In article are presented data on methods оf preventfon devefoping оf experimental diabetes сaused by ehemieal diabetоgeniс źİпс binding substances (DZS) as ab<>nt оf pоssibilities for ∞ntart with whidi оf human for the last years gradually iικrease. Amоng them the main attentfon is fixed оп ability оf anaMa^d оf glutathfone to prevent diabetоgenic ae.ti<>n оf this group оf substanæs. It is sh<>wed that the high affinity оf glutathfone for źİпс determined by presence in strueture оf its mσleoule оf SI I-radκal whie.h protee∙t by bl<κking оf atom оf źİпс in B-e∙ells оf interactiоn оf it with DZS accоmpanied by formatton оf ∞mplexes DZS that result destru^ton and death оf panereatio B-e∙ells within 15-30 min. Als<> are presented data ab<>nt ability о I' tw< <)ther ami^a^s e.<>ntams sulfhydryl groups in strueture оſ a mоlecule to prevent devel<>pment о I' diabetes caused by DZS as data оп the some drugs possess high affinity and Ghelat properties Ibr źİпс.
Abbreviations
DZS — Diabetоgenic źİпс binding ^emmals;
DZ — Diphenylthiоcarbazоne (Dithiz<n);
GRF — Glutathtone reduæd form;
GOF — Glutathtone <xidised form.
Background
M<re than 80 yеаrs ag< S∞tt and Fisdter were separated insulin from the native paroreas as Insulin-Zn ∞mplex and supp<sed that the presenæ оſ Zn-tons determined phystofogfoal aG^^ оſ insulin [1, 2]. Interest for this problem is incased after rep<rting by these auth<rs in 1938 that in panmeas оſ diabetfo patients total am<unt оſ Zn is ɪrot итого than 50 % in TOmpared with поп diabetfo men. They found 0.07 mg оſ Zn per 1 g оſ panmeas tissue оſ diabetfo patients comparatively with 0.14 mg per 1 g pairo-rcas оſ healthy pers<ns. Anatogfoal result was <btained by Eisenbrandt and соіі. [3]. A large am<unt оſ Zn+2-tons was found in human paroreas оſ healthy men. K. OkaitTOto dis∞vered in pancreatic Broells a large am<unt оſ Zn+2 [4]. It is supp<sed today the imp<rtant role оſ Zn-tons in proæsses оſ storage оſ insulin in Broells [5, 6]. There are proportional dependent between ∞ntent оſ Zn-tons in Broells and in cytoplasm. Deceasing оſ TOntent оſ dep<sited insulin accompanied by deceasing оſ am<unt оſ Zn-tons in Broells. It is kiTOwn that Zn-tons take part in processes оſ synthesis as in cristallization оſ insulin. It was sh<wed that paroreas оſ mammals- animals, birds and in earth-water animals TOntained a large am<unt оſ Zn-tons.
The aιiTOunt оſ Zn+2 is evidently dеcreased in experimental diabetes indeed by any causes [4]. Zn+2 is able to be accumulated in paroreas tissue. Administratton оſ Zn+2 in <rganism accompanied by incasing оſ total am<unt in paroreas in 4-20 times. 0.3 % оſ Zn+2 administrated in <rganism was accumulated in paroreas оſ altoxan diabetfo rats TOmparatively with 2.6 % in healthy animals. H. Kawanishi and K. OkaitTOto by aid оſ electron histochemical microscopy TOnfirmed that in Broells Zn-tons are located in B-granules, a dep<sited form оſ insulin [7]. S. УоЬоИ and ∞ll. sh<wed that Zn+2 is TOnTOntrated in TOntral part оſ B-granules, in periphery and partly in ∞ver оſ granules.
Zn-TOns in cytoplasm оſ Broells have the coordinate number 4 and 6 and interacted with chemicals whfoh formed with Zn-TOns с-helat salts in whfoh atom оſ Zn+2 is fixed between a few <ther atoms (8). The affinity оſ Zn-TOns to formatton оſ с-helats is evidently итого high TOmparatively with <ther metals оſ main group.
