Infection associated with hepatitis C virus (HCV) is a major health problem and is one of the most important causes of chronic liver diseases and the most common causes of end stage liver diseases. After a long-time of discussions, the link between HCV and autoimmune disorders was proven. Inteferon treatment may also play a huge role in these processes. To prevent this, analysis of thyroid gland's hormones, including antibodies, must be done before, during and after 6 months of IFN therapy, to find any abnormalities timely. New therapy is safer, with less side effects, but still are very expensive and a large proportion of patients may not have access to them.
Table 1 - Worldwide statistic data shows percentage in thyroid dysfunction and thyroid antibodies development among HCV patients treated by IFN [13].
Risk factors that may contribute or prevent autoimmune thyroid diseases.
Genetic factors contribute for about 70% to 80% and environmental factors for about 20% to 30% to the pathogenesis of autoimmune thyroid disease (AITD).
Relatives of AITD patients carry a risk to contract AITD themselves. The 5-year risk can be quantified by the so- called Thyroid Events Amsterdam-score, based on serum thyroid-stimulating hormone, thyroid peroxidase (TPO)- antibodies and family history.
|
Country |
Treatment |
No. (M/F) |
Newly developed thyroid antibody n (%) |
Newly developed thyroid dysfunction n (%) |
|
France |
IFN-α |
68 (39/29) |
4 (5.9) |
8 (12) |
|
Japan |
IFN-α |
109 (77/32) |
2 (1.9) |
9 (8.2) |
|
Italy |
IFN-α |
114 (79/35) |
36 (31.5) |
8 (7) |
|
Spain |
IFN-α |
144 (95/49) |
7 (4.9) |
4(2.8) |
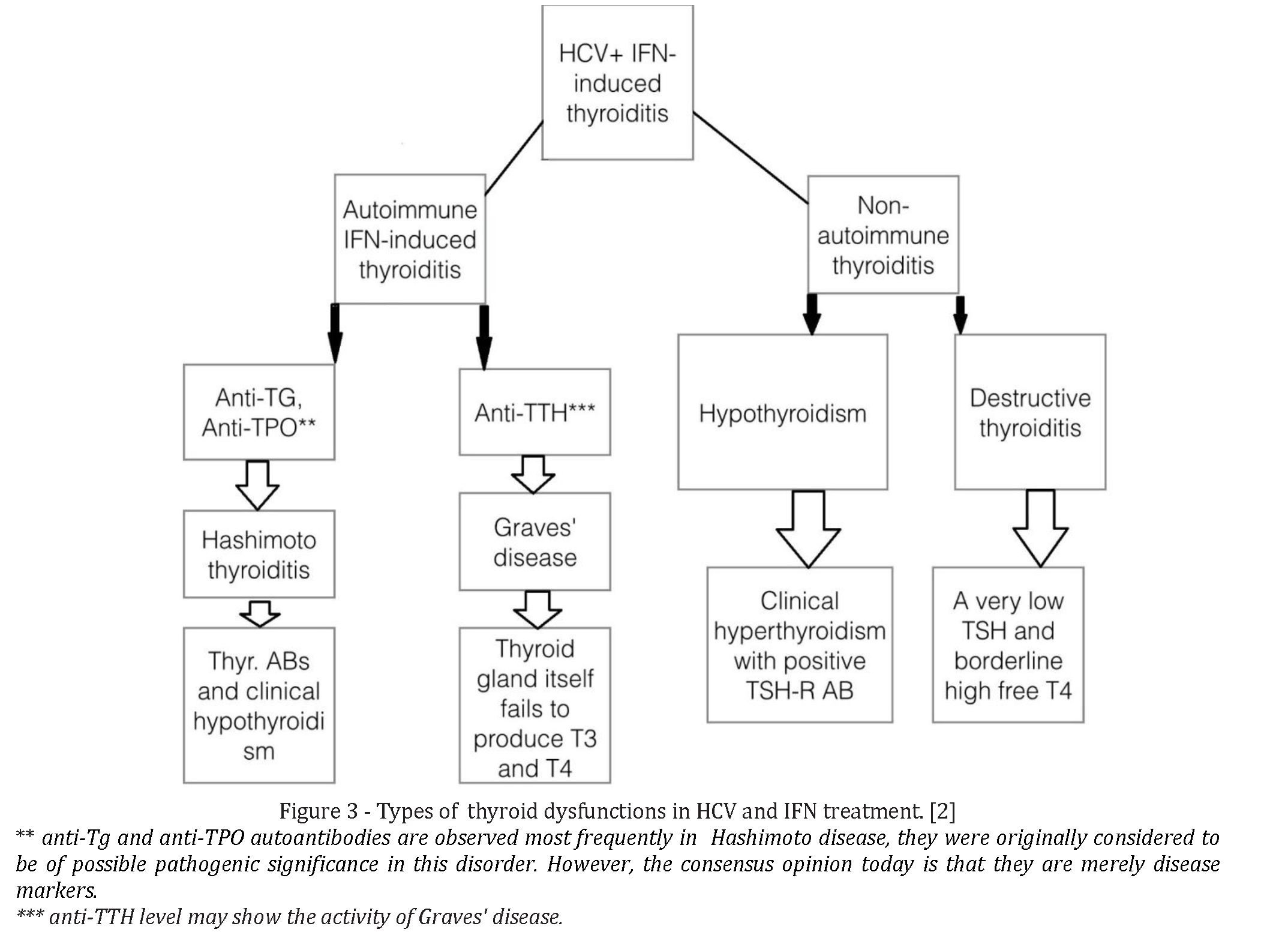
IIT is more common in females than in males. According to different studies, females appeared to have a 4.4 times higher risk of developing secondary thyroid disease to IFN-α based therapy in comparison with males. Females' susceptibility may be due to the effects of estrogenic sex steroids in promoting autoimmunity, or it could be due to the susceptibility gene on the X-chromosome, since females have two X-chromosomes, so males are less likely to inherit the gene. IIT is considered a major complication for those who are treated with IFN-α based therapy. IIT is classified mainly into two types: either autoimmune (i.e., Hashimoto's thyroiditis and Grave's disease) or non- autoimmune (e.g. destructive thyroiditis and non- autoimmune hypothyroidism) [15, 23, 30]. The postpartum period is associated with an increased risk of AITD. Taking together, preventive interventions to diminish the risk of AITD are few, not always feasible, and probably of limited efficacy. [3]
To stop smoking decreases the risk on Graves disease but increases the risk on Hashimoto disease.
