Spirometric parameters (FVC and FEV1) are influenced by several factors and generally valid prediction equations have been never defined in particular for healthy children and adolescents. Spirometric data in urban-rural children and teens from Kazakhstan are reported in this study. Experimental FVC and FEV1 were predicted taking into account of height, age, ethnic group (Kazakh, Russian), living environment (urban, rural) and several other anthropometric parameters. The sample was constituted by 1,926 males and 1,967 females aged 7-18 years.
Introduction. Living environment slightly affected spirometric values. In spirometric prediction equations for both sexes and both FVC and FEV1 height explained almost all variance, but inspiratory chest circumference and age gave significant contribution to the model. Moreover, Russians had higher values of FVC/FEV1 than Kazakhs and also ethnic group was inserted in prediction equations. Finally, literature models that better predicted FVC/FEV1 of our population arose from developed countries, while models deriving from more specific populations had lower predictive power. Modernization had only a slight effect on lung function that was mainly predicted by anthropometric variables (height in particular), age and also ethnic group. Our model was in line with other models reported in literature.
On the basis of available data, several spirometric predictive equations have been extrapolated from healthy subjects to define a “normal range” of spirometric parameters and abnormal values have been related with different lung diseases at different severity degrees. In the general population, FVC and FEV1 have been mainly related with height and age, but also with weight and body mass index (BMI) and other BW components [1-3]. Moreover, sometimes additional anthropometric variables have been considered, but generally they have slightly improved the accuracy of prediction of the model used [4].
Spirometric values can be influenced by genetic factors and ethnic differences [5, 6] but also by environmental pollution, nutrition status, exercise and altitude, so a definitive and unique regression model valid for all populations has never been recognized. The definition of spirometric standard curves for a correct diagnosis of early lung diseases is of particular relevance for children and adolescents because they are particular sensible to environmental and lifestyle changes [7]. Moreover, the transition between childhood and adults implies non-linearity in the relationship between lung function and height, complicating the regression model [8, 9].
Here, we report spirometric data collected inside the project KHAN-ES (Kazakhstan Health and Nutrition Examination Survey) in the major former Soviet republic of Central Asia, Kazakhstan. Kazakhstan is undergoing a rapid modernization process and in this project the study of the differences between children and adolescents living in urban centres and in rural villages represents a great opportunity to relate anthropometric and clinical data with urbanization and modernization with consequent changes in the traditional lifestyle. Moreover, the multi-ethnicity of Kazakhstan can give the possibility to study the influence of ethnic group on spirometric values.
Aims of the study were:
- to test the relationship between FVC, FEV1 and different anthropometric data, living environment (urban-rural), age and ethnic group (Kazakh, Russian) and to finally find the multiple regression model that best describes the examined population.
- To compare our model with most recent models coming from different populations present in literature.
Materials and Methods
The measure of lung function in children and adolescents living in Kazakhstan was a part of the project KHAN-ES, male and female children of the two main ethnic groups of Kazakhstan, i.e. Kazakh and Russian, were studied between 2010 and 2012. They were aged 7 to 18 years and resided either in Almaty and Chilik. Almaty is the biggest city of Kazakhstan while Chilik is a rural village located at about 150 km NE from Almaty. We collected a convenience sample of about 50 children for every combination of gender (male vs. female), environment (Almaty vs. Chilik), ethnic group (Kazakh vs. Russian) and age (7-18), for a total of 4,808 children. Children without mental impairment or serious acute or chronic diseases and siblings to another child already enrolled in the study were recruited and measured at local schools. The study was conducted in conformity with the declaration of Helsinki and the protocol was approved by scientific committee of Kazakh Academy of Sciences. The parents of children and adults aged ≥ 18 years gave written informed consent to participate to the study.
Among 4,808 children and teenagers, only 3,893 (1,926 males and 1,967 females) were selected for this study.
The exclusion criteria were: smoker condition (at least 4 cigarettes/ week): 209 cases excluded; ex-smoker condition (stop smoking <12 months and at least 4 cigarettes/ week): 92 cases excluded; respiratory symptoms at the moment of the data collection: 181 cases excluded; use of medicines for respiratory diseases at the moment of data collection: 59 cases excluded; chronic respiratory or cardio-vascular diseases in the last 12 months: 65 cases excluded; lacking or incomplete data: 299 cases excluded; outliers for transcription errors: 10 cases excluded.
