The anti-inflammatory activity for the promising according to PASS prediction 2-mercapto-4-oxo-3,4-dihydrothieno[2,3-d]pyrimidine-6- carboxylic acid derivatives was studied using carrageenan-induced rat paw edema. In order to prove the mechanism of pharmacological action the modeling of the studied compounds binding with the COX-1 and COX-2 using Autodock Vina program was performed. The computer calculations showed that all of the compounds do not interact effectively with both isoforms of cyclooxygenase. These calculations confirmed the absence of anti-cyclooxygenase mechanism of the tested compounds action. No influence of the synthesized compounds on the central nervous system (locomotor, exploring reactions and emotional reactivity) was ascertained using the open field test.
Introduction.
The derivatives of 2-mercapto-4-oxo-3,4-dihydrothieno[2,3- d]pyrimidine-6-carboxylic acid were reported as the ligands of serotonin receptors [1]; some of the similar compounds are known to be radioprotective and anti-cancer agents [2,3]. The compounds with such structural fragment were also reported as antimicrobials and HIF prolyl hydroxylase inhibitor, useful for treatment of anemia [5]. The series of the papers also describe some derivatives of the similar structure as the substances with non-steroidal anti-inflammatory effect [6,7]. Considering the promising pharmacological opportunities and the results of virtual PASS prediction for the 3-benzyl-2-thio-5-methyl-4-oxo-
- dihydrothieno[2,3-d]pyrimidine-6-carboxylic acid we have studied their biological activity, using suitable models similar to the predicted activities.
Materials and methods.
Chemical part.
In the course of our work we used the methods of organic synthesis, instrumental and chemical methods of organic compounds analysis (1Н NMR, elemental analysis). Meting points were measured using Kofler melting-point apparatus and were not corrected. Elemental analysis for N has been performed by Kjeldahl method. IR-spectra were recorded using FT-IR Bruker Tensor-27 spectrometer. 1H NMR spectra were recorded using Varian Mercury (200 MHz) spectrometer in DMSO-d6, using TMS as internal standard. Chemical shifts (δ) are reported in ppm. Diethyl 3-methyl-5- {[(methylthio)carbonothioyl]amino}thiophene-2,4- dicarboxylate 1 and еthyl 3-benzyl-5-methyl-4-oxo-2-thioxo-
- tetrahydrothieno[2,3-d]pyrimidine-6-carboxylate 2 were prepared using previously reported methods [8,9,10].
Ethyl 3-benzyl-5-methyl-4-oxo-2-thioxo-1,2,3,4- tetrahydrothieno[2,3-d]pyrimidine-6-carboxylate 2 [10] Yield 54%.
Mp. > 238-239°С.
1Н NMR (200 MHz, DMSO-d6) δ: 1,26 (3H, t), 2,66 (3H, s), 4,23 (2H, q), 5,54 (2H, s, CH2), 7,27 (5H, m), 13.86 (1H, br.s). Found, %: N 7,89. C17H16N2O3S2. Calculated, %: N 7,77. M.w. 360,46.
Ethyl 3-benzyl-5-methyl-2-[(4-methylbenzyl)thio]-4-oxo-
- dihydrothieno[2,3-d]pyrimidine-6-carboxylate 3
To the suspension of ethyl 3-benzyl-5-methyl-4-oxo-2-thioxo-
- tetrahydrothieno[2,3-d]pyrimidine-6-carboxylate 2 (1,5 g) in DMF 0,6 ml of triethylamine and 0,6 ml of 4-methylbenzyl chloride were added. Then the reaction mixture was heated at 80°C for 5-7 hours. After cooling water was added to the reaction mixture and the precipitate formed was filtered off and crystallized from 2-propanol-DMF mixture.
Yield 75 %.
Mp. > 248-249°С.
1Н NMR (200 MHz, DMSO-d6) δ: 1,27 (3H, t), 2,22 (3H, s), 2,79 (3H, s), 4,26 (2H, q), 4,41 (2H, s), 5,23 (2H, s), 7,2 (9H, m). Found, %: N 6,34. C25H24N2O3S2. Calculated, %: N 6,03. M.w. 464,61.