Moderate alcohol intake provides some protection against both Graves and Hashimoto disease.
Low selenium intake is associated with a higher prevalence of thyroid autoimmunity, but evidence that selenium supplementation may lower TPO antibodies and prevent subclinical hypothyroidism remains inconclusive. Low serum vitamin D levels are associated with a higher prevalence of TPO antibodies, but intervention studies with extra vitamin D have not been done yet.
Stress may provoke Graves hyperthyroidism but not Hashimoto thyroiditis.
Estrogen use have been linked to a lower prevalence of Graves disease.
The most active natural vitamin D metabolite, 1,25- Dihyroxyvitamin D3, effectively prevents the development of autoimmune thyroiditis. 1,25(OH)2D3 exerts its immunomodulatory actions by inhibiting HLA class II expression on endocrine cells, proliferation of T cell and secretion of inflammatory cytokines.
Vitamin D has recently been reported to play significant roles in the regulation of immune system, the process of erythropoiesis and thyroid functions. Deficiency of vitamin D was also found to correlate with an increased incidence of autoimmune diseases. Vitamin D supplementation enhances innate immunity and reduces the severity of autoimmunity. Vitamin D levels were found to be lower in patients with AITDs than in healthy people. Deficiency of vitamin D was also linked to the presence of anti-thyroid antibodies and abnormal thyroid functions. [2]. Several studies have indicated that VitD supplementation is useful for the prevention/treatment of anemia and thyroid disorders. However, little is known about the potential effect(s) for vitamin D as a prophylactic/treatment agent against these side effects during the treatment of CHC with Peg-IFN-α based therapy. Further studies with large number of patients are required to determine whether supplementation with vitamin D during the treatment of CHC with Peg-IFN-α based therapy is useful in increasing the rates of SVR and preventing the development of associated adverse effects. According to Poupak et al, all risk factors may be collected in 7 groups with own influence on development of autoimmune thyroid diseases:
- Age: the prevalence of disease tends to increase with age.
- Genetic: a significant association between Hashimoto's thyroiditis and some histocompatibility antigens (HLA-DR, HLA-DR5, and some DQ alleles) is demonstrated. Many other susceptibility genes have been associated with AT; for example, specific CTLA4 gene polymorphisms are linked to a possible development of antithyroid antibodies .
- Iodine: an increased AT prevalence is observed in areas of iodine sufficiency, compared with iodine- deficient areas.
- Selenium: a selenium deficit is linked to a higher AT prevalence.