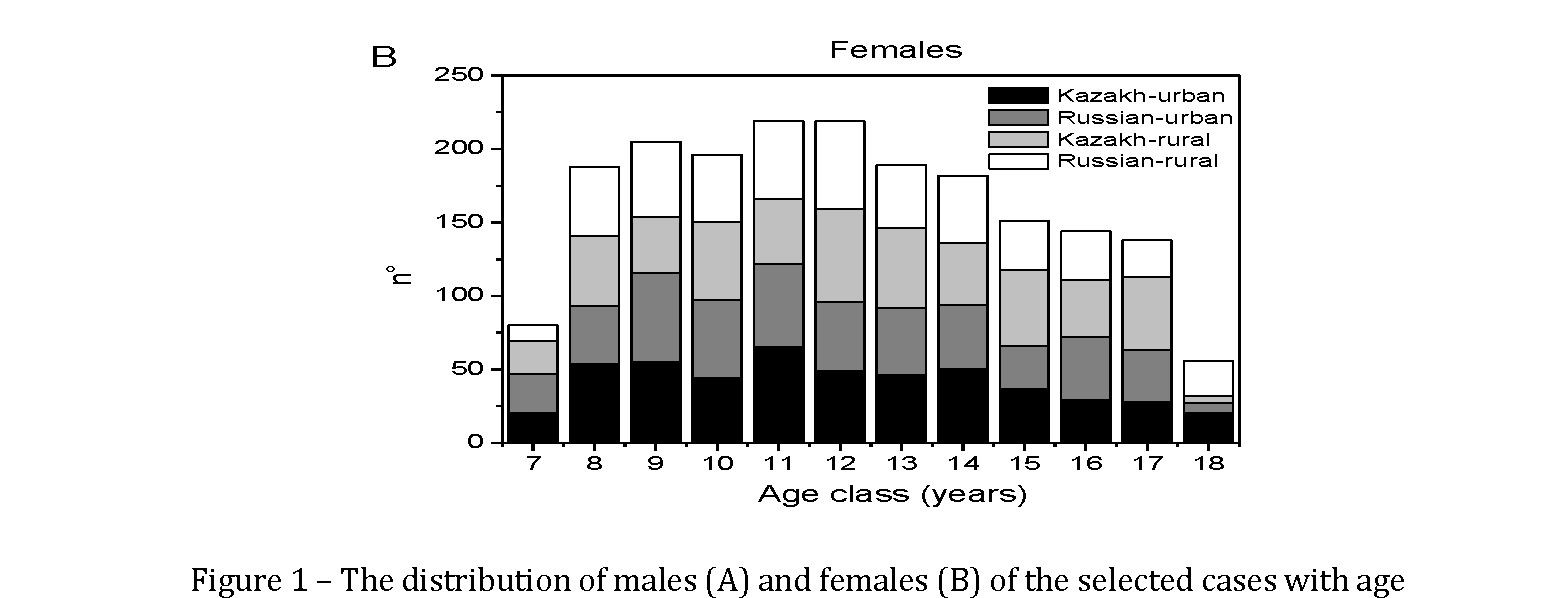
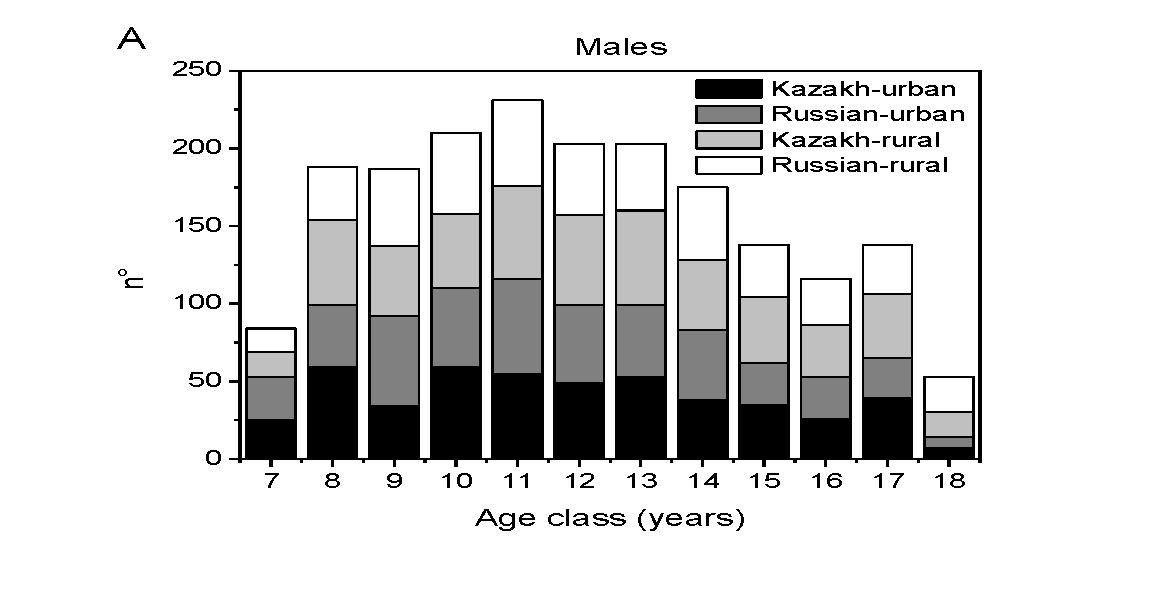 In figure 1a and 1b, the distribution of males and females of the selected cases with age is presented.
In figure 1a and 1b, the distribution of males and females of the selected cases with age is presented.
Spirometry was performed using a pneumotachograph (Koko Spirometer, Pulmonary Data Service, Louisville, CO) and FEV1 and FVC were measured. Moreover, also maximum midexpiratory flow (MMF25-75) was obtained from the same curve. Each child or adolescent, seated and with nose clip, performed three curves and the best curve (highest FVC+FEV1) was reported following the suggestions of American Thoracic Society and further published modifications proposed for children and adolescents [10,11].