{[3-Benzyl-6-(ethoxycarbonyl)-5-methyl-4-oxo-3,4- dihydrothieno[2,3-d]pyrimidin-2-yl]thio}acetic acid 4
To the suspension of ethyl 3-benzyl-5-methyl-4-oxo-2-thioxo-
- tetrahydrothieno[2,3-d]pyrimidine-6-carboxylate 2 (1,5 g) in DMF 1,2 ml of triethylamine and 0,4 g of chloroacetic acid were added. The mixture was heated at 80°С for 5-7 hours. After the cool reaction mixture was diluted with water and acidified with acetic acid. The precipitate formed was filtered off and crystallized from 2-propanol.
Yield 61 %.
Mp. > 192-193°С.
1Н NMR (200 MHz, DMSO-d6) δ: 1,22 (3H, t), 2,85 (3H, s), 4,03 (2H, s), 4,32 (2H, q), 5,25 (2H, s), 7,30 (5H, m).
Found, %: N 6.85. C19H18N2O5S2. Calculated, %: N 6,69. M.w. 418,49.
3-Benzyl-5-methyl-4-oxo-2-thioxo-1,2,3,4- tetrahydrothieno[2,3-d]pyrimidine-6-carboxylic acid 5
Ethyl 3-benzyl-5-methyl-4-oxo-2-thioxo-1,2,3,4-
tetrahydrothieno[2,3-d]pyrimidine-6-carboxylate 2 (2 g) was dissolved in 7 ml of 10% sodium hydroxide solution and the mixture was stirred at 50°С for 10-12 hours. Then the cool reaction mixture was acidified with 1 ml of acetic acid. The precipitate formed was filtered off and washed with plenty of cold water.
Yield 92 %.
Mp. > 255-257 °С.
1Н NMR (200 MHz, ДМСО-d6) δ: 2,64 (3Н, s., СН3); 5,52 (2Н, s., СН2) 7,22-7,25 (5Н, m., Ar-H); 13,75 (2H, broad s., NH + COOH). IR (KBr): 3607, 3461, 3188, 3034, 2627, 1657, 1555, 1535, 1493, 1434, 1378, 1325, 1291, 1185, 1135, 1074 сm-1.
Found, %: N 8,69. C15H12N2O3S2. Calculated, %: N 8,43. M.w. 332,40.
Pharmacology.
Carrageenan-induced rat paw edema was induced by subplantar injection of 0,1 of 1% fresh solution of carrageenan in distilled water into the right-hind paws of each rat [11]. The white albino rats (180-220 g) were used in the experiment. Tablets «Ortofen» produced by the Pharmaceutical company ''Zdorovie'', Ltd. (Kharkiv, Ukraine, series 571112) were used as the reference drug (8 mg per kg dosage); such dose is equal ED50 for the antiexudative effect. The dosage of «Ortofen» was calculated by Yu.R.Rybolovlev's method [11], using the daily dosage in human. The screened compounds were administrated orally (dosage 10 mg per kg) in the form of suspension stabilized with Twin-80 (1 hour before carragenan injection). The animals from the positive control group were treated with the distilled water in the amount equivalent to their mass. Paw thickness was measured after 1, 2, 3, 4 and 5 hours, with the mechanical oncometer. Anti-inflammatory activity (AIA) activity as % was estimated by the ability of the tested compound to suppress the inflammatory reaction in experimental animals vs control ones. Calculation was performed according to the following formula:
|
ПЗА = |
Pc - Pe |
× 100 % |
|
Pc |
Рc - the differences between volumes of the edematous and normal feet in the control; Рe - the differences between volumes of the edematous and normal feet in the experiment.
