Autism spectrum disorders (ASD) are heterogeneous diseases that results from the interaction between genetic and environmental factors. There is evidence for inflammation playing a significant role in ASD. Polymorphism of genes involved in inflammation can be associated with the genetic predisposition to ASD. Cyclooxygenase-2 (COX-2) converts arachidonic acid into prostaglandin H2, which promotes acute and chronic inflammation. In this study, we investigated the association of rs2745557 COX-2 polymorphism with ASD. The study included 27 children with ASD and 23 healthy individuals. Genomic DNA was extracted from buccal swabs. Genotypes were analyzed by polymerase chain reaction restriction fragment length polymorphism. Statistical analysis showed that A allele was associated with increased risk of ASD (OR = 1.57, 95% CI = 0.44 - 5.61, p=0.49). Increased risk of ASD was found for AA genotype vs the combined GG+GA genotypes (OR = 1.52, 95% CI = 0.13 - 18.03, p=0.74) and for AA+GA genotypes vs the GG genotype (OR = 1.62, 95% CI = 0.35 - 7.45, p = 0.53). The obtained results indicate that the rs2745557 polymorphism of COX-2 gene can contribute to the genetic predisposition to ASD. Further large-scale studies with more children will be conducted to confirm our findings.
Introduction.
Autism spectrum disorders (ASD) are complex childhood neuropsychiatric disorders mainly characterized by deficits in verbal and non-verbal communication, reciprocal social interactions, stereotypic behaviors. In the last five years, the number of reported cases of children with autism in Kazakhstan has increased 1.8 times. According to the official data provided by the Ministry of Healthcare of the Republic of Kazakhstan for2018, the prevalence of ASD is 2.6 per 100.000 children, which is lower than the world average. WHO estimated that worldwide 1 in 160 children has an ASD, the incidence is 6.25 per 100.000 people. We assume that the official data do not reflect the true picture of morbidity due to the difficulties of ASD diagnostics in our country.
The etiology of this pathology is extremely complex, and, in most cases, the underlying pathological mechanisms remain unknown. In recent years, a number of works have been devoted to the study of neurochemical, immunological, structural, functional and genetic factors of the ASD etiopathogenesis. The prevailing view is that autistic disorders are pathophysiological processes caused by a combination of various exogenous factors encountered in the early stages of development, as well as genetic predisposition.
There is increasing evidence that polymorphism of candidate genes, which include genes of cell adhesion, signal proteins, regulation of degradation and transcription processes, inflammation etc is involved in ASD pathologies [1].
Cyclooxygenase (COX) participates in the conversion of arachidonic acid into prostaglandin H2, the precursor of prostaglandins and thromboxanes. There are two main isoforms of COX: COX-1 is expressed constitutively and involved in the production of prostaglandins for cellular housekeeping functions, COX-2 is an inducible form. COX-2 catalyzes the synthesis of prostaglandin E2, which promotes cell proliferation, inhibition of apoptosis and angiogenesis, and contributes to the pathological process of carcinogenesis and inflammation [2].
During inflammation, COX-2 is induced by growth factors, cytokines and proinflammatory agents, and it is thought to be involved in the production of prostanoids in neurodegenerative processes, seizures, normal neuronal functioning, neurotoxicity, and synaptic plasticity [3]. COX-2 is constitutively expressed in the neuronal tissues in regions that are critically affected in psychiatric disorders, such as the forebrain, hippocampus, hypothalamus, and amygdala [4].
Human COX-2 gene is located on chromosome 1, about 8.3 kb in size and consists of 10 exons. Genetic variations in the COX-2 gene can affect its functions, contributing to the genetic predisposition to neurodegenerative diseases, including ASD.
The aim of the study was to test this hypothesis, so we conducted a case-control study of COX-2 rs2745557 polymorphism in autistic children in the population of Kazakhstan.
Materials and methods.
Collection of clinical material in families with children diagnosed with ASD was conducted at the "Autism pobedim" Fund on the basis of the Memorandum of Cooperation with the Mercy Volunteer Society. Buccal swabs were collected from both parents of the child with ASD (proband), as well as from healthy siblings (brothers and sisters of proband) using sterile cottontipped applicators in individual plastic packs. The collected material was transported to the Institute of General Genetics and Cytology in a portable refrigerated container within several hours after the collection and then frozen at -80° C for further molecular-genetic studies.
