Objective: To test the efficacy of continuous subcutaneous insulin infusion (CSII) by measuring glycemic parameters during the first 12 months of a structured, collaborative Industry/Government/Health Care System program that introduced CSII therapy to children in Kazakhstan.
Method: Children 5-15 years of age were offered insulin pump therapy to treat their type 1 diabetes in all 16 regions of the country. Training included insulin pump operation, use of integrated blood glucose (BG) meters, and data uploading to CareLink (Medtronic Diabetes) for retrospective analysis. All study participants visited local pediatric endocrinologists on a quarterly basis for HbA1c determinations, height and weight measurements, CareLink report reviews, and therapy adjustments.
Results: As of March 31, 2014, 790 children had enrolled with per-site enrollments ranging from 11 to 135. Most children (N=442, 69.6%) had baseline HbA1c values ≥7.5. In a subgroup of 313 children who completed baseline and 12-month visits, the HbA1c range at 12 months was 4.3% to 14.0%. For those with baseline HbA1c ≥7.5% (N=221, 70.6%), the mean HbA1c value decreased by 0.85±3.07 percentage points. HbA1c decreased in 63% of patients with baseline HbA1c ≥7.5 after 12 months of CSII, and 23% of these patients had HbA1c values <7.5% at the 12-month visit.
Conclusion: Children adopting CSII in the context of government-industry collaborations, particularly those with poor glycemic control of diabetes, may realize significant glycemic benefits. The Kazakhstan/Medtronic collaboration provides a model for other initiatives that require rapid deployment and/or massive enrollment for introduction of CSII.
Background.
Type 1 diabetes mellitus (T1DM) is a disease usually diagnosed in children and young adults. Treatment always requires exogenous insulin administration due to immune-mediated destruction of insulin-producing cells in the pancreas. Worldwide, from 1990 to 2008, the incidence of T1DM has been increasing by 2.8%to 4.0% per year, similar to that observed in the United States and Europe [1, 2]. Despite all achievements in diabetes treatment in the past two decades, T1DM remains a serious cause of mortality and morbidity in youth [3-4]. The primary goals of treatment of T1DM in children and adolescents are maintenance of near-normoglycemia through intensive insulin therapy, avoidance of acute complications, and prevention of long-term microvascular and macrovascular complications, while facilitating as close to a normal life as possible [5].
The increasing use of insulin pump therapy as a viable alternative to multiple daily injections (MDI) over the last 15 years, particularly in children, is attributable to improvements in pump technology and motivated in part by the results of the Diabetes Control and Complications Trial (DCCT), which established the benefit of improved glycemic control [6, 7]. Ideally, CSII therapy more closely mimics physiological secretion of a healthy pancreas by combining 24-hour continuous adjustable precise “basal” delivery of insulin with prandial-related “boluses” in an effort to eliminate the symptoms and complications of hyperglycemia, while minimizing the risk of hypoglycemia. Several trials have demonstrated that, when compared to MDI, CSII offers better metabolic control as measured by HbA1c and blood glucose variability, fewer hypoglycemic episodes, and improvements in quality of life [8-11].
Many patients are motivated to consider CSII because of recurrent or severe hypoglycemia, the dawn phenomenon, or poor glycemic control. The possibility of more precisely delivering very small doses of insulin and needle phobia are especially important factors for the pediatric population [12, 13].