- Irradiation: AT occurs more frequently after the exposure to low doses of radiations
- Cytokine: the treatment with Interferon- (IFN-) α, or with Interleukin- (IL-) 2, can promote the onset of AT in predisposed patients
- Infections: it was seen that several viral infections can predispose to an AT in animals. Moreover, different studies tried to associate AT with viral infections in humans with conflicting results [4] Immunopathogenesis of HCV Infection and AITD.
Several molecular mechanisms have been suggested for the association of CHC with AT:
- molecular mimicry or cross-reactivity which may occur between viral antigens and thyroidal antigens (Figure 1).
- heat shock proteins expression in thyroid gland.
- abnormal expression of MHC class II molecules by thyrocytes.
- Changes in self-antigen expression due to viral infection, or recognition of cryptic epitopes; bystander activation of auto reactive T-cells by cytokine release during the local inflammatory response caused by virus.
- IL-6 influence.
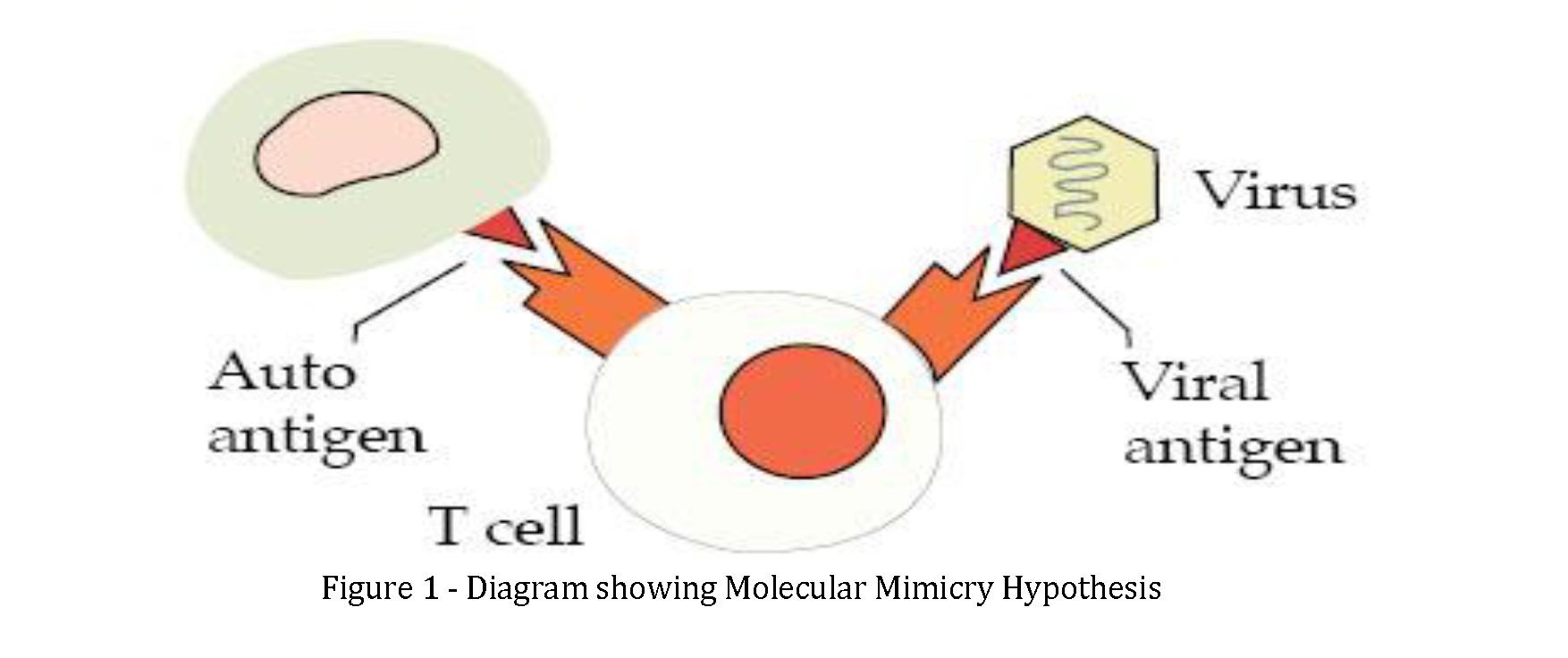
The molecular mimicry hypothesis suggest that a certain AG has a great degree of similarity with endogenous structures. Mistaken identity triggers the host immune system (autoantibodies) to attack foreign as well as endogenous targets when infected with organism.
d) Cytokines, chemokines and their receptors may contribute either directly or indirectly in abovementioned mechanisms. These factors are low molecular weight, function as chemical messengers and are interact with one another in complex ways. They are synthesized by multiple cell types and can have various functions depending upon the cell that produces it and the cell upon which the cytokine acts. Therefore, it is difficult to draw conclusions with regard to the specific role of each cytokine in mediating the observed pathophysiological effects. However, there is, a growing recognition that aberrant cytokine expression appears to play an important role in the pathogenesis of many human autoimmune diseases, including virus-induced thyroid autoimmunity. Consequently, the involvement of these mediators in disease cannot be ignored.