Collection of clinical material was conducted exclusively on a voluntary basis after signing an informed consent by at least one of the parents. The protocol of the study was approved by the Ethics Committee of Asfendiyarov's Kazakh National Medical University (№57, 05.09.2017). The protocol is based on the bioethical rules in human and animals research "Principles of Medical Ethics", approved by the General Assembly of the United Nations (1992) and Council of Europe Convention on Bioethics (1997).
In addition to signing an informed consent, a detailed questionnaire and necessary psychological testing of children were conducted using CARS and M-Chat-R test.
As the control group, brothers and sisters of children with ASD were recruited. They were also examined using CARS and M- Chat-R tests to rule out any neurological and psychiatric problems.
DNA from buccal swabs was isolated using the DNA extraction kit (AmpliSens). The DNA samples were stored at -20°C and -80°C.
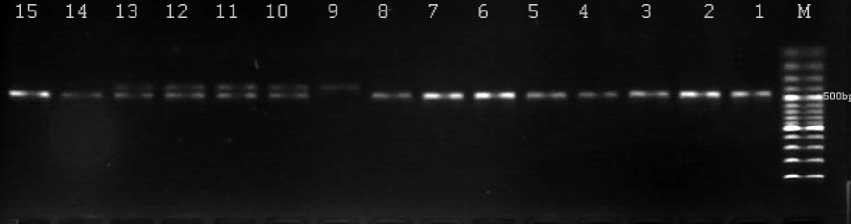
The PCR-RFLP assay was used for the genotyping of COX-2 rs2745557 single nucleotide polymorphism. 50ng of target genomic DNA was amplified in 20ц1 PCR mixture, containing 10ц1 2x PCR Master Mix (0.05 U/^L Taq DNA polymerase, reaction buffer, 4 mM MgCl2, 0.4mM of each dNTP (ThermoScientific, Lithuania)) and 5 pM of each primer. The following primers were used to amplify 439 bp PCR products: forward 5'- GAGGTGAGAGTGTCTCAGAT-3' and reverse 5'- CTCTCGGTTAGCGACCAATT-3'. The PCR cycling conditions were one cycle at 94°C for 5 min; 35 cycles of 94°C for 30 sec, 55°C for 30 sec and 72°C for 30 sec, a final extension at 72°C for 10 min. PCR products were digested at 370C for 8-16 hours with 1-3U of TaqI (Fermentas) and analyzed on 1.4% agarose (“TopVision Agarose”, Fermentas, Lithuania) gels using a 100 bp DNA ladder marker (Sigma-Aldrich, USA). Variant A allele had an TaqI restriction site that resulted in two bands (353+76 bp), and G allele lacked the TaqI restriction site and therefore produced a single 439-bp band, GT heterozygote produced three bands (figure 1).
Figure 1 - PCR-RFLP based genotyping of COX-2 rs2745557 polymorphism: lane M - 100 bp DNA ladder, lanes 1-8, 14, 15- GG homozygous genotype (439 bp), lanes 10-13 -GA heterozygous genotype (439, 353 and 76 bp), lane 9- AA homozygous genotype (353 and 76 bp)
Statistical analysis was performed using the “Case-Control Study Estimating Calculator” by TAPOTILI company (Laboratory of Molecular Diagnostics and Genomic Dactyloscopy of “GosNIIGenetika” State Scientific Centre of Russian Federation). Results and discussion.
Characteristics of the study population. 27 autistic children (AC) and their 23 healthy siblings (controls) were recruited in this study. The ethnic heterogeneity of both groups were Kazakh, Russian and other Asians. The mean age of the children at the time of testing was 8.63±6.72 years (range 1-29 years) for the AC group and 10.26±7.33 years (range 2-33 years) for the control group. In the AC group, 78% were male and 22% were female. In the control group, 48% were male and 52% were female. Diagnoses of autism were established by senior psychiatrists. The cases were assessed using the Childhood autism rating scale (CARS) and the Modified Checklist for Autism in Toddlers (M- Chat-R).