Psychoactive properties of the tested compounds 2 and 5 were studied using the open field test using female albino rats (180-200 g). After randomization the negative control group animals were treated with distilled water in the amount equivalent to their mass. The experimental groups animals were orally treated with the tested compounds (dosages 3 mg per kg and 10 mg per kg). The test was performed after 40 minutes period. The following generally accepted behavioral acts were estimated: locomotor activity, exploring reactions and emotional reactivity. For the integral estimation of all of the behavioral reactions the sum of activities was calculated [11]. The psychoactive properties was evaluated by the changes in locomotor activity, exploring reactions and emotional reactivity of the experimental animals comparatively with the control group animals [11].
Computer modeling and docking studies were performed using the following computer programs: ISISDraw 2.3, Discovery studio Visualizer 4.0, Python molecule viewer and Autodock Vina. Docking was calculated for the flexible ligands and rigid models of proteins. Crystallographic data for COX-1 (PDB ID 1EQG) and COX-2 (PDB ID1CX2) proteins were obtained from the Protein Data Bank [12]. Water molecules and ligands were removed from the protein molecule along with the chains B, C and D. The calibration of the system was performed using the crystallographic data for Ibuprofen complex with COX-1 and results of docking calculations for Ibuprofen molecule model and COX-2.
Results and discussion.
Synthesis of the compounds, which were further used in pharmacological studies, was performed starting from diethyl 3- methyl-5-{[(methylthio)carbonothioyl]amino}thiophene-2,4- dicarboxylate 1 [8,9,10]. At the first step the compound 1 was cyclized with benzylamine in DMF producing ethyl 3-benzyl-5- methyl-4-oxo-2-thioxo-1,2,3,4-tetrahydrothieno[2,3- d]pyrimidine-6-carboxylate 2 [10]. Further modification of the compound 2 was performed either by alkylation of the sulfur atom in position 2 or by hydrolysis of the carbethoxy group of thieno[2,3-d]pyrimidine system. As the result of these transformations 2-S-alkyl derivatives 3 and 4 and the acid 5 were obtained (scheme).
According to the predicted activity and the previous study of the similar compounds of thieno[2,3-d]pyrimidine series [13,14] we have performed the screening of anti-inflammatory for the synthesized compounds 2-5 and the psychoactive properties for the compounds 2 and 5 [11].
The results of the anti-inflammatory activity screening listed in the table 1, evidently confirm that the activity of “Ortofen” tablets have higher anti-exudative effect than the tested compounds. Starting from the 1 hour till the end of the experiment “Ortofen” decreased the inflammation against the positive control, at the third hour of the experiment its anti-inflammatory activity was 77%; because this period of inflammation is characterized by the release of prostaglandins [11,15] it confirms the anticyclooxygenase mechanism of the reference drug. The screened compounds 4 and 5 (dosage 10 mg/kg) showed antiinflammatory activity only during the first hour of the experiment. After administration of these compounds the edema was evidently reduced against the control; their antiinflammatory activity was almost at the same level 65 and 61% respectively.
Administration of the compound 2 (dosage 10 mg/kg) evidently decreased the paw edema against the control pathology group after the first and third hours of the experiment (39% and 31%), but the activity of this compound was lower than the same of “Ortofen”.
The data obtained show that the highest anti-inflammatory activity for the tested compounds 4 and 5 is determined during the period, when inflammation mediators are biogenic amines and kinins (the first hour); the compound 2 displayed its activity in the periods of biogenic amines, kinins and prostaglandins (the first and the third hours) release. Analysis of the antiinflammatory dynamics for the screened compounds in the carrageenan-induced rat paw edema test showed that most of them (except compound 2) have no effect of the production of prostaglandins, which deny possibility of anti-cyclooxygenase mechanism of their action.
From the other side, the absence of the influence on the prostaglandin phase of inflammation decreases the ulcerogenic effect for the tested compounds, which is typical for most of COX inhibitors [15]. This may be considered as a valuable advantage of the tested compounds as potential non-steroidal antiinflammatory drugs.
