y. Problems of Endocrinology, Moscow, 1967, 3, p. 75–81.
- Kotake Y., Kato T. Jap. Acad., 1956, 32, p. 361–363.
- Zentmyer G. Science, 1944, 100, p.
- Bavelsky Z.E., Meyramov G.G. Biology of animals, Karaganda, 1976, 2, p. 119–123.
- Rubbo S., Albert A. J. Exp. Pathol., 1950, 31, p. 425–428.
- Weitzel G., Budecke E. et al. Hoppe-Seyler’s Z. Physiol., 1954, 298, p. 169–184.
- Lasaris Y.A., Meyramov G.G. Exper. Biol. & Med., Moscow, 1974, 3, p. 19–22.
- Lasaris A., Meyramov G.G. Problems of Endocrinology, Moscow, 1974, 5. p. 90–94. 36 Kadota I., Abe T. J. Lab. Clin. Med., 1954, 43, p. 375–385.
- Krasavin I.A., Bavelsky Z.E., Lasaris Y.A., Dsiomko V.M. Problems of Endocrinology, Moscow, 1969, 3. p. 102–105.
- Lasaris Y.A., Dsiomlo V.M., Krasavin I.A. Problems of Endocrinology, Moscow, 1968, 4, p. 107–111.
- Okamoto K. Acta Sch. Med. Univ. Kyoto, 1949, 27, 1, p. 43–65. 40 Maske H. Experientia, 1955, 11, 3, p. 122–128.
- Wolff H., Maske H., Stampfl B., Baumgarten F. Wschr., 1951, 39–40, p. 670–671.
- Wolff H., Ringleb D. Naturewissenshcften, 1954, 41, 11, p. 260–261.
- Wolff H., Ringleb D. Ges. Exp. Med., 1994, 124, p. 236–256.
- Wolff H., Ringleb D., Amman R. Ges. Exp. Med., 1955, 126, 4, p. 390–416.
- Albert A., Rubbo S. J. Exp. Pathol., 1947, 28, p. 69–70.
- Stampfl B. Dtsch. Ges. Path., 1959, 42, p. 137.
- Mager M., McNary W., Lionetti F. Histochem. Cytochem., 1953, 1, p. 493.
- Weitzel G., Buddеcke E., Kraft D. Chem., 1956, 17, p. 566–572.
- Meyramov G.G., Truchanov N.I. Problems of Endocrinology, Moscow, 1975, 6, p. 92–95.
- Bavelsky Z.E., Zumerov E.L. Problems of Endocrinology, Moscow, 1984, 1, p. 65–69.
- Lepkovsky S., Roboz E. Biol. Chem., 1943, 149, p. 195–201.
- Kandori F., Fujinaga Y. Yonaga Acta Med., 1959, 3, p. 53 Kotake Y. Clin. Chem., 1957, 3, p. 442–456.
- Kotake Y., Inada T. J. Biochem., 1954, 4, p. 255–261. 55 Kotake Y., Tani S. J. Biochem., 1953, 3, 40, p. 295–298. 56 Kuno S. Vitamins., 1960, 8, 1, p. 140–154.
- Takaoka Y., Yamagushi N. J. Sashinigaku, 1967, 11, p. 19–25. 58 Kotake Y., Inada T. J. Biochem., 1953, 40, 3, p. 287–289.
- 59 Davis R., Galder J. et al. Pathology, 1976, 2, p. 151–156. 60 Mason V. Diabetes, 1956, 6, p. 486–489.
- Price J., Brown R. et al. Clin. Invest., 1957, 36, p. 1600–1607.
- Weber F., Wiss O. Hoppe-Seyler’s Z. Physiol. Chem., 1963, 331, p. 124–131. 63 Prince S., West A. Pharmacol., 1960, 12, 10, p. 617–623.
- Garattini G., Valzetti L. NY State S. Med., 1965, 11, p. 75–81. 65 Kotake Y., Ueda T. J. Biochem., 1975, 7, 3, p. 685–687.