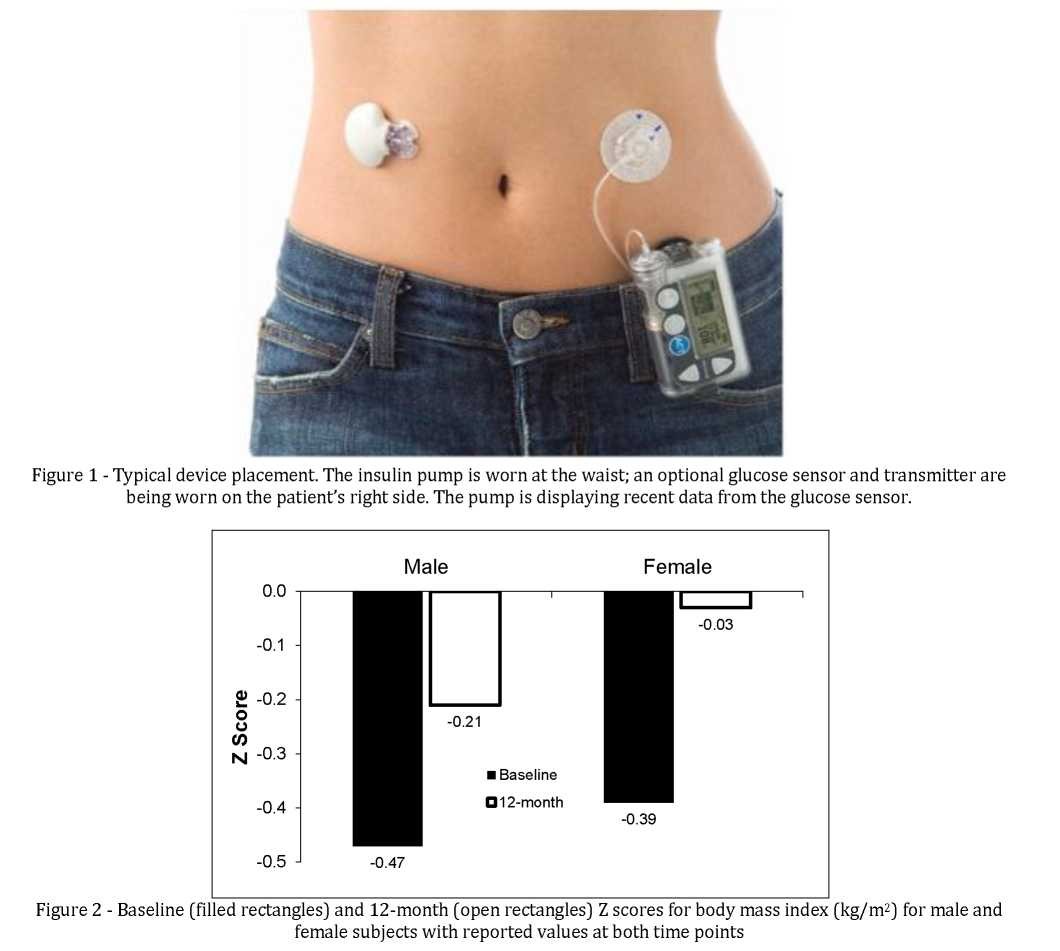
Figure 1 shows the insulin pump used in this study and the placement of an optional sensor for continuous glucose monitoring (CGM). The pump contains a reservoir of insulin which is connected to an infusion set. The tip of the cannula of the infusion set is placed in the subcutaneous space and replaced every two to three days. There are significant regional variations in CSII use. In the United States, about 40% of patients with type 1 diabetes use pumps. Pumps are used by >15% of type 1 diabetics in Norway, Austria, Germany, and Sweden, but by <5% of patients in Spain, the UK, Finland, and Portugal [14]. Less than 1% of patients Central and Eastern European countries use this therapy.
A collaborative project, Project Baiterek, between Medtronic Diabetes, the Kazakhstan Ministry of Health, and 16 regional pediatric diabetes clinics began in January, 2012 with the goal of improving the glycemic control and quality of life of children with diabetes by introducing them to CSII therapy [15, 16].To date, Project Baiterek has enrolled over 790 children ages 5-15. Here we present interim results of the study, which is ongoing.
Material and Methods.
This is an observational study with retrospective data analysis. Consent for data to be entered into the registry was obtained from all parents or guardians, and data collection was approved by local ethics committees. Paradigm® Veo insulin pumps (Medtronic MiniMed, Inc.), consumables, rapid acting insulin analogs, blood glucose (BG) meters, and a monthly supply of 30 BG test strips were funded by Ministry of Health.
Pediatric endocrinologists in each region were identified and attended multiple training sessions supported by Medtronic. Initial two-day long pump therapy training was conducted by two internationally recognized pediatric diabetes experts in Kazakhstan. Advanced training in clinical settings was provided in training centers in Israel, Slovenia, Russia, and USA. Multiple follow-up trainings by industry physicians and educators were conducted in Kazakhstan as well as during international congresses in different locations worldwide. All educational materials for physicians and patients were provided in Russian.