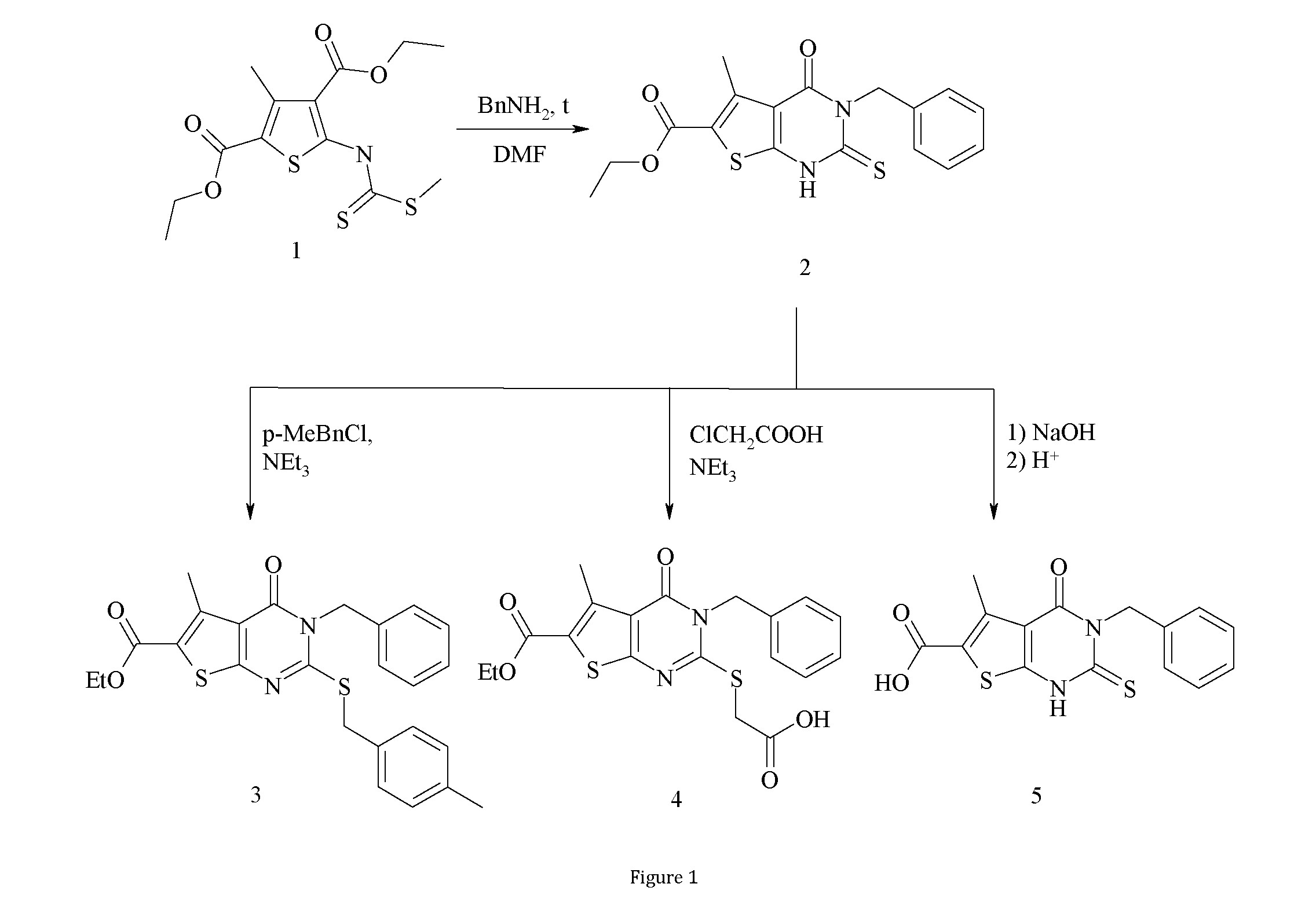
Table 1 - The results of anti-inflammatory screening study for the derivatives of 3-benzyl-2-thio-5-methyl-4-oxo-3,4-dihydrothieno[2,3- d]pyrimidine-6-carboxylic acid 2-5(carrageenan-induced rat paw edema, n=6)
|
Groups |
Change in paw edema, mm |
||||||||||
|
1 hour |
AIA,% |
2 hours |
AIA,% |
3 hours |
AIA, % |
4 hours |
AIA, % |
5 hours |
AIA, % |
||
|
Positive control |
20,00±2,92 |
- |
24,50±2,59 |
- |
25,00±1,77 |
- |
22,83±1,99 |
- |
24,83±2,10 |
- |
|
|
"Ortofen”, 8 mg/kg |
3,00±0,97* |
85 |
4,50±1,12* |
82 |
5,67±1,63* |
77 |
8,17±1,97* |
64 |
14,50±1,12* |
42 |
|
|
I Compounds ∣ |
2, 10 mg/kg |
12,17±1,20*/** |
39 |
18,67±3,49** |
24 |
17,17±4,00*/** |
31 |
17,83±3,75 |
22 |
19,33±3,14 |
22 |
|
3, 10 mg/kg |
14,00±2,21** |
30 |
24,20±2,82** |
1 |
25,80±3,26** |
-3 |
27,20±4,12** |
-19 |
27,60±3,09 |
-11 |
|
|
4, 10 mg/kg |
7,00±1,77* |
65 |
18,83±2,46** |
23 |
22,83±2,37** |
9 |
22,00±2,91 |
4 |
20,17±2,50 |
19 |
|
|
5, 10 mg/kg |
7,83±1,35*/** |
61 |
18,83±2,87** |
23 |
22,50±2,83** |
10 |
20,33±2,49 |
11 |
20,50±3,39 |
18 |
|
- * - statistically significant differences between the positive control, р<0,05;
- ** - statistically significant differences between the “Ortofen” treated group, р<0,05;
- n- number of animals in the group.
In order to investigate the results obtained using the experimental carrageenan-induced rat paw edema model and to analyze the possibility of their application in human therapy, the docking studies of the experimental compounds binding with the enzymes, known to be the typical targets for non-steroidal antiinflammatory drugs (NSAIDs) have been performed.