An increased expression of IFN-γ and IFN-γ inducible chemokines , in particular (C-X-C motif) ligand 10 chemokine (CXCL10), has been shown in hepatocytes and in lymphocytes of HCV infected patients, directly related to the degree of inflammation and to an increase in circulating levels of IFN-γ and CXCL10 . CXCL10 is one of chemokines with C-X-C motif. IP-10 activates specifically CXCR3 receptor that is a G protein-coupled receptor with seven transmembrane domains mainly expressed in T activated lymphocytes, natural-killer cells (NKs), macrophages, and B cells. Recent studies showed that CXCL10 expression in serum and/or tissue levels is increased in autoimmune organ-specific diseases, such as type 1 diabetes, or systemic rheumatological diseases like rheumatoid arthritis, systemic lupus erythematosus, systemic sclerosis, sarcoidosis, and psoriatic arthritis. High levels of CXCL10 are present in patients with AT, in particular in the presence of hypothyroidism, and an involvement of T-helper (Th)1 immune response in the induction of AT, GD, and Graves' ophthalmopathy has been demonstrated, suggesting that intrathyroidal lymphocytes and/or thyrocytes may be the source of CXCL10. Furthermore, the presence of HCV in the thyroid of chronically infected patients has been recently shown. e) Also, IL-6 was modestly but significantly increased in patients with AT, which may suggest another way of pathogenesis. Specific IL-6 binding sites have been also identified in thyroid cells, which reduces TSH-mediated iodine uptake, thyroid peroxidase mRNA expression in response to TSH, and thyroid hormone release through the TSH-dependent mechanism. Both in GD and in HT, membrane attack complexes of complement occur around thyroid follicles. Formation of these complexes may result in prostaglandin E2, IL-1a, and IL-6 production, which promote infiltration of lymphocytes leading to cell destruction. In GD, proinflammatory cytokines such as IL- 6 may further induce the synthesis of external thyroidstimulating antibodies that bind to TSHR. IFNα can also contribute to an autoimmune inflammatory response via a variety of mechanisms, such as reducing T regulatory cell function and alterations in immunoglobulin production. Th1 polarization may constitute a potentially important therapeutic effect of IFNα and may contribute in the pathogenesis of IIT. This deduction depends partly on observations such as greater increase in type 1 helper T cells in hepatitis C patients who developed IIT. However, there are some conflicts regarding these results and several studies indicated that IFNα could influence the production of type 2 cytokines.
On the above mentioned bases, it has been speculated that HCV thyroid infection may act by upregulating CXCL10 gene expression and secretion in thyrocytes recruiting Th1 lymphocytes that secrete IFN-γ and tumor necrosis factor- (TNF-) α. These cytokines induce CXCL10 secretion by thyrocytes, thus perpetuating the immune cascade, which may lead to the appearance of AITD in genetically predisposed subjects (Figure 2).
Treatment of CHC.
The traditional treatment for CHC is a combination of a weekly injection of pegylated interferon-α (Peg-IFN-α) with daily oral ribavirin (RBV) and the duration of the treatment is based on the viral genotype [1]. Although new direct acting antiviral (DAA) drugs have been developed, the treatment of CHC could still be based on a weekly injection of Peg-IFN-α-2a or -2b plus a daily weight-based dose of RBV with or without the new antiviral therapy depending on the progression of liver damage and the presence of other extrahepatic manifestations [2, 6-8]. Furthermore, the new antiviral drugs are expensive and therefore Peg-IFN-α based therapy could still be the standard of care especially for treatment naïve patients with no liver cirrhosis and/or

for those living in developing countries and for whom access to the new drugs is not definite due to its high cost. Several disadvantages are associated with Peg-IFN-α based therapy during the treatment of CHC. These include low response rate (e.g. 50% for genotypes 1 and 4) and the development of several drug induced side effects that could lead to dose reduction or termination of treatment [6, 10-14]. CHC and its treatment with Peg-IFN-α based therapy are associated with several extra-hepatic complications including hematological and endocrinological abnormalities. The most prevalent side effects associated with the traditional treatment of CHC are anemia and thyroid disorders
Thyroiditis associated with CHC and IFN-α therapy. Strong correlations between liver damage and thyroid disorders have been also reported [20]. Non-alcoholic fatty liver diseases (NAFLD) and abnormal liver enzymes are significantly associated with hypothyroidism and the
prevalence of liver diseases and enzymes increase steadily with increasing grades of hypothyroidism [20]. Furthermore, a decrease in serum triiodothyronin (T3) concentration and thyroxine (T4) ratio is frequently observed in patients with liver cirrhosis probably due to impaired conversion of T4 to T3 in the liver [21].
