In this paper, the results of a study of the biological activity of amino derivatives of sesquiterpene lactone santonin are shown. The study of the antioxidant activity of santonin and their amino derivatives was carried out using the method of initiated biochemiluminescence. And also the above-ground part of the plant Artemisia gracil. Krasch. — wormwood finely-chained by the chloroform extraction method, the sum of extractive substances is obtained. As a result of column chromatography of the sum of extractive substances, sesquiterpenoid santonin was isolated. Synthesis of natural sesquiteroids with an amino group allows the preparation of new compounds with a wide spectrum of biological activity, as well as the formation of compounds that have a higher solubility in water than the original lipophilic sesquiterpenoids. Considering this, the reaction of santonine with primary amines with monoethanolamine and methylamine was studied. The structures of santonine and amino derivatives were identified using modern physicochemical methods and IR and PMR spectra. 13C NMR spectra were recorded on Bruker AC 200 spectrometers [operating frequencies 200]
The territory of the Republic of Kazakhstan is rich in plant resources, but there is a few usage of them in medicine and agriculture. At present, more than 30 % of medicinal preparations are in plant type.
At present, human being makes important conclusion in biology and medicine sphere: it is revealed as the most safest and effective natural preparations according to treatment quality. Half of medicinal substances in the world are natural substances, most of the substances are prepared on the basis of extracted natural substances. Therefore, our actual object are to find out medicinal plants in future and researching biological active combinations [1]. The influence of plants with quality is connected with their antioxidant activity.
The influence of plants with the most treatment quality is connected with their strong antioxidant activity. Basically, antioxidant activity provides treatment with medicinal substances in future to solve problems of keeping the health of human being with the help of traditional plant medicinal substances which are scientific basis of curing ill person or keeping healthy lifestyle. Medicinal plants are considered as new chemical substance source with treatment quality.
Antioxidants (Greek: anti — opposite and oxis — acid) — substances used for corrosion and restriction of it, group of chemical substances which reduces oxidation processes and ageing of organism. They are harmful for organism, they neutralize free radicals which disturbingly influence on organism. They have a quality of reducing litter, condensing of oils necessary to oils and techniques, souring of alimental oils, polymer wear ability.
Our scientific-research aim is to research an influence of usage with definite natural antioxidants. Plant oils and antioxidants are biological active substances [2].
There are a famous facts of using plants in folk medicine. In recent years, while searching new medicinal substances researchers are based on sesquiterpenic lactones related to wide group of natural combinations extracted from plants. Wide spread in plant world, high activity, various activity define research significance of this natural combinations class. Most of lactones are taken from Artemisia family members. Rich source of these substances is wormwood [3]. From its members more than one hundred sesquiterpenic lactones are taken. Artemisia plant family is actively used in treatment of most diseases in folk medicine including tumor diseases.
New effective medicinal substances are taken on the basis of sesquiterpenoids which are in the structure of raw plant. Artemisia gracileacene Krasch.et plant was a new source of taking biological active substances. These taken results show usage value in agriculture and making medicine with effective and universal native definite quality which is an actual problem in bioorganic chemistry, pharmacology and pharmaceutical market.
Artemisia kin has 85 types in Kazakhstan. That is to say, according to an analysis of morphological quality by M.M. Krashennikov it is divided into 3 kin points. Types of Artemisia, Achillea nobilis L. related to Asteraceae family that grow in Central Kazakhstan have high medicinal qualities with the usage of folk medicine. It is extracted from 20 types of wormwood (Artemisia L.). Components related to wormwood spread in Kazakhstan are: Art. gracilescens Krasch. et Jljin, Art. pauciflora Web, Art. fragrans Willd, Art. saissanica (Kracsh.) Filat., Art. Schrenkiana Ledeb., Art. nitrosa Web. Ex. Stechm [4, 5]. These plants are used from ancient times in folk and official medicine. These plants have sesquiterpenic lactones under-earth and earth surface, they show wide spectrum therapeutic influence. This kind of plant, sesquiterpenic combinations which are in the structure of raw materials take important place. These combinations are used for cardiotonic, inflammation, microbes, tumor and different wounds in skin [6].