We have performed molecular docking studies by using in silico analysis of the compounds 2-5 with the active site of COX-1 and COX-2 isoforms of cyclooxygenase. The date known about the binding pattern between protein and the molecules of NSAIDs, suggests that most of the COX-inhibitors interact with the following monoacids: His-90, Arg-120, Tyr-355, Tyr-385, Arg- 513, Val-523 and Ser-530 [16,17,18,19]. For the selective COX-2 inhibitors binding with several regions is important such as hydrophobic one (Tyr-385, Trp-387, Phe-518, Ala-201, Tyr-248 and Leu352), hydrophilic (Arg-120, Glu-524, Tyr-355) and additional interaction with His-90, Arg-513, Val-523 fragment [16]. It is also known that Aspirin reversibly inhibits this enzyme by acetylation of Ser-580 [20]. The results of docking studies show that no any from the listed compounds effectively binds the active sites of the both cyclooxygenase isoforms (table 2). Probably the possible binding with one or two aminoacids of the active site is not enough to inhibit the enzyme. The calculations confirm the weak anti-inflammatory effect of the tested compounds and the absence of anti-cyclooxygenase mechanism of their action.
Table 2 - The results of docking studies of 3-benzyl-2-thio-5-methyl-4-oxo-3,4-dihydrothieno[2,3-d]pyrimidine-6-carboxylic acid derivatives
2-5 binding with COX-1 and COX-2*
|
Compound |
COX-1 |
COX-2 |
|
2 |
- |
Arg-120** |
|
3 |
- |
- |
|
His-90** |
Arg-120** |
|
|
4 |
Arg-120** Glu-524** |
|
|
5 |
- |
His-90** |
* + - ligand engagement to the active site; - - no ligand engagement to the active site; ** - amino acids of the active site interacting with the ligand.