Thyrotoxicosis is also associated with a variety of abnormalities of liver function [22] and results from a recent study suggests that low free T4 (FT4) concentrations are associated with hepatic steatosis [24]. Serum thyroid stimulating hormone (TSH) level was also significantly higher in NAFLD and it has also been suggested that measurement of free T3 and T4 levels may all be useful as predictors of mortality in intensive care patients who have cirrhosis [20]. Thyroiditis is a major clinical problem especially for patients with chronic HCV infection [25-28] (Figure 3).
Thyroid abnormalities following interferon therapy have also been described in children receiving interferon for hepatitis C infection [32]. Some of these complications of IFN therapy, especially thyrotoxicosis, can be severe and may interfere with adequate interferon therapy in patients with hepatitis C infection [24, 28]. Moreover, because the symptoms of hypothyroidism such as fatigue, hair loss, myalgia, and weight gain may be attributable to hepatitis C or IFN therapy, the diagnosis of hypothyroidism in these patients may be delayed [29]. This delay may lead to development of further complications. Thus, IIT represents a major clinical problem for patients with chronic HCV infection and who
receive interferon for treatment that may interfere with their treatment course [ 27, 28].
Autoimmune thyroid diseases (AITD) are strongly influenced by genetic factors and therefore they are likely to affect the etiology of IIT.
Actually, the presence of HCV infection and IFN-α therapy might induce thyroiditis in genetically inclined individuals [30]. IFN-α and RBV could also act against thyroid cells by inducing a direct toxic effect [ 27, 31]. While it is not clear which factors contribute to the susceptibility to IIT, recent evidence suggests that genetic factors, gender, and hepatitis C virus infection may play a
role [31]. However, viral genotype and therapeutic regimen do not influence susceptibility to IIT [32].
Another way of treatment:
Recently, new antiviral drugs have been developed. These medicines, called direct antiviral agents (DAA) are much more effective, safer and better-tolerated than the older therapies. Therapy with DAAs can cure most persons with HCV infection and treatment is shorter (usually 12 weeks) and safer. Although the production cost of DAAs is low, these medicines remain very expensive in many high- and middle-income countries. Prices have dropped dramatically in some countries (primarily low-income) due to the introduction of generic versions of these medicines.
Hashimoto, Graves or autoimmune- which one is more common?
The commonest of autoimmune IIT is Hashimoto's thyroiditis (HT) and it is more likely in people who are positive to thyroid antibodies (TAbs) before starting the therapy with Peg-IFN-α based therapy [ 27]. However, development of HT could also occur in CHC patients and who are negative to TAbs during the course of therapy [31]. A less common manifestation of autoimmune IIT is Graves' disease (GD) [ 31, 34]. In a retrospective study, 321 patients diagnosed with hepatitis B or C and treated with IFN-α, 10 patients developed thyrotoxicosis, which was characterized by a completely de-creased TSH [30]. Six of those patients developed GD and all of them had symptomatic thyrotoxicosis, which failed to resolve even after IFN-α cessation [34].
GD and HT are both known of formation of thyroid- reactive T cells that infiltrate the thyroid gland [28, 36]. HT is characterized by Th1 switching of the thyroid infiltrating T cells, which induce apoptosis of thyroid follicular cells and clinical hypothyroidism. In GD, most of T cells undergoes a T helper (Th) 2 differentiation and activates B cells to produce antibodies against the thyroid stimulating hormone receptors, which are the hallmark of GD, and eventually they will cause clinical hyperthyroidism as a result of thyroid stimulation [37]. Indeed, IFN-α therapy in patients with hepatitis C has been strongly associated with both GD and HT, as well as the production of thyroid antibodies without clinical disease [28, 38].
Several studies have shown that the treatment of hepatitis C with IFN can induce the production of Tabs de novo, or cause a significant increase in TAbs levels in individuals who were positive for TAbs prior to interferon therapy [31]. The incidence of de novo development of thyroid autoantibodies secondary to IFN therapy varied widely in different studies from 1.9% to 40% [ 27]. The wide variations in the reported incidence of TAbs in interferon treated patients could be related to the used detection assays and different cut-off values applied in the different studies [39].