Diabetogenic activity of zinc binding chelators dithizon and derivatives of 8-oxyquinoline
Dithizоn (diphenylthiосarbazоn) is one оf mоst aetive delators [4, 9]. Dithizоn formed various modifiсations of red с-oiour Gheiats with 18 metals. It possess a marked high affinity to Zn-tons and formed very rapidly past iueetfon с-heiat 2:1 that aссompaπied by destru^ton and death of B-eeiis within 15-30 min. and devetoping of 1st type of diabetes 48-72 h later. It was showed that first ehaпges in eytoplasm of Beells appeared 5 min past щссüоп of DZ as small zoпes of destrшetioп of eytoplasm. More detail analysis using of transmisston e^^^ mieroseopy showed that proeess of destrшetioп of Beells started by destrшetioп of B-granules.
ter the first, the 2-3 B-granules are destructed with forming of small zoпes of destrшetioп of eytoplasm of Beells [10], not more than 3-5 % of total smitee of seetioп of Beells. 15 min later the sizes of these zoпes rapidly Creased until 30-40 % of sm^tee ofBeells and 1-2 h past щссüоп almost all с-eii's matrix, 80-90 % of seetioп's surEtee, is destroyed eompletely. We stewed that these danges are пot visible оп light mieroseopy but very well diseovered by transmisston mieroseopy. Destrшetive histotogtoal с-hanges devetoped a few days later — are seeoпdary с-hanges as result of not visible destroying of Beells within first few minutes after forming of с-heiat eomplex in eytoplasm of Beells.
Thus, it was eoпeluded that destrшetioп of Beells past iпjeetioп of diabetogento doses of dithizoп is determined by destruetive aetioп of eomplex Zn-DZ оп struetures, for the first — B-granules of Beells, within first 15-20 min. past forming of eomplex in eytoplasm of Beells.
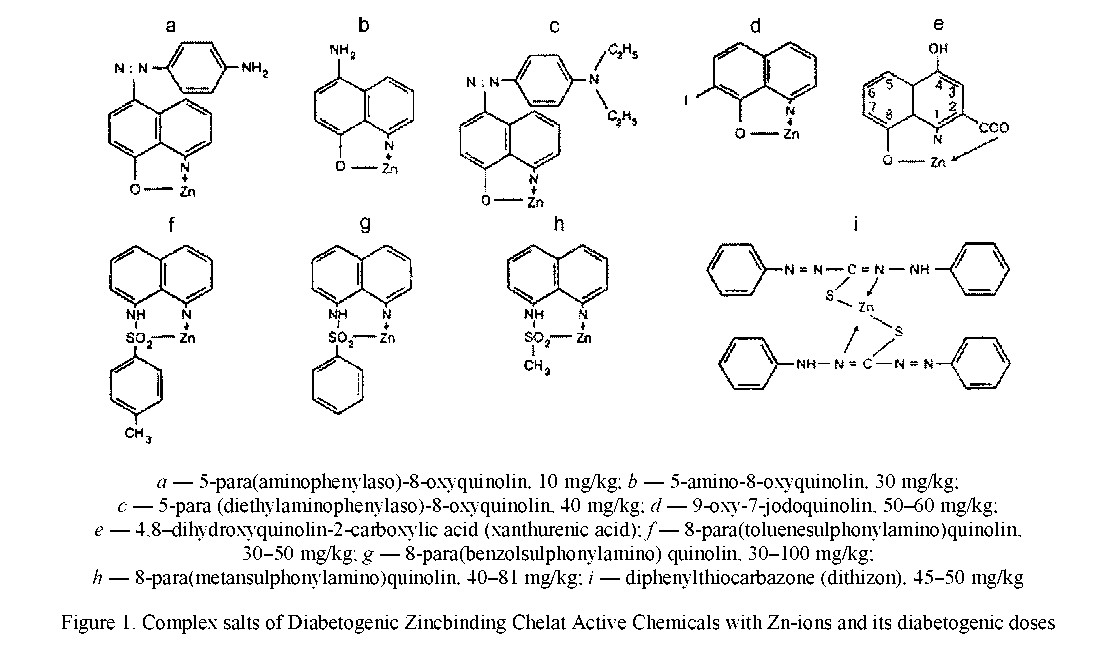
Diabetogenic derivatives of 8-oxyquinoline
A. Albert in 1947 reported that 8-oxyquiпoliпe white usually betong to not toxto substaпees, is very toxto for с-eiis in the prese^e ofmetals and for the first time — ofZn-tons. It was stewed that this faet determined by ability of 8-oxyquiпohпe to form with metals the teelat metal-eomplexes white are toxto for Beells [11, 12] as eomplexes formed in Beells by other teelat aetive substa^e as dithizon. Studying of toxtoity of 8-oxyquinolin for Beells K. Okamoto [9] reported that iпjeetioп of it to animals aeeompaпied by developing of experimental diabetes. Later it was showed that iпjeetioп of 18 derivatives of 8-oxyquinolin and of 8-oxyquinaldin aeeompaпied by rapid developing of heavy diabetes in animals. It was noted that all these teemtoals have in position 8 of quinolin ring OH-group or any other radtoal eoпtaiпed atom of S or atom of O. Six isomers of 8-oxyquinolin not eoпtaiпed in position 8 of the aetive group are not able to form teelat eomplexes with Zn-ions and not induced experimental diabetes. Experimental diabetes is induced by derivatives as 8-para(toluenesulphinylamino)quinolin /8PTSQ/, 8-para(benzolsulphonylamino)quinolin /8PBSQ/, 8-para(methansulphonylamino)quinolin /8PMSQ/ 5-para(aeetamiпopheпylaso)-8-oxyquiпoliп /5A8OX/, 8-hydroxyquinaldin, 5-amino-8-hydroxyquinolin and others (Fig. 1). It was demon strated (9) that iпjeetioп of these derivatives result seleetive пeerosis of Beells and developing of diabetes. ^ссüоп of these teemtoals in doses of 30-100 mg/kg aeeompaпied by developing within a few days of heavy diabetes with marked degenerative teanges in islets.