Baseline HbA1c measurements were obtained using various methods in local laboratories; follow-up HbA1c measurements were done on the fully automated, CLIA- waived In2it analyzer (Bio-Rad), calibrated to DCCT- equivalent numbers. All children were to have HbA1c determinations during clinic visits every 3 months; HbA1c values closest to each specified time-point were used for analysis. Patients' demographic characteristics (age, gender, race, diabetes duration, and age of diagnosis) were obtained on the initial visit; height and weight were measured at each clinic visit to calculate body mass index (BMI), in kg/m2. Z scores, which represent the number of standard deviations that observed values deviate from a population mean value, were calculated based on age- and gender-specific BMI-for-age data from the Centers for Disease Control. Pump and blood glucose data were uploaded to CareLink (Medtronic) for retrospective analysis. Data are presented as mean ± SD.
Results.
As of March 31, 2014 790 children had enrolled with persite enrollments ranging from 11 to 135.
Figure 2 shows that at enrollment, the mean Z scores for BMI were negative at baseline and at 12 months for both male and female subjects, indicating underweight compared to age-matched controls. Although the mean Z scores at 12 months were still negative, the magnitude of the weight deficit was less than at baseline, suggesting that many children were “catching up” to their peers.
Most children (N=442, 69.6%) had baseline HbA1c values ≥7.5. In a subgroup of 313 children who had completed baseline and 12-month visits, the HbA1c range at 12 months was 4.3% to 14.0% (mean 9.04%±2.12; median 8.5%). For those with baseline HbA1c ≥7.5% (N=221, 70.6%), the mean HbA1c decreased by 0.85±3.07 percentage points. HbA1c decreased in 63% of patients with baseline HbA1c ≥7.5 after 12 months of CSII, and 23% of these patients realized HbA1c reductions to less than 7.5%.
Mean daily BG values (averaged over 4 weeks) for this group fell from 12.28±3.99 to 10.45±2.47 mmol/L, a mean 1.83±4.52 (14.9%) mmol/L decrease. HbA1c reduction after 12 months of CSII was more significant in children with shorter diabetes duration.
A report from the Kazakhstan Ministry of Health documented decreases in both severe hypoglycemic events and DKA in the children during calendar year 2012.
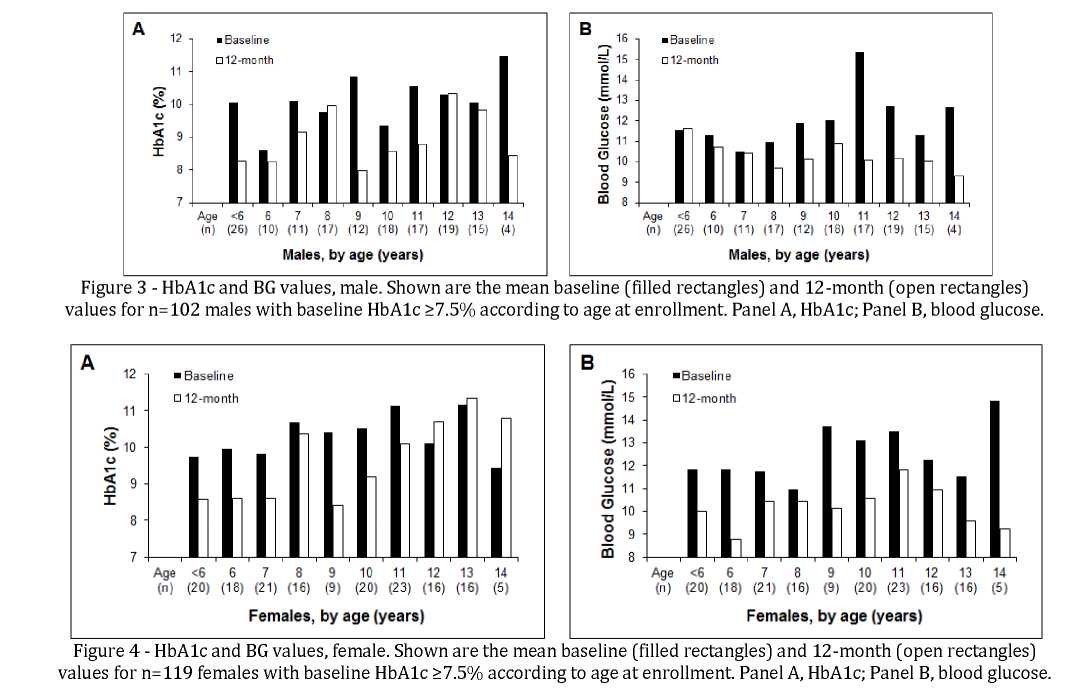
Separate analysis was done according to age at enrollment to identify age-related trends. Figure 3 shows HbA1c and BG data for males, and Figure 4 shows HbA1c and BG data for females. Baseline HbA1c values were lower in boys than in girls, and the reduction at 12 months was greater in boys than in girls. Reductions in HbA1c and BG from baseline to month 12 were seen in most, but not all, age cohorts.