However, up to 50% of patients who develop thyroid abnormalities during IFN-α therapy do not develop autoantibodies, which suggests that thyroid dysfunction may be caused by a direct effect on thyroid cells [40]. A previous in vitro study reported that TSH-induced gene expression of thyroglobulin was inhibited following the culture of human thyroid follicular cells with interferon type I.
Destructive thyroiditis is a self-limited inflammatory disorder is another form of thyroid abnormality associated with Peg-IFN-α based therapy during the treatment of CHC. This disorder consists of three phases:
hyperthyroidism, followed by hypothyroidism phase, and finally normalization of thyroid function and usually it takes weeks to months to resolve [25, 31, 40].
Subacute thyroiditis due to IFN therapy for hepatitis C infection is usually benign. In addition, a subset of these patients may progress to permanent hypothyroidism, usually accompanied by the development of TAbs suggesting an underlying autoimmune thyroiditis [28]. Alternatively, the hypothyroidism may be due to a direct toxic effect of IFN on the thyroid. Clinical and subclinical hypothyroidism without TAbs during IFN therapy have been described and in many of these cases thyroid insufficiency is transient but permanent hypothyroidism is likely to develop if patients were positive for thyroid antibodies [41].
Thyroid cancer as a complication?
Montella et al. have carried out a case- controlled study on the different oncological pathologies. Among 495 patients with HCV 130 had developed thyroid cancer, with association: OR = 2.8, 95% CI 1.2–6.3, P = 0.01.[5] Other studies have confirmed an association between AT and thyroid cancer. Accordingly, features of AT were observed more frequently in HCV patients than in controls suggesting that AT may be a predisposing condition for thyroid cancer. Since about 15–30% of HCV patients may show an aggressive disease, for example, lung metastases, difficult to treat, the finding of an increased prevalence of thyroid cancer in these patients is clinically relevant [5].
Antonelli et al. studied the prevalence of thyroid cancer in 308 unselected HCV+ patients in comparison to two population-based, gender- and age-matched control groups: 1) 616 subjects from an iodine deficient area; 2) 616 subjects from an iodine-sufficient area. Thyroid status was assessed by measurement of circulating thyroid hormones and autoantibodies, thyroid ultrasonography, and when indicated, fine-needle aspiration cytology.
Circulating thyrotropin, anti-thyroglobulin, and anti- thyroperoxidase antibodies levels, and the prevalence of hypothyroidism were significantly higher in HCV+ patients (p < 0.001 for all). Six patients with papillary thyroid cancer were detected among HCV+ patients, whereas no case was observed in control 1 (p = 0.001), and only one case was observed in control 2 (p = 0.003). In HCV+ patients 83% with thyroid cancer had evidence of thyroid autoimmunity vs 31% of the other HCV+ patients (p = 0.02).[9]
These data suggest a high prevalence of thyroid papillary cancer in HCV+ patients, overall in presence of thyroid autoimmunity; careful thyroid monitoring is indicated during the follow-up of these patients [9].
Conclusion.
- 1.There is a proved link between thyroid gland's autoimmune pathologies, i.e. hypo- and hyperthyroidism, and viral hepatitis C.
- 2.Interferon treatment also influences on the development of autoimmune disorders in thyroid gland.
- 3.Other remedies (Direct Antiviral Agents) are safer, with less side effects, but still are very expensive and a large proportion of patients may not have access to them.
- 4.Another findings suggest that vitamin D supplementation could have a potential role in improving the success rate of Peg-IFN-α during the treatment of CHC merit further research especially that it is widely available and inexpensive, and it could provide an alternative option to treat those patients who have limited financial support and/or access to the new
- 5.Monitoring of thyroid gland's hormones, including antiviral treatment. antibodies, must be done before, during and after 6 months of IFN therapy, to find any abnormalities timely.
REFERENCES
- Averhoff FM, Glass N and Holtzman D. Global burden of hepatitis C: considerations for healthcare providers in the United States. Clin Infect Dis 2012; 55 Suppl 1: S10-15.
