Oxidative stress plays an important role in arising, development and progression of atherosclerosis as the main cause of ischemic heart disease (IHD) and myocardial infarction (MI) manifestations. Experimental studies on model objects continue to be a significant tool for revealing molecular and genetic aspects of atherogenesis. However, many questions remain unanswered and results are controversial. In this study, we characterized expression of two major vascular wall proteins - extracellular superoxide dismutase (ex-SOD) and glutathione peroxide dismutase 1 (GPX1) in rabbit atherosclerotic lesions compared to normal rabbit aorta. Using immunohistochemistry, we identified the presence of EC-SOD and GPX1 proteins in rabbit normal aorta and atheroma lesions and established their expression in specific atherosclerotic areas distinctive for both enzymes. Also, we confirmed the higher expression level as for EC-SOD, as for GPX1 proteins in aorta tissues affected by atherosclerosis.
Redox signaling pathways influence many physiological processes providing a strict crosstalk within and between cells in a vessel wall. Oxidative stress induced by inflammation in the coronary artery wall affects excessive oxidizing of lipoproteins, nitric oxide (NO) molecules, DNA and other cell's proteins leading to normal homeostatic state disruption and atherosclerosis development.The main free radical of inflammation - superoxide anion (O2-), was found as a prominentfeatureof the atherosclerotic plaques, produced by activated SMC, macrophages, endothelial cells and some otherscells in abundant amounts. Polyunsaturated fatty acids of lipids in cells membranes, lipoprotein particles (LP) became oxidized in a chain-reaction manner.Highly regulated endothelium derived NO, involved in vascular dilatation, reacts rapidly with O2- forming toxic peroxynitrite (ONOO-).High dose and/or inadequate generation of superoxide anion lead to activation of enzymes involved in O2- removal -superoxide dismutases (SODs), catalase, peroxiredoxins (Prxs), or glutathione peroxidases (GPx). SODs are the first line of antioxidant defense through dismutation of O2- to H2O2. Isoenzyme EC-SOD is of great interest for vascular homeostasis. Its unique role in vasculature is determined by theextracellular localization, anchoringirreversibly to type I collagen and heparan sulfate and thus on guard of vascular tissue integrity. EC-SOD was found to be highly expressed in human[1], mouse and rabbit atherosclerotic lesions [] with time-dependent downregulationtendency.
Formed through dismutation reaction H2O2 is a second signaling molecule in tissues, but generated in high amounts under oxidative stress, became a dangerous agent. Enzymatic inactivation of derived H2O2 is achievedprincipally by glutathione peroxidases superfamily. Clinical and experimental studies suggest a crucial role for GPX1 isoenzyme in atherogenesisand cardiovascular disease promotion[2], [3], [4]. But, available data on expression and regulation of GPX1 protein under atherosclerosis especially in rabbit experimental model are rare and incomplete.
Based on the importance of these proteins for the development of atherosclerosis pathology, we sought to determine and compare the exact locations of GPX1 and EC-SOD proteins and itsexpression level under atherosclerotic lesions. In our study we used a suitable atherosclerotic model object - rabbit, normal and after six-months fed high cholesterol diet.
Materials and methods.
Rabbit experimental atherosclerosis model
Total 10 male Chinchilla rabbits of 6 months old, initial weight 3500g and without any physical pathology was investigated. Atherosclerosis was induced in 5 male Chinchilla rabbits by high 2% cholesterol diet "Purina rabbit chow" (Dytes Inc., USA) lasting for 6 months. Five rabbits were on a regular diet (food "Purina rabbit chow", Dytes Inc., USA) and served as a control. After 6 months, cervical dislocation under anesthesia were performed and thoracic aorta tissue (0.5x0.7 mm)were prefixed in 10% buffered formalin solution and embedded in paraffin blocks by classical method.
Animal experimental procedures were approved by the Local Bioethics Committee of the Institute of Cardiology and Internal Diseases (Protocol № 10 of March 12, 2009).
Immunohistochemistry.
Immunohistochemistry was done according to avidin-biotin peroxidase complex methodby using rabbit monoclonal antibodies to Glutathione Peroxidase 1(Abcam, USA) and extracellular Superoxide Dismutase 3 (Abcam, USA). Before immunostaining, blocking of endogenous peroxidase activity was performed by incubation with 3% H2O2 for 15 min at room temperature with subsequent antigen retrieval by heating slides in 10mM citrate buffer (pH-6).