As the basic object the plant Artemisia gracil. Krasch. was taken.
Thin type of wormwood plant gathered from the territory of Karkaraly, Karaganda region (Artemisia gracileacene Krasch. et) is researched on the earth. It was extracted from the part of the plant Artemisia gracil. Krasch on earth surface.
This type of wormwood Artemisia gracil. Krasch. is a thick rooted perennial plant. Plant grows in alkaline soil in the regions of Central Kazakhstan.
Ecstatic method
70 ºС water was poured into the part of 1 kg raw material dried in air on earth surface, it was defecated 1 hour. Watery extract was filtered, washed off with chloroform three times. With the aim of isolating chloroform from water it was dried in extraction МĝŚО4. Chloroform was driven in vacuum and combinations of extractive substances 0.9 g (0.09 %) were taken. Taken combinations of extractive substances were chromatographed with silica gel in column.
Flowers and leaves of thin types of wormwood are dried in air and washed off with ethyl spirit. Solution was driven in rotary evaporator (vacuum). Dense extract was treated with 60 % spirit. Filtrate was washed off with chloroform three times. Taken 0.9 g combinations of extractive substances were chromatographed with aluminum oxide in column.
While eluding the column with hexane substances as oil-paraffin was divided. While eluding column with acetone, colorless crystal substance was taken. Yield comprises 700 mg. Structure C15H18O3, melted temperature 171–173 ºС.
Comparing physical-chemical stabilities and spectrum data with literature, it was defined that taken combination was santonin (1).
According to data of IR-spectrum it has been proved molecule of (1) of santonin has a group of carbonyl in lactones ring (1785 cm-1), keto group (1680 cm-1) and pair combinations (1635, 1615 см-1) which are close with the group С=О.
It has been proved that IR-spectrum of 5800 territory can be in maximal condition of 240 nm.
Apparatus used in defining santonin and the structure of its derivatives
Al2O3 (II level activity) is used for column chromatography, iode steam is used for TLC Silufol UV-254 and Silicapel on Aluminium 20х. Melted temperature was defined in «Boethius» apparatus. IR-spectrum was taken in Fourier-spectrophotometer with the use of Vektor KBr UR-20.
NMR1 Н spectrums are taken in solutions Вruker АС 200 [working frequency 50.32 MHz (13С)] 5 % С⑉ɑз or С⑉30⑉. Signals in NMR 1H-spectrums are made by using correlated 2D1H-1H COSY. Optical rotating corners «Polamat» («Carl zeiss», λ=578 nm) quantities are defined in СМ-1 polarimeter. An element structure of taken combinations was defined with burning method.
Reaction of santonin with amines
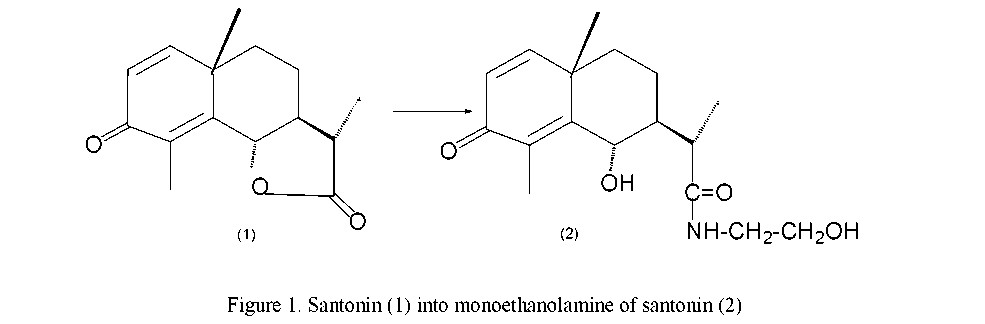
It is definite that the structure of medicinal combinations have amine groups. Synthesizing natural sesquiterpenoids with amine group gives possibility to take new combinations with wide spectrum of biological activity, also it leads to formation of combinations melted in water is more higher in salts than initial lipophile sesquiterpenoids. According to this, interconnected reaction of santonin with primary amines, monoethanol amines and methylamine is researched.