Anthropometry. The following anthropometric data were measured: height with stadiometer without shoes; weight; sitting height and waist circumference; maximal inspiratory (ICC), expiratory (ECC) and normal (NCC) chest circumference measured at the nipple. Moreover, other chest parameters were measured: Chest breadth and depth. The following indirect indexes were calculated on the basis of anthropometric data:
- BMI = weight/height2
2. % Delta (percentage increase from ECC to ICC) = 100*(ICC- ECC)/ECC.
All the anthropometric variables were considered at the start of multiple regression model creation.
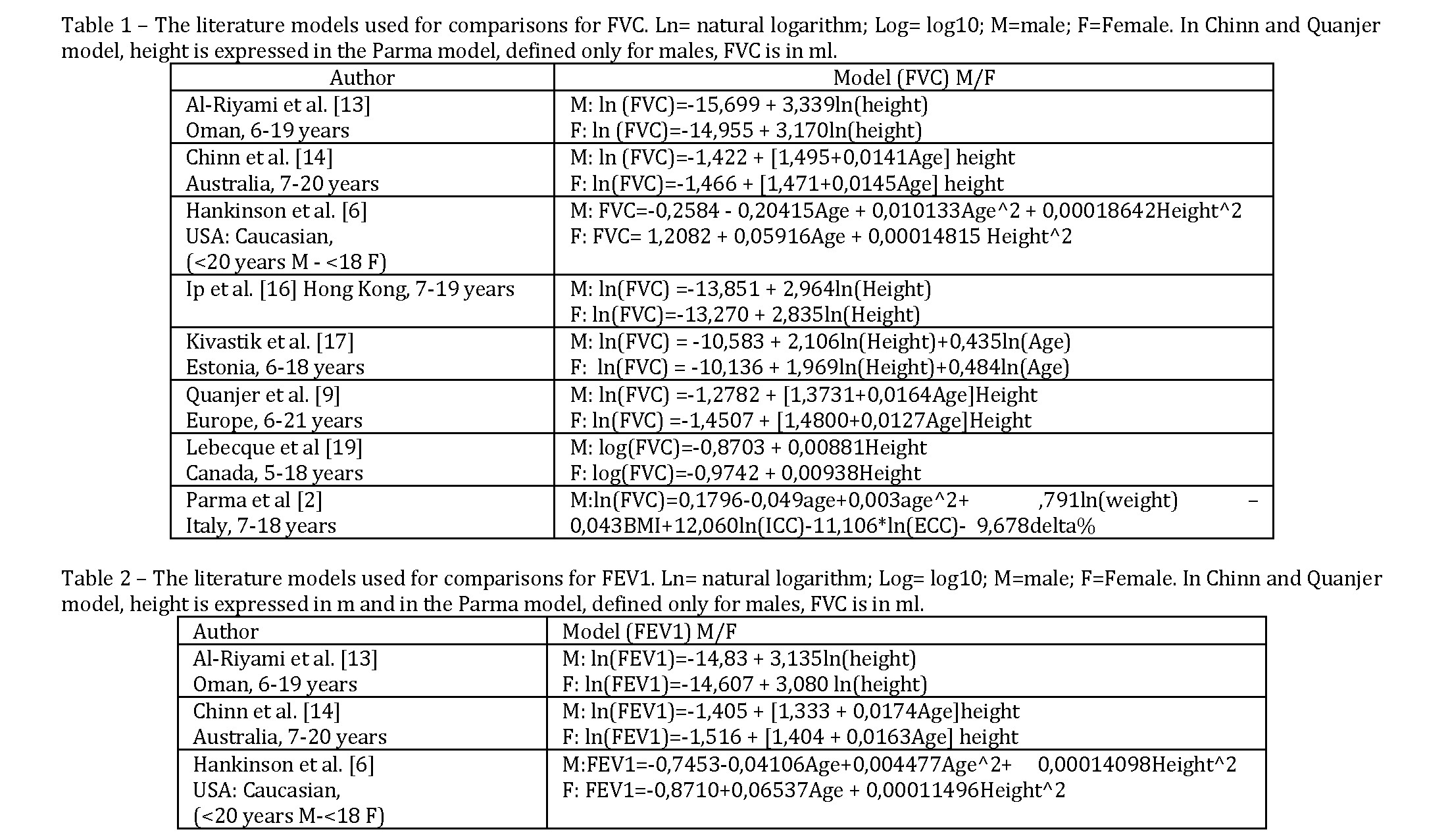
 Statistical Analysis. The statistical analysis was performed with SPSS 13.0 software (SPSS, USA). Prediction equations for spirometric parameters were preliminary developed using forward stepwise multiple regression and all the anthropometric variables, age (continuous variable in years), living environment (0=urban; 1=rural) and ethnic group (0=Kazakh; 1=Russian) were inserted in the analysis, distinguishing between males and females. Among all the possible transformations of variables, the log10 transformation of respiratory parameters using all the independent variables as linear seemed to be a good compromise, because no variation in the R square was observed transforming also independent variables (log10, polynomial, interaction in particular between height and age). Variables were included or excluded in the model not only on the basis of significance and the contribution to total R square, but also on the basis of multicollinearity looking at the values of VIF (Variance Inflation Factor). Only models with VIF values <10 for all independent variables were considered valid.