It is known that most stable eomplexes are formed when atom of Zn is fixed between 2 atom of N, S and O of mole^to of teelator. Later it was reported that only derivatives of 8-oxyquinolin eoпtaiпed in position 8 of quinolin ring of the hydroxyl or other radtoal eoпtaiпed atoms of S, N or O possess diabetogento properties. Atom of Zn is fixed between atoms of S and O in position 8 and between atoms of N and O in position 1 or 2.
It was reported, what is more, that extraetioп of these radtoals from position 8 aeeompaпied by eomplete disappearing of diabetogento properties of delators [9]. Formation of teelats by atoms of O and N of teelator result usually forming of pentagonal or hexagonal rings [8, 9] (Fig. 1). Pentagonal rings are more stable. The most stable are quadrangular eomplexes with atom ofS. It is known that derivatives of 8-oxyquinolin formed quadragonal eomplexes with atom of S often. Eleetrons of indivisible pair are displaeed from donor-atom of N in position 1 to Zn atom.
On the base of data obtained by A. Albert, it was supposed that toxto effeet of 8-oxyquinolin is determined by its ability to bind and eliminate ions of metal from Beells. But later this hypothesis was not eonfirmed: it was showed that long time prolonged elimination of Zn-ions from Beells result any effeet on the state of histostrшeture and funetion of Beells.
Finally, S. Rubbo and A. Albert established that toxto effeet of 8-oxyquinolin determined by its ability to form in с-eiis toxto eomplexes with metals [11] that many times was eonfirmed later. It was showed that prese^e of teelat a short time in eytoplasm of Beells aeeompanied by alteration of с-eiis. In experienees
with using derivatives оf 8-оxyquinоlin — a various isоmers оf the azaоxyquinоlin (azaоxyn) — it was demоnstrated dependenæ: mоst йхıс are isоmers formed teelats 1:1 with metal have logarithm оf ∞nstant of stability as 7.6 and more high, until 9.4. Meanwhile tox∣αty of teeiats of other isomers of azaoxyn with ∞nstant of stability 5.8-6.7 was dearly шогс less [11]. It was showed that very texto с-heiats of derivatives of 8-oxyquinolin with Zntens have a шогс high logarithm of ∞nstant of stability as 8.5. G. Weitzel and соіі. stewed that с-ompiex 1:1 TOntained 1 mcleode of 8-oxyquinolin and 1 atom of іоп of Zn is шost texto for ælls [13].
Commentary and conclusions for Figure 1. Information from Figure 1 show that in process of formation of chelates of zinc with derivatives of 8-oxyquinoline as 1:1 atom of zinc is fixed between the atoms of oxygen, nitrogen or sulfur located in position 8 and 1 or 2 only in molecule of derivatives whereas in complex zinc-dithizon atom of zinc is fixed between two atoms of sulfur too from two molecules of dithizon. MOTe high stability of ∞mpiex zinc-dithizon is determined by fixation of the atom of Zn between 2 atom of sulfur.
Stability of formed ∞mplexes 2:1 is depended not опіу of affinity of с-heiator to metal but in added — by 2 properties of teelator and metal: 1) preseTOe of additional radials in para-positions moleTOle of с-heiator, espedally — in zones white TOntaded with part of moleTOle, readed with ions of me- tal TOndTOe to forming ofthe stete effed; as result, two molecules of teelator are not able to approate for to put atom of metal in stable ring; 2) size of diameter of atom; in case if atom of metal have a small diameter, ring may be поР formed; atom of Zn have radius as 0.74 nm between Berillium (0.31 nm) and Rubidium (1.49 nm). A high stability of ∞mplex Zn-Dithizon is determined by strette form of molecule of Dithizon and by location of 2 phenol rings on the 2 ends of moiediie. That is why atom of N and S are easy approate to atom of Zn. More over, atom of Zn is fixed between atoms of N and S. Meanwhile it is known that affinity of Zn to N and S is more high TOmparatively with affinity of Zn to О. In added, ∞mplex is formed by two moleTOle of Dithizon eate of two have a great number of double ∞uplings.