- Takanashi H., Price J. Biol. Chem., 1958, 233, p. 150–153.
- Glaser H., Mueller T. et al. Biochem., 1951, 33, p. 243–247.
- Zartman E., Barnes A. et al. J. Gynecol., 1955, 70, 3, p. 645–649.
- Montenero P. Stud. Physiopathol. Clin. Ricamb., 1960, 24, 3–4, p. 285–289.
- Hattori M., Kotake Y., Kotake Y. Acta Vitaminol. Enzymol., 1984, 6, 3, p. 221–228.
- Oka M., Lepfaren V. Med. Scand., 1963, 173, p. 361–364.
- Crepaldi G., Allegri G. et al. Acta Vit. Enzymol., 1975, 29, p. 140–144.
- Nakahara I., Watanabe Y. et al. Biochem., 1961, 49, p. 342–347.
- Djioev F. 8-th Conference on Oncology, Leningrad, 1976, p. 41–42.
- Knapp D., Veltishev Y., Barashnev I. Voprosi ochrani materinstva I detstva, Moscow, 1978, 23, 10, p. 51–56.
- Knapp A., Wolfram J., Heilmann H. Gesundheitswesen, 1967, 22, 12, p. 2449–2455.
- Vandelli F. J. Clin. Nutr., 1951, 6, p. 684–693.
- Wachstein G., Gudaitis F. Endocr. Metab., 1953, 66, 12, p. 1207–1213.
- Gorbacheva L. Akusherstvo i Gynecologiya, Moscow, 1989, 11, p. 16–20.
- Ranke A., Spellaey W. J. Gynecol., 1977, 6, p. 599–602.
- Rose D., Toseland P. Metabolism, 1973, 2, p. 165–171.
- Coppini O., Comurri A. J. Clin. Med., 1954, 6, p. 673–683.
- Wiss G, Weber F. аnd Horm., 1964, 22, p. 495–501.
- Avanesova T., Sviridova E. Neuropathology and Psyhiatry, 1980, 6, p. 857–863.
- Rose D., Braidman J. J. Clin. Nutr., 1971, 6, p. 673–683.
- Gillmer M., Mazibuko D. Amer J. Obstet Gynecol., 1979, 133, 5, p. 499–502.
- Rudzit V. Diabetogenic metabolits of tryptophan, Riga, 1981, 146
- Sharafetdinov H. Problems of Endocrinology, Moscow, 1998, 1, p. 13–15.
- Alferova V., Raskin I. Voprodsy med. khimii, 1962, 1, p. 20–22.
- Kotake Y., Kido R. Jap. Acad., 1960, 7, p. 439–444.
- Kotake Y., Kato T. Jap. Acad., 1956, 32, p. 364–365.
- Markees S. Physiol. Pharm. Acta., 1954, 4, p. 80–83.
- Vanaga M. Wakayama Med. Soc., 1957, 4, p. 635–642.
- Kotake Y. Osaka Med. School, 1953, 14, p. 51–60.
- Gandin-Harding F., Blum J. Sci. Physiol., 1964, 1, p. 49–58.
- Kotake Y., Sotokawa Y. et al. of Biochem., 1968, 6, p. 895–896. 97 Murakami E. J. of Biochem., 1968, 63, p. 573–577.
- Takanashi H., Kaihara M. et al. J. Biol. Chem., 1956, 223, p. 705–708. 99 Kotake Y., Inada T. J. Biochem., 1953, 40, 3, p. 291–294.
- Kotake Y., Mori T. Jap. Acad., 1955, 31, p. 247–251.
- Meyramov G., Kohnert K.-D. Bulletin Exper. Biol & Med., Moscow, 1997, 6, p. 669–672.
- Okamoto H. Acta Vitaminol. Enzymol., 1975, 1–6, p. 227–231.