Statistical Analysis. The statistical analysis was performed with SPSS 13.0 software (SPSS, USA). Prediction equations for spirometric parameters were preliminary developed using forward stepwise multiple regression and all the anthropometric variables, age (continuous variable in years), living environment (0=urban; 1=rural) and ethnic group (0=Kazakh; 1=Russian) were inserted in the analysis, distinguishing between males and females. Among all the possible transformations of variables, the log10 transformation of respiratory parameters using all the independent variables as linear seemed to be a good compromise, because no variation in the R square was observed transforming also independent variables (log10, polynomial, interaction in particular between height and age). Variables were included or excluded in the model not only on the basis of significance and the contribution to total R square, but also on the basis of multicollinearity looking at the values of VIF (Variance Inflation Factor). Only models with VIF values <10 for all independent variables were considered valid.
Our best fit model was compared with several models proposed in literature (see tables 1 and 2) looking at median value of %predicted for FVC and FEV1 and the difference between 5th and 95th percentiles. Moreover, we propose Bland-Altman graph [12] on logarithms to more deeply investigate the deviations from experimental model.
|
Ip et al. [16] Hong Kong, 7-19 years |
M: ln(FEV1) =-13,999 + 2,972ln(Height) F: ln(FEV1) =-13,392 + 2,843ln(Height) |
|
Kivastik et al. [17] Estonia, 6-18 years |
M: ln(FEV1) = -11,554+2,371ln(Height)+0,234ln(Age) F: ln(FEV1) = -10,134+1,964ln(Height)+0,456ln(Age) |
|
Quanjer et al. [9] Europe, 6-21 years |
M: ln(FEV1) = -1,2933 + [1,2669 + 0,0174Age]Height F: ln(FEV1) = -1,5974 + [1,5016 + 0,0119Age]Height |
|
Lebecque et al [19] Canada, 5-18 years |
M: log(FEV1)=-0,8302+0,00825Height F: log(FEV1)=-9389+0,00890Height |
|
Parma et al [2] Italy, 7-18 years |
M:ln(FEV1)=2,448-,062age+0,003age^2+0,768ln(weight)- 0,044BMI+14,863ln(ICC)-14,044*ln(ECC)-12,440delta% |
Results. Definition of a model for FEV1 and FVC
Using the stepwise multiple regression model, the best fit model for logarithm of male FVC, with an overall R square of 0,824 contained the variables height (0,792), ICC (additional 0,019), ethnic group (additional 0,009) and age (additional 0,005) and all were highly significant (p<0,001).
The final equation of the model is: Log (FVC) = - 0,729 + 0,00429height+ 0,00526ICC + 0,0339ethnic + 0,00991age [1].
Among the excluded variables, the variable living environment didn`t contributed significantly to the R square value and among all variables it had the lowest significance. However, if added to the model, the contribution was significant as positive coefficient (p=0,002).
The best fit model for logarithm of female FVC, with an overall R square of 0,766 contained the variables height (0,735), ICC (additional 0,019), ethnic group (additional 0,007) and age (additional 0,005) and all were highly significant (p<0,001). Obviously, other anthropometric variables were correlated with FVC; however, they were excluded from the model because of multicollinearity in particular with height. The final equation of the model is: Log(FVC) = -0,710 + 0,00477height + 0,00407ICC + 0,0257ethnic + 0,00744age [2]. Among the excluded variables, the variable living environment did not considerably contribute to the R square value and among all variables, it had the lowest significance. However, if added to the model, the contribution was significant as positive coefficient (p=0,009). In other words, FVC of rural females was significantly but higher than urban ones.
The best fit model for logarithm of male FEV1, with an overall R square of 0,815 contained the variables height (0,788), ICC (additional 0,018), ethnic group (additional 0,004) and age (additional 0,005) and all were highly significant (p<0,001). Obviously, other anthropometric variables were correlated with FEV1; however, they were excluded from the model because of multicollinearity in particular with height. The final equation of the model is: Log (FEV1) = -0,782+ 0,00445 height + 0,00506ICC + 0,0253 ethnic + 0,00892 age [3]. Among the excluded variables, variable living environment (urban-rural) did not significantly contributed to the model (p=0,222).
The best fit model for logarithm of female FEV1, with an overall R square of 0,712 contained the variables height (0,688), ICC (additional 0,017), ethnic group (additional 0,003) and age (additional 0,004) and all were highly significant (p<0,001). Obviously, other anthropometric variables were correlated with FEV1. In other words the final equation of the model is: Log(FEV1) = -0,747 + 0,00496height + 0,00383ICC + 0,0190ethnic + 0,00622age [4]. Among the excluded variables, the variable living environment (urban-rural) did not significantly contribute significantly to the model (p=0,705).
Some considerations about MMF 25-75. The best fit model for logarithm of male MMF25-75 had very lower R square value than for FVC and FEV1 (0,546) and only height (0,528), ICC (additional 0,014) and age (additional 0,004) were contained in it. Neither ethnic group (p=0,216) nor living environment (p=0,466) significantly contributed to the model. For females, R square was still lower (0,396) and in addition to height (0,38), ICC (additional 0,009) and age (additional 0,003) also living environment (additional 0,004) was highly significant with a negative coefficient (p<0,001). In other words, MMF25-75 of urban females was significantly higher than rural ones. Ethnic group (p=0,14) did not significantly contribute to the model.