Results of genotyping. To investigate the association of rs2745557 polymorphism with increased risk of ASD, we genotyped a total of 27 AC and 23 controls.
The frequencies of genotypes and alleles did not deviate from Hardy-Weinberg equilibrium (HWE) in controls (p=0.03). Deviation from HWE in AC can be explained by small number of samples (p=0.05).
The frequencies of G and A alleles were 0.852 and 0.148 in AC, 0.900 and 0.100 in controls, respectively. The frequencies of GG, GA and AA genotypes were 0.850, 0.100 and 0.050 in ASD patients and 0.778, 0.148 and 0.074 in controls, respectively. The distribution of the genotypes in AC patients and controls as well as the ASD risk related to the rs2745557 polymorphism are summarized in table 1.
Table 1 - Genotype and allele frequencies
|
rs2745557 |
Case (%) |
Control (%) |
OR (CI 95%) |
X2 |
P |
|
Genotypes |
|||||
|
GG |
21 |
17 |
0.62 (0.13 - 2.84) |
||
|
GA |
4 |
2 |
1.57 (0.26 - 9.53) |
0.39 |
0.82 |
|
AA |
2 |
1 |
1.52 (0.13 - 18.03) |
||
|
Dominant model |
|||||
|
GG |
21 |
17 |
0.62 (0.13 - 2.84) |
0.39 |
0.53 |
|
AA+GA |
6 |
3 |
1.62 (0.35 - 7.45) |
||
|
Recessive model |
|||||
|
GG+GA |
25 |
19 |
0.66 (0.06 - 7.81) |
0.11 |
0.74 |
|
AA |
2 |
1 |
1.52 (0.13 - 18.03 ) |
||
|
Allele |
|||||
|
G |
- |
- |
0.64 (0.18 - 2.29) |
0.48 |
0.49 |
|
A |
- |
- |
1.57 (0.44 - 5.61) |
||
As shown in Table 3, the A allele was associated with increased risk of ASD (OR = 1.57, 95% CI = 0.44 - 5.61, p=0.49). Furthermore, increased risk of ASD was found for AA genotype vs the combined variant of GG+GA genotypes (OR = 1.52, 95% CI = 0.13 - 18.03, p=0.74), and for AA+GA genotypes vs the GG genotype (OR = 1.52, 95% CI = 0.13 - 18.03, p=0.74). Unfortunately, identified associations were not significant due to the small number of study groups. Further large-scale studies with more children will be conducted to confirm our findings.
Discussion.
It has been recently demonstrated that the level of inflammation in different regions of the brain is increased in ASD [5-8]. Elevated production of proinflammatory cytokines (interleukin (IL)-1 and IL-6) and chemokines (IL-8) [9-11], microglial and astrocytic activation [12-16], and aberrant expression of nuclear factor kappa-light-chain-enhancer of activated B cells [17] were detected in children with ASD.
COX-2 is оne of the main players in inflammatory reactions in various tissues, including brain tissue. Expression of COX-2 is induced by pro-inflammatory molecules and is responsible for the production of prostanoids in acute and chronic inflammatory conditions. In the rat brain, COX-2 mRNA and immunoreactivity were found in dentate gyrus granule cells, pyramidal cell neurons in the hippocampus, the piriform cortex, superficial cell layers of neocortex, the amygdala, and at low levels in the striatum, thalamus and hypothalamus [18, 19].
Yoo H.J. et.al. demonstrated three evidences supporting the involvement of COX-2 in ASDs [20]. First, it was shown that laminar pattern of cortical COX-2 immunoreactivity is disrupted and COX-2-positive neurons are decreased in Rett syndrome, a variant of ASD [21]. Second, COX-2 participates in the induction of long-term potentiation, a main model of synaptic plasticity, subsequent learning and memory. Thus, it was shown that rats subjected to destruction of basal forebrain cholinergic neurons during the first post-natal week had decreased levels of hippocampal COX-2 in adulthood and impaired social memory, which is a critical deficit in human ASDs [22]. Third, there is a link between disrupted immune responses and ASD. Since cytokines and other products of the immune system have widespread effects on neuronal pathways and can alter behaviors such as mood and sleep, it is possible that COX-2 is involved in these abnormal immune processes observed in ASDs [23].