Table 3 - The results of psychoactive properties screening for the derivatives of 3-benzyl-2-thio-5-methyl-4-oxo-3,4 dihydrothieno 2,3-ď| pyrimidine -6-carboxylic acid 2-5 Ме (Q25÷Q75)
|
Groups |
Behavioral acts |
|||||||
|
Line crossing |
Number of rearings |
Number of hole investigations |
Number of defecations |
Number of urinations |
Number of grooming |
Sum of activities |
||
|
Negative control |
16 (11÷18) |
4 (3÷5) |
8,5 (5÷12) |
3 (2÷4) |
0 (0÷1) |
0 (0÷0) |
33,5 (25÷36) |
|
|
Compund |
2, 3 mg/kg |
8 (2÷25) |
4 (0÷7) |
5,5 (5÷7) |
0,5 (0÷2) |
0,5 (0÷3) |
0 (0÷0) |
24 (6÷43) |
|
2, 10 mg/kg |
11 (8÷17) |
2,5 (2÷4) |
7,5 (5÷10) |
0 (0÷0)* |
2,5 (0÷4) |
0,5 (0÷1) |
29,5 (17÷31) |
|
|
5, 3 mg/kg |
15 (10÷18) |
4,5 (4÷8) |
7,5 (4÷8) |
3 (0÷5) |
1,5 (0÷2) |
0 (0÷2) |
33,5 (26÷37) |
|
|
5, 10 mg/kg |
25 (16÷35) |
4,5 (3÷8) |
7,5 (5÷10) |
0 (0÷1)* |
2,5 (0÷3) |
0,5 (0÷1) |
42 (35÷50) |
|
- n=6 - number of animals in the group;
- * - statistically significant differences between the negative control group, p≤0,05.
The PASS prediction also indicated the possible psychoactive properties for the compounds 2 and 5. One of the most popular and meaningful test to determine this type of activity is the open field test [11]. The results of screening as to the sum of activities compared with activities of negative control animals indicate the absence of the compounds 2 and 5 influence on the exploring reactions and emotional reactivity of the experimental animals (table 3). The increase in the dosage of the compound 2 up to 10 mg/kg evidently decreased only the number of defecations (emotional activity index).
Conclusions.
The results of the investigation performed, allowed to identify two the most promising compounds from the series of 3-benzyl- 2-thio-5-methyl-4-oxo-3,4-dihydrothieno[2,3-d]pyrimidine-6- carboxylic acid derivatives with anti-inflammatory activity. The mechanism of their action probably includes suppression of such inflammatory agents as kinins and biogenic amines release. The docking study of the compounds 2-5 binding with COX-1 and COX-2 active sites showed no effective interaction of the screened compounds with the both isoforms of the enzyme, which is the additional evidence for the absence of anticyclooxygenase mechanism of the tested compounds action. The results of the open field test screening for compounds 2 and 5 showed no influence on the central nervous system (locomotor, exploring reactions and emotional reactivity).
REFERENCES
- Modica M., Santagati M., Russo F., et al. High affinity and selectivity of [[(arylpiperazinyl)alkyl]thio]thieno[2,3-d]pyrimidinone derivatives for the 5-HT1A receptor. Synthesis and structure - affinity relationships // Eur. J. Med. Chem. — 2000. — Vol. 35, №7-8. — P. 677 - 689.
- Ghorab M.M., Osman A. N., Noaman E., Heiba H.I., Zaher N.H. The Synthesis of Some New Sulfur Heterocyclic Compounds as Potential Radioprotective and Anticancer Agents // Phosphorus, Sulfur and Silicon and the Related Elements. — 2006. — Vol. 181, № 8. — P. 1935 1950.
- Ghorab M.M., Osman A.N., Noaman E., Heiba H.I., Zaher N.H. The Utility of Isothiocyanato Thiophenes in the Synthesis of Thieno[2,3- d]pyrimidine Derivatives as Possible Radioprotective and Anticancer Agents // Phosphorus, Sulfur and Silicon and the Related Elements. 2006. — Vol. 181, № 9. — P. 1983 - 1996.
- Hussein H.A.R. A Facile and Efficient Synthesis of Polynuclear Heterocycles:Azolothienopyrimidines and Thienothiazolopyrimidines with Antimicrobial Activity // Phosphorus, Sulfur and Silicon and the Related Elements. — 2007. Vol. 182, № 9. — P. 2069 - 2085.