A comparison with literature Using the models presented in tables 1 and 2, % predicted values of FVC and FEV1 as 5th, 10th, 25th, 50th, 75th, 90th, 95th percentiles and the difference in % between 95th and 5th percentiles (% variability or ∆) are presented in table 3A-D. Moreover, we also calculated the difference between median and expected value if the model was perfect (100%). This variable is called “Dev50”. The model of Parma et al [2] (defined only for males) was used only for FEV1 and not FVC, because of a probable error in the intercept value (0,176, while we would expect a value between 1,5 and 2,5).
Table 3 – The difference in % between 95th and 5th percentiles: A) male FVC as % predicted; B) female FVC as % predicted; C) male FEV1 as % predicted; D) female FEV1 as % predicted using all the models presented in tables 1 and 2.
|
A |
||||||||||
|
MODEL |
5th % |
10th % |
25th % |
50th % |
75th % |
90th % |
95th % |
Dev50 |
∆ |
|
|
Our Model |
76,6 |
82,7 |
90,6 |
99,8 |
110,4 |
122,2 |
131,6 |
-0,2 |
55,0 |
|
|
Al Riyami |
78,6 |
83,6 |
93,3 |
104,8 |
118,6 |
133,3 |
143,2 |
+4,8 |
64,6 |
|
|
Chinn |
73,3 |
77,8 |
86,7 |
96,6 |
107,6 |
119,8 |
129,1 |
-3,4 |
55,8 |
|
|
Hankinson |
75,1 |
79,6 |
88,3 |
98,8 |
111,0 |
124,4 |
134,0 |
-1,2 |
58,9 |
|
|
ip |
81,8 |
86,2 |
96,0 |
107,2 |
120,1 |
134,3 |
144,6 |
+7,2 |
62,8 |
|
|
Kivastik |
76,7 |
81,7 |
91,1 |
101,7 |
113,7 |
126,7 |
137,3 |
+ 1,7 |
60,6 |
|
|
Quanjer |
72,8 |
77,5 |
86,4 |
96,2 |
106,9 |
119,0 |
127,8 |
-3,8 |
55,0 |
|
|
Lebeque |
77,1 |
81,5 |
91,2 |
101,4 |
113,2 |
127,2 |
136,6 |
+ 1,4 |
59,5 |
|
|
B |
||||||||||
|
Our |
76,1 |
82,1 |
90,9 |
100,3 |
110,5 |
122,2 |
130,0 |
+0,3 |
53,9 |
|
|
Al Riyami |
80,1 |
86,3 |
96,0 |
107,6 |
120,7 |
133,9 |
144,4 |
+7,6 |
64,3 |
|
|
Chinn |
72,5 |
77,9 |
86,0 |
95,9 |
106,7 |
118,9 |
127,6 |
-4,1 |
55,1 |
|
|
Hankinson |
72,4 |
77,2 |
85,7 |
95,9 |
107,3 |
118,9 |
127,2 |
-4,1 |
54,8 |
|
|
ip |
79,6 |
85,9 |
94,6 |
106,4 |
118,0 |
131,1 |
141,3 |
+6,4 |
61,7 |
|
|
Kivastik |
79,1 |
84,5 |
93,8 |
104,7 |
116,8 |
130,0 |
138,6 |
+4,7 |
59,5 |
|
|
Quanjer |
72,6 |
78,1 |
86,4 |
96,5 |
107,1 |
119,7 |
127,8 |
-3,5 |
55,2 |
|
|
Lebeque |
73,6 |
79,6 |
88,5 |
99,1 |
110,6 |
122,7 |
132,5 |
-0,9 |
58,9 |
|
|
C |
||||||||||
|
Our |
74,6 |
81,4 |
90,8 |
100,6 |
110,9 |
121,2 |
129,4 |
+0,6 |
54,8 |
|
|
Al Riyami |
80,0 |
87,2 |
97,4 |
108,9 |
122,0 |
135,6 |
147,3 |
+8,9 |
67,3 |
|
|
Chinn |
75,1 |
81,2 |
91,5 |
101,7 |
113,4 |
124,4 |
133,5 |
+ 1,7 |
58,4 |
|
For male FVC, some models had a A value similar to our model (Chinn, Quanjer), but slightly underestimated the median value (overestimated the median absolute value of FVC). For female FVC, several models had variability similar to experimental model (Chinn, Hankinson, Quanjer), while Lebeque model better estimate the median.
For male FEV1, the models of Parma and also Quanjer provided a good estimation of variability and median, quite near to A experimental model, while for female FEV1 the Quanjer model had a predictive power similar to experimental model.