Protein extraction from paraffin embedded rabbit aorta tissues Total protein extracts from paraffin embedded rabbit aorta tissues were isolated according to Kimimasa Ikeda et al.[5]. Protein assay.
Concentration of total protein lysates was measured by M. Bradford method [6]. BSA solution in different dilutions was used as a reference standard.
SDS-PAGE and Western blot (WB) analysis 20 ng of total protein extracts were separated in 12% SDS-PAGE by electrophoresis at a constant 160 volts. Proteins were transferred into PVDF membrane overnight at constant 30mA, overnight, +4C°. Nonspecific binding was inhibited by blocking buffer (0.1% casein in 0.2x PBS, 0.1% Tween-20) during overnight at +4C°. The membranes were incubatedwith the primary antibody at +4C° for 2 hr at the 1:3000 dilutions. After being washed with TBST (50 mMTris-HCl, pH 7.5, 150 mMNaCl, 0.1% Tween-20), the membranes were incubated for 1 h with anti-rabbit IgG secondary antibody conjugated with horseradish peroxidase (HRP).Membranes were developed with the Blue Basic Autorad Film 8x10 (ISC Bio Express) in 1.25mM luminol, 0.68mM coumaric acid and 0.01% H2O2 buffer.
Statistical analysis.
All data results are expressed as mean ±SD.A p-value of <0.05 was considered statistically significant. Distribution of variables between two-rabbit groups was compared by using a Student's t- test.
Results.
Immunohistochemical identification of EC-SOD and GPX1 proteins were studied in 10 different sets of rabbit thoracic aorta tissue samples.Positive EC-SOD and GPX1 proteins immunostaining were found in normal and atherosclerotic aortas. Pic 1A through 1D shows representative microphotographs of GPX1 immunostaining in serial sections of normal (Picture A, C) and atherosclerotic aortas (B, D). A strong immunostaining of GPX1 was seen in intimalayer under advanced atheroma lesions (Picture 1D). Also, positive, but less intensity GPX1 immunoreactivity was detected in early atherosclerotic lesions in the intima layer (Picture 1B). In control sections there were no intensive immunoreactivity to GPX1 protein.
Staining with EC-SOD antibody was also positive in normal rabbit aorta tissues sections, but low intensity (Picture 1 E). EC-SOD signal was observed only in the intima layer mostly directly under epithelium. There were no visible EC-SOD signals in media and adventitia layers (Picture 1 G). In contrast, atherosclerotic aorta tissues sections showed positive and intense staining signal across the intima, especially under advanced atheromas (Picture 1 F). Also, the intensive EC-SOD antibody signal was noted in the media layer near the adventitia (Picture 1H).


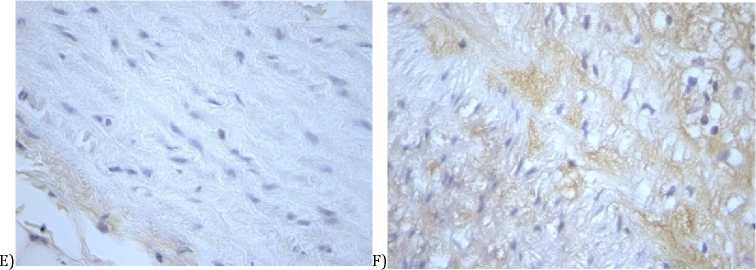
Picture 1 - GPX1 and EC-SOD immunohistochemical localization in aorta tissue. A - GPX1 in normal aorta x86; B - GPX1 in atherosclerotic aorta x86; C - GPX1 in normal aorta x20; D - GPX1 in atherosclerotic aorta x20; E - EC-SOD in normal aorta x86; F - EC-SOD in atherosclerotic aorta x86; G - EC-SOD in normal aorta x20; H - EC-SOD in atherosclerotic aorta x20

Western blots revealed bands of EC-SOD and GPX1 proteins at the expected molecular size 22kDa and 26kDa respectively (Picture 2 A and B). Densitometryanalysis of expression also showed increased levelsfor both enzymes in atherosclerosis compared with the control group (Picture 2 A and B).
B
Picture 2 - Western blot (WB) analysis of EC-SOD and GPX1 in normal and atherosclerotic aorta tissues. A -expression of EC-SOD protein in aorta tissue from control and atherosclerosis groups (SDS-PAGE and immunoblotting with rabbit monoclonal antibody to SOD3). B - densitometric analysis of WB for EC-SOD protein, p=0.000. C - expression of GPX1 protein in aorta tissue from control and atherosclerosis groups (SDS PAGE and immunoblotting with rabbit monoclonal antibody to GPX1). D - dencitometric analysis of WB for GPX1 protein, p=0.002.