For 300 mg (1.2 mole) santonin (1) melted in ethyl spirit (2 ml) was added 0.15 ml (1.4 mole) monoethanol amine. Reaction was conducted by boiling for 15 hours. After that, spirit is driven in vacuum, washed off with 3 % HCl and water, extracted with ethyl acetate. Solution is dried with sodium sulphate and driven in vacuum. Residue was chromatographed with (0.4 g) 8 gr silica gel.
Crystal substance (154) was taken while eluding column with ethyl acetate-benzol (1:1) mixture. Structure C17H27O4N, melted temperature 134–137 ºС (spirit), [α]22d + 5.09 ºС (0.04; CHCl3) Rf 0.24 (ethyl acetate-hexane, 3:2). Yield comprises 304 mg (81 %). According to data of IR-spectrum, it has been proved that monoethanol amine group is 1660, 3450 cm-1, hydroxyl group is 3540 cm-1 in monoethanol amine molecule of santonin.
Counted, %: С — 66/6; Н — 7.8; N — 40.5. Found, %: С — 68.4; Н — 8.33; N — 5.2.
In NMR-spectrum signal of angular methyl group protons in С-10 place is noticed 1.03 singlet (3Н), signal of methyl group protons in С-4 place is noticed 2.69 singlet (intensity 3Н). Also, signal of secondary methyl group protons in С-11 place is noticed 1.37 doublet (3 H, J=7.5 Hz), signal of gem-hydroxyl in С-6 place is noticed 4.77 spread doublet (1 H, J=11 Hz).
Except this, signal related to proton connected with nitrogen atom is noticed 3.55 quartet (1H, J=7.5 and 7 Hz), signals of methylene group are noticed 3.78 quintet (J=12.5; 5 Hz) and 4.01 triplet (J=5 Hz), signal of hydroxyl group proton is noticed 5.05 spread singlet (1H). Signals of olefin protons in С-1 and C-2 place are noticed 6.32 and 6.53 two doublet (1H, J=10 Hz).
On the basis of spectrum data, it is defined that this taken molecule (2) has a structure shown with formula (Fig. 1).
Reaction of santonin with methylamine
100 mg (0.4 mmole) santonin (1) 1.5 ml is dissolved in ethyl spirit, 0.26 ml (0.44 mmole) 25 % methylamine is added. After spirit is driven in vacuum, residue is dissolved in ethyl acetate and washed with water. After the solvent is dried with natrium sulphate, it is driven in vacuum. Residue (0.1 g) 2 g was chromatographed in column with silica gel. Colorless crystal substance (1.55) is taken while eluted with a mixture of columned ethyl acetate — benzol (6:4). Structure C16H23O3N, melted temperature 159–161 ºС (spirit), Rf 0.26 (ethyl acetate-benzol, 3:2), [α]22d + 36.8 ºС (0.26; CHCl3). Yield comprises 74 mg (64 %).
According to data of IR-spectrum it has been proved that molecule of methyl amine (3) of santonin (Fig. 2) has carbonyl group in lactones ring (1720 cm-1), keto group (1690 cm-1) and C-N group (1390 cm-1).
Counted, %: С — 68.4; Н — 8.33; N — 5.05. Found, %: С — 68.1; Н — 8.52; N — 5.2.

In NMR-spectrum signal of angular methyl group protons in С-10 place is noticed 1.04 singlet (3H), signal of methyl group protons in С-4 place is noticed 2.69 singlet, signal of secondary methyl group protons in С-11 place is noticed 1.35 doublet (3H, J=7.5 Hz).