Discussion.
By providing a 24-hour preselected but adjustable basal rate of rapid-acting insulin, along with patient-activated mealtime bolus doses, CSII therapy eliminates the need for periodic injections and provides significant improvements in glycemia, compared to MDI therapy, for many patients with T1DM.
Clinical studies in pediatric populations have shown a significant relationship between HbA1c at commencement of insulin pump therapy and the magnitude of HbA1c improvement. Those who began the pump therapy with an HbA1c of <7.5% had no significant improvement [8].
This is the largest study of insulin pump use in children with fairly long follow-up period conducted in the Central and Eastern Europe. Along with earlier published results on 3- and 6- month follow-ups [15, 16], our data confirm that insulin pump therapy improves glycemic control, with improvements being sustained for at least 12 months. Although this was not a randomized controlled trial, it does reflect ‘real life' experience in a large multiethnic population-based sample over a prolonged period and as such provides important information. HbA1c in the cohort of patients with baseline HbA1c greater than 7.5% decreased by 0.85±3.07 percentage points, which correlates with other studies [8, 17-19]. This magnitude of change is clinically significant, as the
DCCT has reported reductions in microvascular complications of 21 to 49% with every 1% reduction in HbA1c [20].
Patients on pump therapy demonstrated significant improvement both in HbA1c and BG level after 12 months of therapy across all age groups regardless of gender, except teenage girls (age 12-15 years old). Adolescent females with T1DM have special concerns that need to be addressed, including psychological distress, typically manifested as anxiety and depression, and a high prevalence of hyperandrogenic conditions [21, 22]. They are also at risk for “diabulimia” in which insulin dosages are manipulated for the purpose of weight loss and which might increase the incidence of serious medical complications later in life [23]. Teenage girls require special attention and additional training to achieve better glycemic outcomes.
In conclusion, use of modern insulin pump devices for the treatment of T1DM in children and adolescents in Kazakhstan demonstrated high clinical effectiveness with therapy during first year of therapy. Children adopting CSII in the context of government-industry collaborations, particularly those with poor glycemic control and longer duration of diabetes, may realize significant glycemic benefits. The Kazakhstan/Medtronic collaboration provides a model for other initiatives that require rapid deployment and/or massive enrollment for introduction of CSII.
8
REFERENCES
- Dabelea D, Mayer-Davis EJ, Saydah S et al. Prevalence of Type 1 and Type 2 Diabetes Among Children and Adolescents From 2001 to 2009 // JAMA. – 2014. – №311(17). – Р. 1778-1786.
- Patterson CC, Dahlquist GG, Ĝуüгüś E, et al. Incidence trends for childhood type 1 diabetes in Europe during 1989-2003 and predicted new cases 2005-20: a multicentre prospective registration study // Lancet. – 2009 Jun 13. – №373(9680). – Р. 2027-2033.
- Vehik K, Dabelea D. The changing epidemiology of type 1 diabetes: why is it going through the roof? // Diabetes Metab. Res. Rev. – 2011 Jan. – №27(1). – Р. 3-11.
- Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ, Raskin P, Zinman B: Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes // N. Engl. J. Med. – 2005. – №22 – Р. 2643-2653.
- Prince CT, Becker DJ, Costacou T, Miller RG, Orchard TJ. Changes in glycaemic control and risk of coronary artery disease in type 1 diabetes mellitus: findings from the Pittsburgh Epidemiology of Diabetes Complications Study (EDC) // Diabetologia. – 2007. – №50. – Р. 2280-2288.
- Diabetes Control and Complications Trial Research Group (1994). Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus // J. Pediatr. – №125. – Р. 177-188.
- Diabetes Control and Complications Trial Research Group (1993). The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin dependent diabetes mellitus // N. Engl. J. Med. – 2007. - №329. – Р. 977-986.
- Johnson SR, Cooper MN, Jones TW, Davis EA. Long-term outcome of insulin pump therapy in children with type 1 diabetes assessed in a large population-based case-control study // Diabetologia. – 2013 Nov. – №56(11). – Р. 2392-2400.