- Jin J., Schulz M.J., Wang Y. Pat. WO201022308. — 2010. — Заявл. 21.08.2008. Опубл. 25.02.2010.
- Hafez H. N., El-Gazzar A.-R. B.A., Nawwar G. A. M.. Synthesis, biological and medicinal significance of S-glycosido-thieno[2,3-d]- pyrimidines as new anti-inflammatory and analgesic agents // Eur. J. Med. Chem. — 2010. — Vol. 45, № 4. — P. 1485 - 1493.
- Basile L., Álvarez S., Blanco A., et al. Sulfonilamidothiopyrimidone and thiopyrimidone derivatives as selective COX-2 inhibitors: Synthesis, biological evaluation, and docking studies // Eur. J. Med. Chem. — 2012. — Vol. 57. — P. 149 - 161.
- Ivachtchenko A.V., Kovalenko S.M., Tkachenko O.V., Parkhomenko O.O. Synthesis of Substituted Thienopyrimidine-4-ones // J. Comb. Chem. — 2004. — Vol. 6, № 4. — Р. 573 - 583.
- Коваленко С.Н., С.В.Власов, А.И.Федосов, В.П.Черных. Синтез и превращения алкил 3-амино-5-метил-4-оксо-2-тиоксо-1,2,3,4- тетрагидротиено[2,3-d]пиримидин-6-карбоксилатов // Journal of organic and pharmaceutical chemistry.- 2007.- Т. 5, № 3.- С. 3440.
- Tkachenko O.V., Vlasov S.V., Kovalenko S.M., Zhuravel' I.O., Chernykh V.P. Synthesis and antimicrobial activity of ethyl 5-methyl-2- (alkylthio)-4-oxo-3,4-dihydrothieno[2,3-d]pyrimidine-6-carboxylates // Journal of organic and pharmaceutical chemistry. - 2013. - Т. 11, №3 (43). - С. 9-15.
- Доклiнiчнi дослiдження лiкарських засобiв. Методичнi рекомендаций/ За ред. О.В. Стефанова. — Киŕв. — 2001. - 528 с.
- http://www.rcsb.org/pdb
- Черних В.П., Коваленко С.М., Яковлева Л.В., и др. Пат. UA86561. — 2009.- Заявл. 20.10.08. Опубл 27.04.09.
- В.П. Черних, С.М. Коваленко, Л.В. Яковлева, и др. Пат. UA87088. — 2009.- Заявл. 20.10.08. Опубл. 10.06.09.
- Di Rosa M., Giround J.P., Williughby D.A. Studies on the mediators of the acute inflammatory response induced in rats in different sites by carrageenen and turpentine // J. Pathol. - 1971. - Vol. 104. - № 15. - P. 29.
- D'Mello P., Gadwal M. K., Joshi U., Shetgiri P. Modeling of COX-2 inhibitory activity of flavonoids // International Journal of Pharmacy and Pharmaceutical Sciences. — Vol 3. №4. — 2011. - P. 33-40.
- Price M.L.P., Jorgensen W.L. Rationale for the observed COX-2/COX-1 selectivity of celecoxib from Monte Carlo simulations // Bioorg. Med. Chem. Lett. — 2001. — Vol. 11, № 12. - P. 1541 - 1544.
- Kiefer J.R., Pawlitz J.L., Moreland K.T., et al. Structural insights into the stereochemistry of the cyclooxygenase reaction // Nature. —
- — Vol. 405, № 6782. —P. 97-101.
- Llorens O., Perez J., Palomer A., Mauleon D. Differential binding mode of diverse cyclooxygenase inhibitors // Journal of Molecular Graphics and Modelling. — 2002. — Vol. 20, № 5. - P. 359-371.
- Adinarayana K.P.S., Ashoka Reddy P., Ajay Babu P. Structural studies on docking selective COX-2 inhibitors // Journal of Bioinformatics and Research. — 2012. — Vol. 1, №1. - P. 21-26.