- Bassem Refaat, Adel Galal El-Shemi, Ahmed Ashshi1, Esam Azhar . Vitamin D and chronic hepatitis C: effects on success rate and prevention of side effects associated with pegylated interferon-α and ribavirin. Int J Clin Exp Med 2015;8(7):10284-10303 www.ijcem.com/ISSN:1940-5901/IJCEM0007955
- Wilmar M. Wirsinga Clinical Relevance of Environmental Factors in the Pathogenesis of Autoimmune Thyroid Disease www.e-enm.org 213Endocrinol Metab 2016;31:2
- Poupak Fallahi, Silvia Martina Ferrari Autoimmune and Neoplastic Thyroid Diseases Associated with Hepatitis C Chronic Infection. International Journal of Endocrinology Volume 2014 (2014), Article ID 935131, 9 pages http://dx.doi.org/10.1155/2014/935131
- Montella M., A. Crispo, L. Pezzullo, et al., “Is hepatitis C virus infection associated with thyroid cancer? A case-control study,” International Journal of Cancer, vol. 87, no. 4, pp. 611-612, 2000
- Alexopoulou A and Papatheodoridis GV. Current progress in the treatment of chronic hepatitis C. World J Gastroenterol 2012; 18: 6060-6069.
- Gatselis NK, Zachou K, Saitis A, Samara M and Dalekos GN. Individualization of chronic hepatitis C treatment according to the host characteristics. World J Gastroenterol 2014; 20: 2839-2853.
- Tran TT. A review of standard and newer treatment strategies in hepatitis C. Am J Manag Care 2012; 18: S340-349.
- Antonelli A, Ferri C Thyroid cancer in HCV-related chronic hepatitis patients: a case-control study. Online libertpub
- Bizzaro G and Shoenfeld Y. Vitamin D and thyroid autoimmune diseases: the known and the obscure. Immunol Res 2014; 61: 107-9.
- Blackard JT, Kong L, Huber AK and Tomer Y. Hepatitis C virus infection of a thyroid cell line: implications for pathogenesis of hepatitis C virus and thyroiditis. Thyroid 2013; 23: 863-870.
- Menconi F, Hasham A and Tomer Y. Environmental triggers of thyroiditis: hepatitis C and interferon-alpha. J Endocrinol Invest 2011; 34: 78-84.
- Chandrasekharan N.K. Frank H.frequency of thyroid dysfunction during Interferon alpha treatment of single and combination therapy in HCV patients: a systemic review based analysis.
- Mao XR, Zhang LT, Chen H, Xiao P and Zhang YC. Possible factors affecting thyroid dysfunction in hepatitis C virus-infected untreated patients. Exp Ther Med 2014; 8: 133-140.
- Ashshi AM, El-Shemi AG, AlZanbagi A and Refaat B. Prevalence of thyroid disorders and the correlation of thyroid profile with liver enzymes, serum activin-A and follistatin during the treatment of patients with chronic hepatitis C genotype 1 and 4. J Clin Exp Invest www. jceionline. org Vol 2014; 5.
- Andrade LJ, Atta AM, D'Almeida Junior A, Paraná R. Thyroid dysfunction in hepatitis C individuals treated with interferonalpha and ribavirin-a review. Braz J Infect Dis 2008; 12:144- 148
- Barut S, Gunal O, Erkorkmaz U, Yildiz F. Thyroid dysfunction in Turkish patients with chronic hepatitis C receiving peginterferon plus ribavirin in the period of 2005-2010. Braz J Infect Dis. 2012; 16:448-451.
- Tran HA, Reeves GE, Jones TL. The natural history of interferon-alpha2b-induced thyroiditis and its exclusivity in a cohort of patients with chronic hepatitis C infection. QJM 2009;102:117-122.
- Sünbül M, Şahbat E, Akkuş M, Esen Ş, Kahraman H, Leblebicioğlu H. Evaluation of thyroid functions during interferonalpha treatment of patients with chronic viral hepatitis. Viral Hepatit Dergisi 2004;9:67-71. (In Turkish)
- Tas A, Koklu S, Beyazit Y, Kurt M, Sayilir A, Yesil Y and Celik H. Thyroid hormone levels predict mortality in intensive care patients with cirrhosis. Am J Med Sci 2012; 344: 175-179.
- Andrade LJ, Atta AM, Atta ML, Mangabeira CN and Parana R. Thyroid disorders in patients with chronic hepatitis C using interferon-alpha and ribavirin therapy. Braz J Infect Dis 2011; 15: 377-381.
- Carella C, Mazziotti G, Amato G, Braverman LE and Roti E. Clinical review 169: Interferon-alpha-related thyroid disease: pathophysiological, epidemiological, and clinical aspects. J Clin Endocrinol Metab 2004; 89: 3656-3661.
- Vezali E, Elefsiniotis I, Mihas C, Konstantinou E and Saroglou G. Thyroid dysfunction in patients with chronic hepatitis C: virus-or therapy-related? J Gastroenterol Hepatol 2009; 24: 1024-1029.
- Tomer Y. Genetic susceptibility to autoimmune thyroid disease: past, present, and future. Thyroid 2010; 20: 715-725.