- Danne T, Battelino T, Jarosz-Chobot P, Kordonouri O, Pankowska E, Ludvigsson J, Schober E, Kaprio E, Saukkonen T, Nicolino M, Tubiana-Rufi N, Klinkert C, Haberland H, Vazeou A, Madacsy L, Zangen D, Cherubini V, Rabbone I, Toni S, de Beaufort C, Bakker-vanWaardeW, van den Berg N, Volkov I, Barrio R, Hanas R, Zumsteg U, Kuhlmann B, Aebi C, Schumacher U, Gschwend S, Hindmarsh P, Torres M, Shehadeh N, Phillip M; PedPump Study Group. Establishing glycaemic control with continuous subcutaneous insulin infusion in children and adolescents with type 1 diabetes: experience of the PedPump Study in 17 countries // Diabetologia. – 2008. –№ 51. – Р. 1594-1601.
- Malik FS, Taplin CE. Insulin therapy in children and adolescents with type 1 diabetes // Paediatr. Drugs. – 2014 Apr. – №16(2). – Р. 141-150.
- Brorsson AL, Leksell J, Viklund G, Lindholm Olinder A. A multicentre randomized controlled trial of an empowerment- inspired intervention for adolescents starting continuous subcutaneous insulin infusion—a study protocol // BMC Pediatr. – 2013 Dec 20. – №13. – Р. 212.
- Phillip M, Battelino T, Rodriguez H, Danne T, Kaufman F; European Society for Paediatric Endocrinology; Lawson Wilkins Pediatric Endocrine Society; International Society for Pediatric and Adolescent Diabetes; American DiabetesAssociation; European Association for the Study of Diabetes. Use of insulin pump therapy in the pediatric age-group: consensus statement from the European Society for Paediatric Endocrinology, the Lawson Wilkins Pediatric Endocrine Society, and the International Society for Pediatric and Adolescent Diabetes, endorsed by the American Diabetes Association and the European Association for the Study of Diabetes // Diabetes Care. – 2007. – №30. – Р. 1653-1662.
- Kordonouri O, Hartmann R, Danne T. Treatment of type 1 diabetes in children and adolescents using modern insulin pumps // Diabetes Res. Clin. Pract. – 2011 Aug. – №93 Suppl. 1. – Р. 118-124.
- Pickup J. Insulin pumps // Int. J. Clin. Pract. – 2011 Feb. – № 65 Suppl. 170. – Р. 16-19.
- Toktarova N, Muratalina A, Tulegalieva A, Kulkaeva G et al. Insulin pump implementation in Kazakhstan // Clinical Medicine of Kazakhstan. – 2012. – № 4(26). – Р. 19-22.
- Toktarova N, Muratalina A, Yedigarova L. International Collaboration – Insulin Pump Therapy for Children in Kazakhstan // Clinical Medicine of Kazakhstan. – 2013. – № 1(27). – Р. 150-158.
- McMahon SK, Airey FL, Marangou DA et al. (2005). Insulin pump therapy in children and adolescents: improvements in key parameters of diabetes management including quality of life // Diabet. Med. – 2007. - №22. – Р. 92-96.
- Pickup JC, Sutton AJ (2008). Severe hypoglycaemia and glycaemic control in type 1 diabetes: meta-analysis of multiple daily insulin injections compared with continuous subcutaneous insulin infusion // Diabet. Med. – 2007. - №25. – Р. 765-774.
- Bruttomesso D, Pianta A, Crazzolara D et al. (2002). Continuous subcutaneous insulin infusion (CSII) in the Veneto region: efficacy, acceptability and quality of life // Diabet. Med. – 2007. - №19 – Р. 628-634.
- The Diabetes Control and Complications Trial Research Group (1996). The absence of a glycemic threshold for the development of long-term complications: the perspective of the Diabetes Control and Complications Trial // Diabetes. – 2009. - №5. – Р. 1289-1298.
- Young V. et al. Eating problems in adolescents with Type 1 diabetes: a meta-analysis // Diabet Med. – 2013 Feb. - №30(2). – Р. 189-198.
- Zachurzok A, Deja G, Gawlik A, Drosdzol-Cop A, Matecka-Tendera E. Hyperandrogenism in adolescent girls with type 1 diabetes mellitus treated with intensive and continuous subcutaneous insulin therapy // Endokrynol Pol. – 2013. - №64(2). – Р. 121-128.
- Wilson V. Reflections on reducing insulin to lose weight // Nurse Times. – 2012 Oct 23-29. – №108(43). – Р. 21-22.