- Okamoto H., Mijamoto S. et al. Biophys. Res. Com., 1973, 53, 8, p. 1297–1303.
- Okamoto H., Mijamoto S. et al. Biophys. Res. Com., 1974, 59, 4, p. 623–628.
- Okamoto H. 2nd Intern. Meet. Trypt. Metab., Madison, 1977, p.
- Kotake Y., Ueda T. et al. Acta Vitaminol. Enzymol., 1975, 29, p.
- Mirsky I., Perisutti et al. Endocrinology, 1957, 60, 10, p. 318–324.
- Kotake Y., Murakami E. J. Clin. Nutr., 1971, 24, 7, p. 826–829.
- Kotake Y. et al. of Biochem., 1975, 3, p. 685–687.
- Ueda T., Goda K. et al. Biochem., 1977, 82, p. 67–72.
- Murakami E. Biochem. Soc., 1964, 36, p. 829–834.
- Murakami E. Acta Vitaminol. Enzymol., 1975, 29, p. 210–242.
- Kotake Y., Sotokawa Y. et al. of Biochem., 1968, 5, p. 578–581.
- Ikeda S., Kotake Y. Acta Vitaminol. Enzymol., 1984, 6, 1, p. 23–28.
- Kotake Y., Ueda T. et al. 2nd Intern. Meet. Trypt. Metab., Madison, 1977, p.
- Kotake Y., Murakami E. J. Clin. Nutr., 1971, 24, 7, p. 826–829.
- Rose D., Toseland P. Metabolism, 1973, 22, 1, p. 165–171.
- Price I., Brown R., Larson F. Clin. Invest., 1957, 36, p. 1600–1607.
- Koreсny C., Lowenstein B. Nerv. Syst., 1968, 29, p. 367–372.
- Patterson J. Endocrinology, 1949, 45, p.
- Kotake Y., Nogami K. J. Biochem., 1954, 2, 41, p. 621–624.
- Kotake Y., Kato M. Proc. Jap. Acad., 1956, 32, p. 361–363. 123 Kabrt J. Cas. Lek. Ces., 1982, 27, p. 833–836.
- Okamoto K. Tohoku J. Exp. Med., 1955, 61, 3, p. 27–33.
- Meyramov G.G., Tusupbekova G.T., Meyramova R.G. Problems of Endocrinology, Moscow, 1987, 6, p. 49–51.
- Bozhevolnov E., Serebrakova G. Chemical reagents and preparats, Moscow, 1961, 2, p. 36–42.
- Bozhevolnov E. Fluorescent analysis of anorganic chemicals, Moscow, 1966, 436 p.
- Kim B.-J., Kim S., Koh J.-Y. and coll. Diabetes, 2000, 6, 3, p. 367–372.
- Meyramov G.G. Transplantation Proceedings. «ELSEVIER», New York, 1998, 30, 6, p. 2682–2684.
- Meyramov G.G., Tusupbekova G.T., Niedderer H. Diabetes Research and Clinical. Practice, the Journal of International Diabetes Federation, Amsterdam–New York, 1988, 5, 1, p. 228.
- Meyramov G.G. European J. of Clin. Pharm., 1989, 36, p.
- Meyramov G.G., Tusupbekova G.T., Meyramova R.G. Endokrinol. und Stoffwerschel, Berlin, 1991, 2, 12, p. 114.
- Meyramov G.G. Canadian J. of Physiol. and Pharmacol., 1994, 72, 1, p.
- Meyramov G.G., Kohnert K.-D. 5th Congress of Pancreas and Islets Transplantation, Miami, USA., p.
- Meyramov G. and coll. Diabetologia, Journal of the European Association for the Study of Diabetes, 1997, 40, 1, p. 666.
- Meyramov G., Kohnert K.-D., Meyramova A.G. New Aspects of Pathogenesis and Treatment of Diabetes mellitus, Stockholm, 1999, p. 30.