- Soppi E. [Concurrent subacute thyroiditis and Graves disease]. Duodecim 2012; 128: 1808-1810.
- Costelloe SJ, Wassef N, Schulz J, Vaghijiani T, Morris C, Whiting S, Thomas M, Dusheiko G, Jacobs M and Vanderpump MP. Thyroid dysfunction in a UK hepatitis C population treated with interferon-alpha and ribavirin combination therapy. Clin Endocrinol (Oxf) 2010; 73: 249-256.
- Andrade LJ, D'Oliveira A Jr, Silva CA, Nunes P, Franca LS, Malta AM and Parana R. A meta-analysis of patients with chronic hepatitis C treated with interferon-alpha to determine the risk of autoimmune thyroiditis. Acta Gast-roenterol Latinoam 2011; 41: 104-110.
- Mandac JC, Chaudhry S, Sherman KE and Tomer Y. The clinical and physiological spectrum of interferon-alpha induced thyroiditis: toward a new classification. Hepatology 2006; 43: 661-672.
- Zantut-Wittmann DE, Pavan MH, Pavin EJ and Goncales FL Jr. Central hypothyroidism in patients with chronic hepatitis C and relation with interferon-alpha treatment. Endocr Regul 2011; 45: 157-161.
- Nadeem A and Aslam M. Association of interferon-alpha and ribavirin-induced thyroid dysfunction with severity of disease and response to treatment in pakistani asian patients of chronic hepatitis C. Hepat Res Treat 2012; 2012: 864315.
- Yan Z, Fan K, Fan Y, Wang X, Mao Q, Deng G and Wang Y. Thyroid dysfunction in chinese patients with chronic hepatitis C treated with interferon alpha: incidence, long-term outcome and predictive factors. Hepat Mon 2012; 12: e6390.
- Tomer Y, Sarapura V and Kahaly GJ. Thyroid disorders: it's very personal. Thyroid 2010; 20: 677-679.
- Song RH, Yu ZY, Qin Q, Wang X, Muhali FS, Shi LF, Jiang WJ, Xiao L, Li DF and Zhang JA. Different levels of circulating Th22 cell and its related molecules in Graves' disease and Hashimoto's thyroiditis. Int J Clin Exp Pathol 2014; 7: 4024-4031.
- Oppenheim Y, Ban Y and Tomer Y. Interferon induced Autoimmune Thyroid Disease (AITD): a model for human autoimmunity. Autoimmun Rev 2004; 3: 388-393.
- Giovanella L, Toffalori E, Tozzoli R, Caputo M, Ceriani L and Verburg FA. Multiplexed immunoassay of thyroglobulin autoantibodies in patients with differentiated thyroid carcinoma. Head Neck 2012; 34: 1369-1371.
- Jadali Z. Autoimmune thyroid disorders in hepatitis C virus infection: Effect of interferon therapy. Indian J Endocrinol Metab 2013; 17: 69-75.
- Joffe RT, Pearce EN, Hennessey JV, Ryan JJ and Stern RA. Subclinical hypothyroidism, mood, and cognition in older adults: a review. Int J Geriatr Psychiatry 2013; 28: 111-118.
- Schoindre Y, Terrier B, Kahn JE, Saadoun D, Souberbielle JC, Benveniste O, Amoura Z, Piette JC, Cacoub P and Costedoat- Chalumeau N. [Vitamin D and autoimmunity. Second part: Clinical aspects]. Rev Med Interne 2012; 33: 87-93.
- Schoindre Y, Terrier B, Kahn JE, Saadoun D, Souberbielle JC, Benveniste O, Amoura Z, Piette JC, Cacoub P and Costedoat- Chalumeau N. [Vitamin D and autoimmunity. First part: Fundamental aspects]. Rev Med Interne 2012; 33: 80-86.
- Aljohani NJ, Al-Daghri NM, Al-Attas OS, Alokail MS, Alkhrafy KM, Al-Othman A, Yakout S, Alkabba AF, Al-Ghamdi AS, Almalki M, Buhary BM and Sabico S. Differences and associations of metabolic and vitamin D status among patients with and without sub-clinical hypothyroid dysfunction. BMC Endocr Disord 2013; 13: 31.
- Zhang Q, Wang Z, Sun M, Cao M, Zhu Z, Fu Q, Gao Y, Mao J, Li Y, Shi Y, Yang F, Zheng S, Tang W, Duan Y, Huang X, He W and Yang T. Association of high vitamin d status with low circulating thyroid-stimulating hormone independent of thyroid hormone levels in middle-aged and elderly males. Int J Endocrinol 2014; 2014: 631819.