Discussion.
Chinchilla rabbit atherosclerosis status. In our study, we used experimental model of atherosclerosis in rabbits (Chinchilla breed). Historically, rabbits were the first and successful model for atherosclerosis basic research [7]. Despite the existing differences between rabbits and human in terms of lipid metabolism [8], [9] and in arterial susceptibility to
atherosclerosis[10], studies in translational medicine show that this model of atherosclerosis is the most suitable than mice and rats[11].Hypercholesterolemia and atherosclerosis promotion in rabbits is directly proportional to the dietary cholesterol and/or fats uptake [12], [13], [14].
TakingintoaccountthesefeaturesandtheChinchillabreedhyperchol esterolemiainduction[15],we used a standard "Purina rabbit chow" with the addition of 2% cholesterol for 6 months. This time was enough for advanced atherosclerotic plaques formation, without systemic lipid degeneration in other organs. Also, due to differences in plasma cholesterol metabolism between rabbit males and females [16], we used only males in the study.
EC-SOD and GPX1 proteins expression profile. According to numerous studies, EC-SOD and GPX1 proteins areundoubtedly cardiovascular diseases associated proteins. Manifestation of arterial hypertension[17][18][19], atherosclerosis[20][21][22], ischemia[23], myocardial infarction and other cardiovascular disorders provoking states may be aggravate by an expression and regulation of EC-SOD and GPX1[24].The precise regulatory mechanisms for the EC-SOD and GPX1 genes expression under normal and atherosclerosis states are unknown. Several mechanisms for the EC-SOD have been proposed[24][25][26], [27]. Mikko O. Laukkanen et al., assumedthe existence of the additional possible mechanism throughde/methylation pattern ofthe EC-SOD gene in atherosclerosis[28].
The data obtained by us also support the above-listed studies and views on the important role of antioxidant defense enzymes in the development of atherosclerosis. The increased expression of EC-SOD and GPX1 proteins in rabbit aortic tissues indicates a shift in the oxidation-reduction potential in the tissue and intensification of antioxidant processes. Deficiency and/or overexpression of these proteins may speed up[22][29]or slow down[30]the atherosclerosis pathological states.Identification of specific biochemical pathways requires further studies on suitable experimental models and human.
REFERENCES
- Landmesser U. et al. Vascular Extracellular Superoxide Dismutase Activity in Patients With Coronary Artery Disease: Relation to Endothelium-Dependent Vasodilation // Circulation. - 2000. - Vol. 101, №19. - P. 2264-2270.
- Guo Z.M. et al. Changes in expression of antioxidant enzymes affect cell-mediated LDL oxidation and oxidized LDL-induced apoptosis in mouse aortic cells // Arterioscler. Thromb. Vasc. Biol. - 2001. - Vol. 21, №7. - P. 1131-1138.
- 'T Hoen P.A.C. et al. Aorta of ApoE-deficient mice responds to atherogenic stimuli by a prelesional increase and subsequent decrease in the expression of antioxidant enzymes // Circ. Res. - 2003. - Vol. 93, № 3. - P. 262-269.
- Blankenberg S. et al. Glutathione peroxidase 1 activity and cardiovascular events in patients with coronary artery disease. // N. Engl. J. Med. - 2003. - Vol. 349, № 17. - P. 1605-1613.
- Ikeda K. et al. Extraction and Analysis of Diagnostically Useful Proteins from Formalin-fixed, Paraffin-embedded Tissue Sections // J. Histochem. Cytochem. - 1998. - №4. - Р. 88-97.
- Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding // Anal. Biochem. - 1976. - №2. - Р. 52-57.
- Steinberg D. Thematic review series: the pathogenesis of atherosclerosis. An interpretive history of the cholesterol controversy, part I // J. Lipid Res. - 2004. - №12. - Р. 21-27.
- Koike T. et al. Expression of human ApoAII in transgenic rabbits leads to dyslipidemia: A new model for combined hyperlipidemia // Arterioscler. Thromb. Vasc. Biol. - 2009. - №6. - Р. 64-72.
- Koike T. et al. Human C-reactive protein does not promote atherosclerosis in transgenic rabbits // Circulation. - 2009. - №11. - Р. 136147.