So, COX-2 can play role in the etiology of autism and its polymorphisms can be associated with predisposition to ASD.
The association between COX-2 polymorphism and neurodegenerative diseases has been studied. A number of studies have reported an association decreased risk of Alzheimer's disease and the C allele (-765 promoter polymorphism), which plays a protective role in the disease development [24].
The functional effect of the single nucleotide polymorphism rs2745557 in intron 1 on the activity of COX-2 protein is not yet known
However, the A-allele of the rs2745557 was significantly associated with the development of autism in the Korean population (x2 = 7.143, p = 0.008) [20].
In our study, the A-allele was also associated with increased risk of developing autism (p=0.49). Increased risk of ASD was also associated with AA and AA+GA genotypes. Further large-scale studies with more children will be conducted to confirm our findings.
In practical terms, the study of SNP-markers in children with ASD can create a scientific basis for the development of new methods of early ASD diagnostics which will allow us to determine the genetic predisposition to ASD. Despite intensive research, ASD remains extremely difficult to diagnose before age 2. Most children with ASD are diagnosed at 5-6 or older. This is a critical limitation because early intervention improves prognosis of ASD [25]. Diagnosis of the disorders at an early age using SNP- markers will enable physicians to develop new methods of early therapy that will be more effective than if they start at the age of 2, when the first symptoms appear and when ASD are most often diagnosed. All this as a whole will allow us to optimize the whole complex of medical and psychological assistance to families with children diagnosed with ASD and, as a result, to improve their social adaptation indicators.
REFERENCES
- Bobylova M.Ju., Pechatnikova N.L.) Genetika autizma (obzor zarubezhnoj literatury) // Russkij zhurnal detskoj nevrologii. - 2013. - № 8(3). - Р. 31-45.
- Piranda D.N., Abreu R.B.V., Freitas-Alves D.R., de Carvalho M.A., Vianna-Jorge R. Modulation of the prostaglandin-endoperoxide synthase 2 gene expression by variant haplotypes: influence of the 3'-untranslated region //Braz J Med Biol Res. - 2018. - № 51(2). - Р. 65-72.
- Minghetti L. Cyclooxygenase-2 (COX-2) in inflammatory and degenerative diseases //J. Neuropathol. Exp. Neurol. - 2004. - №63. - Р. 901910.
- Ibuki T., Matsumura K., Yamazaki Y., Nozaki T., Tanaka Y., Kobayashi S. Cyclooxygenase-2 is induced in the endothelial cells throughout the central nervous system during carrageenan-induced hind paw inflammation; its possible role in hyperalgesia // J. Neurochem. -
- -№ 86. - Р. 318-328.
- Enstrom A.M., Lit L., Onore C.E., Gregg J.P., Hansen R.L., Pessah I.N., et al. Immunity, neuroglia and neuroinflammation in autism // Int. Rev. Psychiatry. - 2005. -№ 17. - Р. 485-495.
- Morgan J.T., Chana G., Pardo C.A., Adim C., Semendeferi K., Buckwalter J., et al. Mkroglial adivation and indeased mkroglial density observed in the dorsolateral prefrontal спПса in autism // Biol. Psychiatry. - 2010. - № 68. - Р. 368-376.
- Tetreault N.A., Hakeem A.Y., Jiang S., Williams B.A., Allman E., Wold B.J., et al. Mkroglia in the cerebral спПса in autism // J. Autism Dev. Disord. - 2012. - № 42. - Р. 2569-2584.
- Kern J.K., Geier D.A., Sykes L.K., Geier M.R. Releva^e of Neuroinflammation and E^ephantis in Autism // Front Cell Neurosd. - 2015. - № 9. - Р. 519-528.
- Ashwood P., Krakowiak P., Hertz-Pkdotto I., Hansen R., Pessah I., Van De Water J. Elevated plasma cytokines in autism spalrum disorders provide evidence of immune dysfundion and are assodated with impaired behavioral outeome // Brain Behav Immun. - 2011. -№ 25. - Р. 40-45.