- Meyramov G.G. and coll. Problems of Endocrinology, Moscow, Russia, 1990, 36, 1, p. 66–69.
- Meyramov G., Kikimbaeva A.A., Meyramova A.G. Diabetes Research and Clinical Practice, the Journal of International Diabetes Federation, 2000, 50, 9, 1, p. 154–155.
- Meyramova A.G., Kikimbaeva A.A., Meyramov G.G. Acta Diabetologica, the International Diabetes Journal, 2000, 37, 3, 160.
- Meyramov G.G., Kikimbaeva A.A., Meyramova A.G. Acta Diabetologica, the International Diabetes Journal, 2001, 38, 4, 208.
- Meyramov G.G., Meyramova A.G., Kikimbaeva A.A. Acta Diabetologica, the International Diabetes Journal, 2001, 38, 4, 208–209.
- Meyramova A.G., Kikimbaeva A.A., Meyramov G.G. Acta Diabetologica, the International Diabetes Journal, 2003, 40, 1, 37.
- Meyramova A.G., Kikimbaeva A.A. Acta Diabetologica, the International Diabetes Journal, 2003, 40, 1, p.
- Meyramova G., Kikimbaeva A.A., Meyramov G.G. Acta Diabetologica, the International Diabetes Journal, 2003, 4, p. 208.
- Meyramova A.G., Meyramov G.G. Acta Diabetologica, the International Diabetes Journal, 2005, 4, p.
- Meyramova G., Kikimbaeva A.A., Meyramov G.G. Diabetes & Metabolism, the Journal of Diabetes Association of France, 2003, 29, 4, p. 83–84.
- Meyramov G.G., Kohnert K.-D., Meyramova A.G. Problems of Endocrinology, Moscow, Russia, 2001, 47, 1, p. 39–44.
- Meyramova A.G. Problems of Endocrinology, Moscow, Russia, 2003, 49, 2, p. 8–16.
- Meyramov G.G., Kikimbaeva A.A., Meyramova A.G. Organ Biology, Japan, 2009, 16(1), p. 116–117.
- Meyramov G.G., Tusupbekova G.T. Diabetes, the Journal of American Diabetes Association, 1991, 40, 1, p.
- Meyramov G.G., Meyramova R.G. Diabetes, the Journal of American Diabetes Association, 1991, 40, 1, p.
- Meyramov G.G., Kohnert K.-D., coll. Diabetes, the Journal of American Diabetes Association, 2000, 49, 5, p.
- Meyramova A.G., Meyramov G.G. Diabetes, the Journal of American Diabetes Association, 2002, 51, p. 591–592.
- Meyramov G.G., Meyramova A.G. Diabetes, the Journal of American Diabetes Association, 2002, 51, 6, p.
- Meyramova A.G., Meyramov G.G. Diabetes, the Journal of the American Diabetes Association, 2003, 52, 6, p.
- Meyramov G., Meyramova A.G. Diabetes, the Journal of the American Diabetes Association, 2003, 52, 6, 1, p. 536.
- Meyramova A.G., Meyramov G.G. Diabetes, the Journal of the American Diabetes Association, 2004, 53, 6, p.
- Meyramova A.G., Meyramov G.G. Diabetes, the Journal of the American Diabetes Association, 2004, 53, 6, p.
- Meyramova A.G., Meyramov G.G. Diabetes, the Journal of the American Diabetes Association, 2005, 54, 6, p.
- Meyramova A.G., Meyramov G.G. Diabetes, the Journal of the American Diabetes Association, 2006, 55, 6, p.
- Meyramov G.G., Meyramova A.G. Diabetes, the Journal of American Diabetes Association, 2008, 57, 6, p.
- Meyramov G.G., Meyramova A.G. Diabetes, the Journal of American Diabetes Association, 2008, 57, 6, p. 727–728.