- Fan J. et al. Rabbit models for the study of human atherosclerosis: From pathophysiological mechanisms to translational medicine // Pharmacol. Ther. - 2015. - №3(8). - Р. 88-97.
- Endo A. The discovery and development of HMG-CoA reductase inhibitors // J. Lipid Res. - 1992. - №5. - Р. 52-61.
- Bocan T.M.A. et al. The relationship between the degree of dietary-induced hypercholesterolemia in the rabbit and atherosclerotic lesion formation // Atherosclerosis. - 1993. - №1. - Р. 149-152.
- Kritchevsky D. et al. Experimental atherosclerosis in rabbits fed cholesterol-free diets // Atherosclerosis. - 1982. - №3. - Р. 246-258.
- Kritchevsky D. Trans fatty acid effects in experimental atherosclerosis // Fed. Proc. - 1982. - №12. - Р. 47-56.
- Henderson J.D., Webster W.S., Clarkson T.B. Diet-induced hypercholesterolemia in the chinchilla and rabbit // Atherosclerosis. - 1970. - №7. - Р. 22-27.
- Roberts D.C.K. et al. Plasma cholesterol concentration in normal and cholesterol-fed rabbits. Its variation and heritability // Atherosclerosis. - 1974. - №13. - Р. 108-117.
- Gomez-Marcos M.A. et al. Serum Superoxide Dismutase Is Associated with Vascular Structure and Function in Hypertensive and Diabetic Patients. // Oxid. Med. Cell. Longev. - 2016. - №9. - Р. 69-72.
- Lob H.E. et al. Induction of hypertension and peripheral inflammation by reduction of extracellular superoxide dismutase in the central nervous system // Hypertension. - 2010. - №5. - Р. 112-117.
- Ardanaz N. et al. Lack of glutathione peroxidase 1 accelerates cardiac-specific hypertrophy and dysfunction in angiotensin II hypertension // Hypertension. - 2010. - №6(7). - Р. 71-77.
- Takeshita S. et al. Shear stress enhances glutathione peroxidase expression in endothelial cells. // Biochem. Biophys. Res. Commun. - 2000. - №4. - Р. 151-157.
- Callegari A. et al. Gain and loss of function for glutathione synthesis: Impact on advanced atherosclerosis in apolipoprotein E-deficient mice // Arterioscler. Thromb. Vasc. Biol. - 2011. - №4. - Р. 31-39.
- Cheng F. et al. Impact of Glutathione Peroxidase-1 Deficiency on Macrophage Foam Cell Formation and Proliferation: Implications for Atherogenesis // PLoS One. - 2013. - №8. - Р. 42-52.
- Lim C.C. et al. Glutathione peroxidase deficiency exacerbates ischemia-reperfusion injury in male but not female myocardium: insights into antioxidant compensatory mechanisms // Am. J. Physiol. Heart Circ. Physiol. - 2009. - №11. - Р. 28-37.
- Fukai T. et al. Extracellular superoxide dismutase and cardiovascular disease // Cardiovascular Research. - 2002. - №18. - Р. 169-181.
- Fattman C.L., Schaefer L.M., Oury T.D. Extracellular superoxide dismutase in biology and medicine // Free Radical Biology and Medicine. - 2003. - №1. - Р. 248-256.
- Laukkanen M.O. et al. Extracellular superoxide dismutase regulates the expression of small GTPase regulatory proteins GEFs, GAPs, and GDI // PLoS One. - 2015. - №4. - Р. 12-24.
- Laatikainen L.E. et al. SOD3 decreases ischemic injury derived apoptosis through phosphorylation of Erk1/2, Akt, and Foxo3a // PLoS One. - 2011. - №6. - Р. 103-115.
- Laukkanen M.O. et al. Local hypomethylation in atherosclerosis found in rabbit ec-sod gene // Arterioscler. Thromb. Vasc. Biol. - 1999. - №13. - Р. 66-75.
- Lewis P. et al. Lack of the antioxidant enzyme glutathione peroxidase-1 accelerates atherosclerosis in diabetic apolipoprotein E-deficient mice // Circulation. - 2007. - №17. - Р. 29-38.
- Weiss N. et al. Overexpression of cellular glutathione peroxidase rescues homocyst(e)ine-induced endothelial dysfunction. // Proc. Natl. Acad. Sci. U. S. A. - 2001. - №15. - Р. 66-72.