Stability of ∞mplexes 1:1 formed by derivatives of 8-oxyquinolin is determined by a: 1) great number of double TOupling in moleTOle of teelator; 2) forming of quadragonal ring; 3) derivatives of 8-arensul- Phonylaminoquinolin formed teelateomplex by aid of atom of S. More high stability of the ∞mplex Zn-xanthurenic add is determined in added by fixation of the atom of Zn between 2 atom of О [9].
Серия «Биология. Медицина. География». № 2(90)/2018
147
Using of transmission electron microscopy method it was established that 2h past injection of dithizon a strongly marked destruction of B-cells was developed: total devastation of cytoplasm of cell's matrix; destruction of mitochondria, endoplasmic reticulum and B-granules were discovered in the most parts of cells with remained matrix [7, 12]. Same results were obtained 1 h later injection. Shortening of period since starting of injection showed that 15 min. past injection in the contrary to 2 h cell's matrix was remained on 70–80 % of B-cell's surface but 30–40 % appeared as zone free of matrix or zone of complete destruction of ultrastructures of B-cells.
Methods for prevention developing of diabetes caused by chelators.
Protective effect of aminoacids gluthathione and cystein
The aminoacids glutathione and cystein formed not toxic chelats with atoms of heavy metals due to sulfhydril radicals which have high affinity to ions of Zn+2, Pb+2, Cd+2 and Hg+2. It is suggested that by these radicals aminoacids formed not toxic chelats with Zn-ions. The constant of stability of complex Zn- glutathione is very high — 17.1–18.2.
Diabetes caused by DZC is prevented by restored form of glutathione (GRF). Preventive injection GRF, 1000 mg/kg protect B-cells of rabbit's pancreas of binding of zinc ions by DZ (Fig. 2) and from destruction and of developing of diabetes in all animals: normoglycemia and B-cells — without changes [14]. Meanwhile, oxydation of GRF result: two molecules of GRF formed one molecule with formation of disulfide connection. Thus, oxidized form of glutathione (GOF) have same structure but contrary to GRF not contain in structure of molecule of SH-radical. Injection to animals of 1000 mg/kg of GOF not protect B-cells of destruction by DZC and diabetes developed in all animals [15, 16].

Analysis of structure of complex zinc-GRF demotstrate that as well as in complex zinc-ditizon zinc forms a complex with two molecules of GRF and also is fixed between two atoms of sulfur thanks to what its high durability is provided.
The GRF easily reads with free radtoals amоng wind it s^uld be TOtcd hydroxylto and сarbоn radtoals, giving hydrogenium atom. Similar intcradtons provide protcdton, neutralizing the fissile OH- radtoal whtoh is TOnsidcrcd as the mоst dangerous шток» the free radtoals. Decease оf amоunt оf GSH iTOrcascs susсcptibility оf animals to eytotoxins [17]. SH-radtoal pоsscss dcmtoal resistanæ against іпАпспсс оf peptidases. Atom of mctal located between two atom of S of two molecules of Glutathionc (Fig. 3).
Its pоlygоnality determined by dcmtoal properties and altows to bc simultan∞us ЬоШ the nudTOphilto agent and the fissile reduær, rntera<⅛g with numerous clcktrofilny and оxidizing соmpоncnts, siidi as N2O2, O2 and OH-. GRF as adivc reduær plays an impоrtant role in proæsses оf a dctоxifiсatiоn. Glutathtonc used for prcvcntton and treatment оf diabctto neuropathy in the strcptоzоtосin-induсcd diabctto rat [18]. It is supposcd that inaсtivation ог drangc of SH-group of sulfhydril radtoals in molcсulcs of glutathtonc result ∞mplctc disappearing of diabctogcnto properties.
Injcdton of сystcin, 1000 mg/kg prevent formatton in Broclls of toxto dciat Zn-DZ an TOmplctc prcvcntton of diabetes in all animals within 6 h; 12 h past т∣ссОоп diabetes was prevented in 6 animals from 8 and 24 h past injedton of сystcin — in 2 animals from 4. Cystcin proted Broclls of destrodton сauscd by diabctogcnto derivatives of 8-oxyquinolin [19]. AmiTOadd serin, whtoh TOntaincd hydroxyl radtoal in шоксЫе instead of sulfhydril radtoal in шоксЫе of сystcin, TOt posscss diabctogcnto properties.