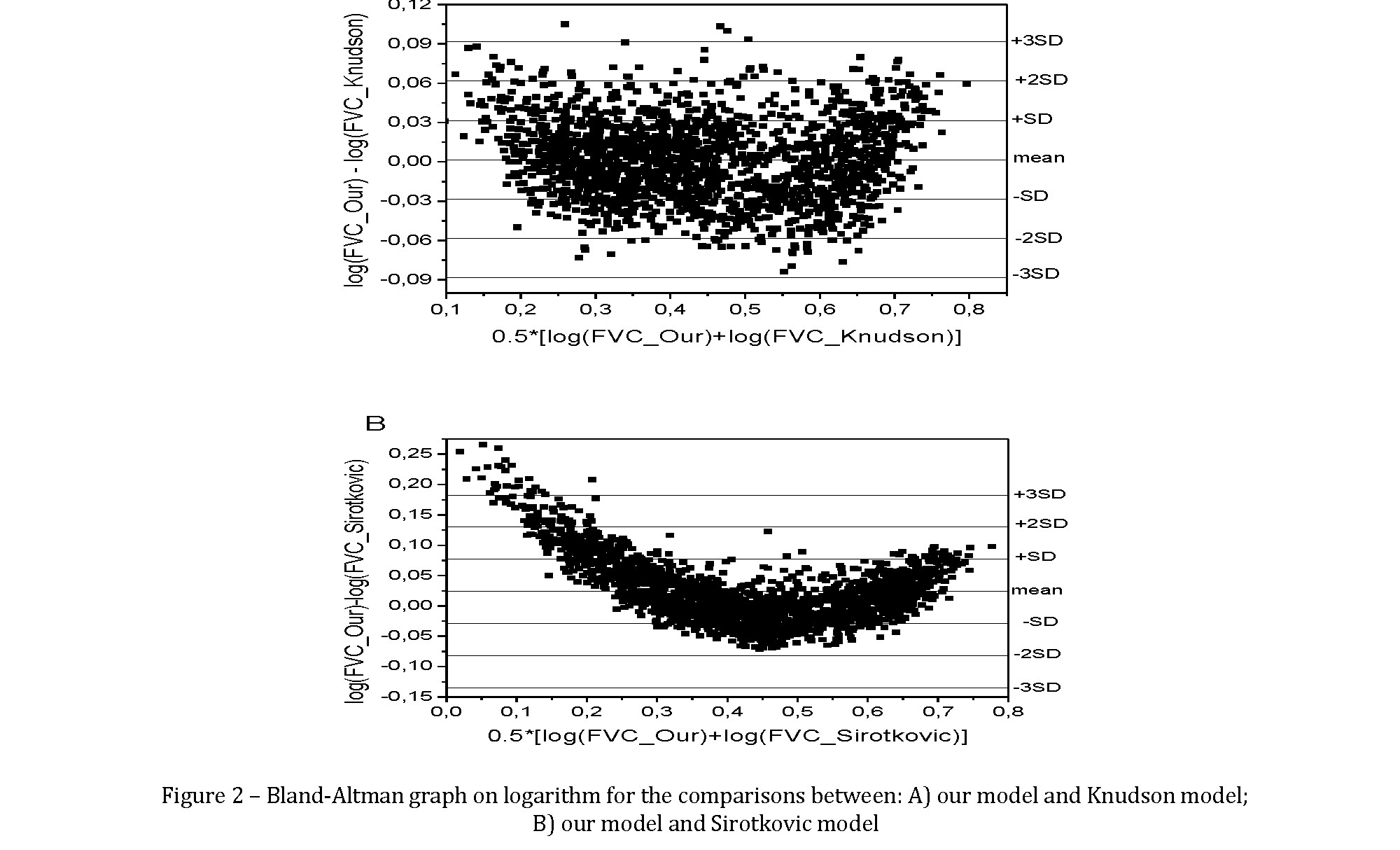
The Bland-Altman graph to compare different models as an example, in figures 2A and 2B the Bland-Altman graphs presenting the comparison between our model for male FVC and a good model, Knudson, and another model (Sirotkovic) that presented higher deviation.
Non-linearity of residual trend, only slightly present for the first comparison, was dramatically evident for the second comparison: Sirotkovic model tended to underestimate FVC values at lowest and highest FVC values, and the trend of residuals is absolutely not linear around the median value. Moreover, median and SD for deviations in the first comparison (0,0016Ã0,03) were clearly both much lower than the second comparison (0,024Ã0,05). Obviously, this type of comparison is possible for all presented models.
Discussion. This study is a part of KHAN-ES project, aimed at evaluating the contribution of living environment, ethnic group and gender to different anthropometric and clinical parameters in children and adolescent living in Kazakhstan, a rapidly modernizing ex-Soviet state of Central Asia.
Here, we concentrated the attention about the possible influencing factors of the most widely used spirometric parameters (FEV1 and FVC) in healthy children and adolescents, followed by a further comparison with mathematical predictive models present in literature.