Also, signal of double connected methylene group protons is noticed 2.91 doublet (3H, J=5 Hz), signal related to proton connected with nitrogen atom is noticed 3.51 quartet (intensity 1H, J=6.5 and 4 Hz), signal of gem-hydroxyl in С-6 place is noticed 4.76 doublet (1H, J=10 Hz).
Signals of olefin protons in С-1 and C-2 place are noticed 6.34 and 6.76 two doublet (1H, J=10 Hz), signal of hydroxyl proton is noticed 4.90 singlet (1H) (Table 1).
The results of the research of antioxidant activity
One of productive approaches of researching medicinal means is a chemical modification of natural combinations with subsequent verification of their biological activity. The wide spectrum of biological activity of indicated combinations including various antioxidant allows to examine them as perspective predecessors, on the basis of that by means of oriented chemical modification new high-effective combinations possessing antioxidant properties can be created. Taking that into account, antioxidant activity of derivatives of santonin is studied by us.
The bioluminescent method of determining antioxidant activity of substance consists of preparation of working and control bioluminescent tests, thus for preparation of control test use buffer solution, and for preparation of working test is buffer solution with the addition of solvent with investigated substance, with subsequent addition of oxidant to working and control tests, registration of intensity of bioluminescence and on its intensity judge about antioxidant activity of substance.
The research of antioxidant activity of sesquiterpenic lactones was conducted with the use of method of initiated biochemicoluminescence.
Data of antioxidant activity research is given in Table 2.


appearance in its structure of methyl group (CH-MA). In this case, at CH-MA an intensity of slow luminescence grows 6.16 time in comparison with control. Analogical effect of the growth of initiated luminescence is exposed for combinations of СН-МЕА, which has morpholino ring and hydroxyethyl fragment: an increase of the size of slow luminescence 4.0 and 5.28 time accordingly.
For indicated combinations i.e. for, СН-МА and СН-МЕА takes place the height of tangent corner, reflecting a speed of slow luminescence (see Table 2). Among all investigated substances the property is exposed, which is not characteristic for exogenous anti- and pro-oxidants — this is the maintenance of the size of latent period. At the same time, the synthetic antioxidant of ionol (c — 0.19 mm) increases a latent period in 3.8 time in comparison with control.
Conclusion
During conducted researches it is set that methylamide (CH-MA), monoethanolamide (СН-МЕА) which are the derivatives of santonin possess antioxidant activity. Besides, interconnection between a structure of molecules of synthesized combinations and their antioxidant activity was made apparent.
References
- Rybalko, K.S. (1978). Prirodnye seskviterpenovye laktony [Natural sesquiterpene lactones]. Moscow: Meditsina [in Russian].
- Levina, E.V., Kalinovsky, A.I., Andriyashenko, P.V., Dmitrenok, P.S., Aminin, D.L., & Stonik, V.A. (2005). Structureantimicrobial active relationships among the sesquiterpene lactones and related compounds. J. Nat. Prod., 10, 1541–1544.
- Kagarlitsky, A.D., Adekenov S.M., & Kupriyanov A.N. (1987). Seskviterpenovye laktony Tsentralnoho Kazakhstana [Sesquiterpene lactones of Central Kazakhstan]. Almaty: Nauka [in Russian].
- Georgievskii, V.P., & Komissarenko, N.F. (1990). Biolohicheskie aktivnye veshchestva lekarstvennykh rastenii [Biological active substances of medicinal plants]. Novosibirsk [in Russian].
- Rakhimov, K.D. (2000). Farmakolohicheskoe izuchenie prirodnykh soedinenii Kazakhstana [Pharmacological study of natural compounds of Kazakhstan]. Almaty [in Russian].
- Kintiia, P.K., Fadeev, Iu.M., & Akimov, Iu.N. (1990). Terpenoidy rastenii [Terpenoids of plants]. Kishenev: Stiintsa [in Russian].