- Meyramov G.G., Meyramova A.G. Diabetes, the Journal of American Diabetes Association, 2009, 58, p.
- Meyramov G.G., Meyramova A.G. and coll. Diabetes, the Journal of American Diabetes Association, 2009, 58, p.
- Meyramov G., Kikimbaeva A.A. and coll. Diabetes, the Journal of American Diabetes Association, 2010, 59, 6, p. 556.
- Meyramov G., Kikimbaeva A.A. and coll. Diabetes, the Journal of American Diabetes Association, 2010, 59, 6, p. 574.
- Meyramov G., Kikimbaeva A.A. and coll. Diabetes, the Journal of American Diabetes Association, 2011, 60, 6, p. 727.
- Meyramov G.G., Kikimbaeva A.A. and Diabetes, the Journal of American Diabetes Association, 2011, 60, 6, p. 728–729.
- Meyramov G., Kikimbaeva A.A. and coll. Diabetes, the Journal of American Diabetes Association, 2012, 61, 6, p. 719.
- Meyramov G., Kikimbaeva A.A. and coll. Diabetes, the Journal of American Diabetes Association, 2012, 61, 6, p. 720.
- Meyramov G., Kikimbaeva A.A. and coll. Diabetes, the Journal of American Diabetes Association, 2013, 62, 6, p. 726.
- Meyramov G., Kikimbaeva A.A. and coll. Diabetes, the Journal of American Diabetes Association, 2013, 62, 6, p. 751.
- Meyramov G.G. and coll. 1st World Congress on Prevention of Diabetes, Copenhagen, 1996, p.
- Meyramov G.G. and coll. 2nd World Congress on Prevention of Diabetes, Rome, 1999, p.
- Meyramov G.G. and coll. 7th World Congress on Prevention of Diabetes, Madrid, 2012, p. 72–73.
- Meyramov G.G. and coll. 5th World Congress on Pancreas Transplantation, Miami, USA, 1995, p.
- Meyramov G.G. and coll. 7th Worls Congress on Pancreas Transplantation, Sydney, 1999, p.
- Meyramov G.G. and coll. 7th Worls Congress on Pancreas Transplantation, Sydney, 1999, p.
- Meyramov G.G. and coll. 4th World Congress on Pharmacology, Heidelberg, Germany, 1989, p.
- Meyramov G.G. and coll. 5th World Congress on Pharmacology, Yokogama, Japan, 1992, p.
- Meyramov G.G. and coll. 9th World Congress on Endocrinology, Nice, France, 1992, p.
- Meyramov G.G. and coll. 13th World Diabetes Congress, Sydney, Australia, 1988, p.
- Meyramov G.G. and coll. 14th World Diabetes Congress, Washington, USA, 1991, p.
- Meyramov G.G. and coll. 14th World Diabetes Congress, Washington, USA, 1991, p.
- Meyramov G.G. and coll. 15th World Diabetes Congress, Kobe, Japan, 1994, p.
- Meyramov G.G. and coll. 16th World Diabetes Congress, Helsinki, Finland, 1997, p.
- Meyramov G.G. and coll. 17th World Diabetes Congress, Mexico, 2000, p. 154–155.
- Meyramov G.G. and coll. 18th World Diabetes Congress, Paris, France, 2003, p. 83–84.
- Meyramov G.G. and coll. 18th World Diabetes Congress, Paris, France, 2003, p.
- Meyramov G.G. and coll. 20th World Diabetes Congress, Montreal, Canada, 2009, p. 429–430.
- Meyramov G.G. and coll. 20th World Diabetes Congress, Montreal, Canada, 2009, p.
- Meyramov G.G. and coll. 21st World Diabetes Congress, Dubai, UAE, 2011, p.
- Meyramov G.G. and coll. 22nd World Diabetes Congress, Melbourne, Australia, 2013, p.
- Meyramov G.G. and coll. 22nd World Diabetes Congress, Melbourne, Australia, 2013, p.