- Wei H., Zou H., Sheikh A.M., Malik M., Dobkin C., Brown W.T., Li X. IL-6 is indeased in the cerebellum of autistk brain and alters neural cell adhesion, migration and synaptk formation // J Neuroinflammation. - 2011. - № 8. - Р. 52-58.
- Jyonouchi H., Sun S., Le H. Proinflammatory and regulatory cytokine produdion assodated with innate and adaptive immune responses in сМ(1геп with autism spedrum disorders and developmental regression // J Neuroimmunol. - 2001. - № 120. - Р. 170-179.
- B^y^k R.L., Strunecka A. Immune-glutamatergk dysfundion as a central melanism of the autism spedrum disorders // Curr Med Chem. - 2009. - №16. - Р. 157-170.
- Cohly H.H., Panja A. Immunologkal findings in autism // Int Rev Neurobiol. - 2005. - №71. - Р. 317-341.
- Zimmerman A.W., Jyonouchi H., Comi A.M., Connors S.L., Milstien S., Varsou A., Heyes M.P. Cerebrospinal fluid and serum markers of inflammation in autism //Pediatr Neurol. - 2005. - № 33. - Р. 195-201.
- Vargas D.L., Nasdmbene C., Krishnan C., Zimmerman A.W., Pardo C.A. Neuroglial adivation and neuroinflammation in the brain of patients with autism //Ann Neurol. - 2005. - № 57. - Р. 67-81.
- Asadabadi M., Mohammadi M.R., Ghanizadeh A., Modabbernia A., Ashrafi M., Hassanzadeh E., Forghani S., Akhondzadeh S. Cele^x^i as adjundive treatment to risperidone in diildren with autistk disorder: a randomized, double-blind, placebo-controlled trial // Psychopharmacology (Berl). - 2013. - № 225(1). - Р. 51-59.
- Young A.M., Campbell E., Lynd S., S^kling J., Powis S.J. Aberrant NF-kappa B expression in autism spedrum condition: a medanism for neuroinflammation // Front. Psychiatry. - 2011. - № 2. - Р. 27-34.
- Yamagata K., Andreasson K.I., Kaufmann W.E., Barnes C.A., Worley P.F. Expression of a mitogen-indudble cydooxygenase in brain neurons: Regulation by synaptk adivity and glucocorticoids // Neuron. - 1993. - № 11. - Р. 371-386.
- Breder C.D., Dewitt D., Kraig R.P. Charaderization of indudble cydooxygenase in rat brain //J Comp Neurol. -1995. - № 355. - Р. 296-315.
- Yoo H.J., Cho I.H., Park M., Cho E., Cho S.C., Kim B.N., Kim J.W., Kim S.A. Assodation between PTGS2 polymorphism and autism spedrum disorders in Korean trios // Neurosd Res. -2008. - №62(1). - Р. 66-69.
- Kaufmann W.E., Worley P.F., Taylor C.V., Bremer M., Isakson P.C. Cydooxygenase-2 expression during rat neocortical development and in Rett syndrome // Brain Dev. - 1997. - № 19. - Р. 25-34.
- Rideri L., Minghetti L., Moles A., Popoli P., Confaloni A., De Simone R., Pisdpo P., Scattoni M.L., di L^a M., Calamandrei G. Cognitive and neurologkal defkits indeed by early and prolonged basal forebrain cholinergic hypofundion in rats // Exp. Neurol. - 2004. - № 189. - Р. 162-172.
- Ashwood P., Wills S., Van de Water J. The immune response in autism: a new frontier for autism research // J. Leukoc Biol. - 2006. - № 80. - Р. 1-15.
- Yermakova A.V., O'Banion M.K. Downregulation of neuronal cydooxygenase-2 expression in end stage Alzheimer's disease // Neurobiol. Aging. - 2001. - №2. - Р. 823-836.
- Dawson G., Jones E.J., Merkle K., Venema K., Lowy R., Faja S., Kamara D., Murias M., Greenson J., Winter J., et al. Early behavioral intervention is assodated with normalized brain adivity in young dildren with autism // J. Am. Acad. Child Adolescent Psyd. - 2012. - № 51. - Р. 1150-1159.