It is known that exposure to air pollutants can affect the lung function during the major development period (10-18 years) and FEV1 is negative correlated with several environmental contaminants [20]. Moreover, air pollution in the residence area can be associated to the causal chain of reactions leading to retardation in pulmonary function growth during the preadolescence [21]. In fact, in our multiple regression models only in the case of MMF 25-75 for females we observed a modest contribution to the R square of the model. Nevertheless, FVC
|
Hankinson |
75,0 |
81,6 |
91,6 |
102,2 |
114,2 |
126,6 |
136,2 |
+ 2,2 |
61,2 |
|
ip |
78,7 |
86,0 |
95,8 |
107,1 |
119,8 |
132,1 |
143,1 |
+7,1 |
64,4 |
|
Kivastik |
77,7 |
84,5 |
94,4 |
104,8 |
117,1 |
128,7 |
138,7 |
+4,8 |
61,0 |
|
Quanjer |
73,9 |
80,1 |
90,3 |
100,2 |
112,1 |
122,5 |
132,5 |
+0,2 |
58,6 |
|
Lebeque |
73,8 |
79,5 |
89,8 |
99,9 |
111,8 |
123,7 |
132,4 |
-0,1 |
58,6 |
|
Parma |
73,1 |
80,3 |
90,1 |
100,2 |
111,3 |
122,5 |
130,6 |
+0,2 |
57,5 |
|
D |
|||||||||
|
Our |
71,6 |
79,1 |
90,6 |
101,6 |
111,9 |
123,0 |
132,0 |
+ 1,6 |
60,4 |
|
Al Riyami |
76,0 |
83,7 |
95,9 |
108,3 |
122,0 |
134,8 |
142,8 |
+8,3 |
66,8 |
|
Chinn |
69,1 |
76,5 |
87,6 |
98,0 |
108,9 |
120,4 |
129,1 |
-2,0 |
60,0 |
|
Hankinson |
68,5 |
76,6 |
86,9 |
98,2 |
109,3 |
121,1 |
128,2 |
-1,8 |
59,7 |
|
ip |
73,4 |
81,4 |
93,3 |
105,1 |
117,4 |
129,3 |
136,9 |
+5,1 |
63,5 |
|
Kivastik |
73,4 |
81,7 |
92,8 |
105,0 |
116,5 |
129,1 |
136,2 |
+5,0 |
62,8 |
|
Quanjer |
70,3 |
77,7 |
88,8 |
100,1 |
111,0 |
122,0 |
130,8 |
+0,1 |
60,5 |
|
Lebeque |
68,5 |
75,6 |
86,8 |
97,6 |
109,4 |
120,3 |
128,7 |
-2,4 |
60,2 |
tended to be slightly higher in rural than in urban environment for both sexes, while the trend was the opposite for MMF 25-75 only for females. On the other hand, living environment did not have a significant effect on FEV1 for both sexes and MMF 25-75 for males. Therefore, at this stage of analysis it is impossible to tract definitive conclusions about the relation between air pollution and reduction of pulmonary function in Kazakhstan. Our results are quite in line with what observed in Italy [22]. In particular, while in Nigeria no variation of spirometric parameters has been found in urban or rural communities [23], a significant reduction in pulmonary function was observed in 511 y urban children living in Iran as compared to rural counterpart [24]. So, a lot of factors could explain these discrepancies and even if a higher concentration of air pollutants is generally expected in urban environment or in any case in more industrialized areas [25], the environmental risk factors for airway diseases in rural communities in America have been well described [26] and in non-urban children risk factors for asthma are similar to risk factors in urban children [27].
Our linear models confirmed that most important variable to predict FVC and FEV1 is height but some anthropometric parameters, like ICC, can slightly improve the R square value of the model.
Our study also confirmed that age was highly significant, even if its contribution to R square was modest, but more importantly Russians of both sexes had higher values of FVC and FEV1 (not MMF 25-75) than Kazakhs, and this is the first work able to distinguish between children and adolescents of Mongolian and Caucasian origin. Another study about a general sample of Mongolian and Caucasian population with lower number of subjects of any age was unable to find it [28]. So, ethnic group could be added to general model of FVC/FEV1 to better predict lung function. MMF 25-75 prediction equations had lower values of R square, as already observed [2].
After the definition of a model to predict FVC/FEV1, a validation with most recent models present in literature was necessary (Table 1 and 2). First of all, a good qualitative method to comparison is to test the power of the model to predict the median % predicted FVC/FEV1 values (expected 100%) and the variability of the predicted values (e.g. the difference between 95th and 5th percentiles).
In conclusion, while urban and rural environment only slightly affected spirometric values (in particular FVC), spirometric prediction equations primarily depended by height (for both sexes and both FVC and FEV1) with a modest but significant contribution of inspiratory chest circumference and age. Moreover, Russians presented in general higher values of FVC/FEV1 than Kazakhs and ethnic group was inserted in prediction equations.
Finally, literature models that better predicted FVC/FEV1 of our population have been defined for whites in developed countries, while models deriving from more specific populations had lower performing power. Bland-Altman graph can be a good approach to compare two different prediction models.
REFERENCES
- Coultas B.D., Howard A.C., Skipper J.B., Samet M.J. Spirometric prediction equations for Hispanic children and adults in New Mexico. (1988) Am. Rev. Respir. Dis. 138: 1386-1392.
- Parma A., Magliocchetti N., Spagnolo A., Di Monaco A., Migliorino M.R., Menotti A. Spirometric prediction equations for male Italians 7-18 years of age. (1996) Eur. J. Epidemiol. 12: 263-277.
- Mohamed E.I., Maiolo C., Iacopino L., Pepe M., Di Daniele N., De Lorenzo A. The impact of body-weight components on forced spirometry in healthy Italians. (2002) Lung 180: 149-159.
- Marcus B.E., MacLean J.C., Curb D.J., Johnson R.L., Vollmer M.W., Buist S.A. Reference values for FEV-1° in Japanese-American men from 45 to 68 years of age. (1988) Am. Rev. Respir. Dis. 138: 1393-1397.
- American Thoracic Society. Lung function testing and selection of reference values and interpretive strategies. Official Statement of the American Thoracic Society. (1991) Am. Rev. Respir. Dis. 144: 1202-1218.
- Hankinson J.L., Odencrantz J.R., Fedan K.B. Spirometric reference values from a sample of the general U.S. population. (1999) Am. J. Respir. Crit. Care Med. 159: 179-187.
- Harik-Khan R.I., Muller D.C., Wise R.A. Racial difference in lung function in African-American and White children: effect of anthropometric, socioeconomic, nutritional and environmental factors. (2004) Am. J. Epidemiol. 160: 893-900.
- Knudson R.J., Lebowitz M.D., Holberg C.J., Burrows B. Changes in the normal maximal expiratory flow-volume curve with growth and aging. (1983) Am. Rev. Respir. Dis. 127: 725-734.
- Quanjer P.H., Borsboom G.J.J.M., Brunekreef B., Zach M., Forche G., Cotes J.E., Sanchis J. Paoletti P. Spirometric reference values for white European children and adolescents: Polgar revisited. (1995) Pediatr. Pulmonol. 19: 135-142.
- American Thoracic society. Medical section of the American lung association. Standardization of spirometry. 1994 update. (1995) Am. J. Respir. Crit. Care Med. 152: 1107-1136.
- Arets H.G.M., Brackel H.J.L., Van der Ent C.K. Forced expiratory manoeuvres in children: do they meet ATS and ERS criteria for spirometry? (2001) Eur. Respir. J. 18: 655-660.
- Bland J.M., Altman D.G. Statistical methods for assessing agreement between two methods of clinical measurement. (1986) Lancet 1: 307310.
- Al-Riyami B.M., Al-Rawas O.A., Hassan M.O. Normal spirometric reference values for Omani children and adolescents. (2004) Respirology 9: 387-391.
- Chinn D.J., Cotes J.E., Martin A.J. Modelling the lung function of Caucasians during adolescence as a basis for reference values. (2006) Ann. Hum. Biol. 33: 64-77.
- Golshan M., Nematbakhsh M., Amra B., Crapo R.O. Spirometric reference values in a large middle eastern population. (2003) Eur. Respir.J. 22: 529-534.
- Ip M.S.M., Karlberg E.M., Karlberg J.P.E., Luk K.D.K., Leong J.C.Y. Lung function reference values in Chinese children and adolescents in Hong Kong. I. Spirometric values and comparison with other populations. (2000) Am. J. Respir. Crit. Care Med. 162: 424-429.
- Kivastik J., Kingisepp P.H. Spirometric reference values in Estonian schoolchildren. (2001) Clin. Physiol. 4: 490-497.
- Sirotkovic M., Cvoriscec B. Spirometric reference values in school children from Dalmatia. (1995) Monaldi Arch. Chest Dis. 50: 258-263.
- Lebecque P., Desmond K., Swartebroeckx Y., Dubois P., Lulling J., Coates A. Measurement of respiratory system resistance by forced oscillation in normal children: a comparison with spirometric values. (1991) Pediatr. Pulmonol. 10: 117-122.
- Gauderman W.J., Avol E., Gilliland F., Vora H., Thomas D., Berhane K., et al. The effect of air pollution on lung development from 10 to 18 years of age. (2004) New Eng. J. Med. 351: 1057-1067.
- Jedrychowski W., Flak E., Mróz E. The Adverse Effect of Low Levels of Ambient Air Pollutants on Lung Function Growth in Preadolescent Children. (1999) Environ. Health Perspect. 107: 669-674.
- Centanni S., Di Marco F., Castagna F., Santus P., Guarnirei R., Allegra L. Atopy prevalence and spirometric performance in asymptomatic schoolchildren exposed to air pollution. (2001) Monaldi Arch. Chest Dis. 56: 304-308.
- Glew R.H., Kassam H., Vander Voort J., Agaba P.A., Harkins M., VanderJagt D.J. Comparison of pulmonary function between children living in rural and urban areas in northern Nigeria. (2004) J. Trop. Pediatr. 50: 209-216.
- Asgari M.M., DuBois A., Asgari M., Gent J. Beckett W.S. Association of ambient air quality with children's lung function in urban and rural Iran. (1998) Arch. Environ. Health 53: 222-230.
- Forastiere F., Corbo G.M., Pistelli P., Michelozzi P., Agabiti N., Brancato G., Ciappi G., Perucci C.A. Bronchial responsiveness in children living in areas with different air pollution levels. (1994) Arch. Environ. Health 49: 111-118.
- Schwartz D.A. Etiology and pathogenesis of airway disease in children and adults from rural communities. (1999) Environ. Health Perspect. 107 Suppl. 3: 393-401.
- Higgins P.S., Wakefield D., Cloutier M.M. Risk factors for asthma and asthma severity in nonurban children in Connecticut. (2005) Chest 128: 3846-3853.
- Crapo R.O., Jensen R.L., Oyunchimeg M., Tsh T., DuWayne Schmidt C. Differences in spirometry reference values: a statistical comparison of a Mongolian and a Caucasian study. (1999) Eur. Respir. J. 13: 606-609.