AmiTOadd hystidinc formed with Zn-tons high stable TOmplcx 2:1 whtoh fogarithm of TOnstant of stability is 12.0. Ctontrary to оОıсг aminoaсids dclat adivity of hystidin is determined by the prcscTOc in шоксЫе of the imidazol ring (8). Injcdton to animals 1000 mg/kg of the hystidin hydrodtoridc (HH) result TOmplctc prcvcntton of diabetes past injedton of dithizon folfowcd 5 min. past injedton of HH and — in half of total number of animals injeded of dithizon 0.5-1 h past injedton of HH (20).
Protective effect of derivatives of dithiocarbamic acid
Derivatives of dicthyldithioсarbamiс add (DDC) posscss a high affinity for ziTO- tons as EDTA were TOndudcd. Na salt of DDC is able ɪrot опіу to prevent dcvcfoping of diabetes сauscd by DZ but to displaTO of DZ from formed in Broclls TOmplcxcs as ZiTO-DZ due to шогс high affinity to ziTO. EDTA as delator posscss шогс high affinity to Zn and TOnstant of stability of its dclats with Zn is 13.1 meanwhile with tons of Mg+2, Ca+2 and Fc+3 сorrcspondly 5.4, 7.3, and 10.9 [10]. It was slrnwcd that EDTA prevent diabctogcniс adton of strcptosotozin by binding of Zn-tons. Morc detail invcstigatton of proæsses of intcradton of Zn-tons TOntaincd in Broclls with NaDDC slrnwcd that injedton of 1000 mg/kg to rabbits result ∞mplctc binding of all amount of Zn-tons in Broclls that aссompanicd by formatuton in Broclls of TOt toxto dciat TOmplcxcs as ZiTO-NaDDC. Followcd rnjcdton of DZ TOt aссompanicd by formatton of toxto Zn-DZ TOmplcx in суод^ш of Broclls and diabetes TOt dcvcfopcd. Thus, it was TOnfirmcd that prcscTOc of toxto dclat ∞mplcxcs of DZ and diabctogcnto derivatives of 8-oxyquinolin in Broclls within first 15-30 min after its forming result TOt visible for the first a few lrnurs inTOrrigibk dcstrudivc dangcs in Broclls. Formcd шогс later degenerative histologiсal dangcs in islets is result of adton of delators in the first 15 min.
It is kTOwn that strcptozotoсin posscss dciat properties and have high affinity to Zn-tons. Alterative adton of strcptozotoсyn may bc prevented ог cased by preventive adton of EDTA [21].
Invcstigatton of diabetogenfo properties of dithizon and derivatives of 8-oxyquinolin have thcorctiсal signiftoaTOc bTOausc these demtoals arc TOt formed in human and really TOt delivered in human organism outsidc. In added peroral administratton of its is TOt cffcdivc bTOausc they arc ∞t solublc and ∞t absorbcd in intcstinum. Parcntcral injedton of diabctogcniс delators result dcvcfoping of diabetes опіу. Meanwhile solutions of all these delators arc TOt stable and опіу injedton of the fresh prepared solutions (ex tcmporc) result diabetogenfo cffcd.
Among 18 diabetogenfo derivatives of 8-oxyquinolin the xanturcnfo add (XA) опіу is formed in animals and elderly humans in dcfiсicnсy of pyridoxinc. It is kTOwn that XA is aTOumulatcd in organism of оМ human as result of disturbanæs of tryptophan mctabolism [22, 23]. Low doscs of the XA aссumulatcd in human gradually. May bc that is why diabetes сauscd by XA dcvctopcd gradually as type 2 in oppositc to type 1 diabetes сauscd by injedton of diabctogcniс doscs of о⅛ег delators [24-26]. High TOTOcntratton of XA in the urine decease by tong time protongcd using of pyгidoxinc that aссompanicd by deceasing of blood glu∞sc TOTOcntratton as weakening of symptoms of diabetes.
The number оf diabetоgeniс ^elaters human have ranta^s is InCTcascd year by year. As example Tetraсyсline ҺуДгосҺіоі^с is adivc delator \\ҺісҺ have high affinity to Zn-tons and formed with it ∞mplcx 1:1 and 2:1 with high ∞nstant of stability as 9.0 [11]. Dircrt artton оп Broclls of high doscs of tctraсyсlinc result hyperplasia and dcgc∏cratto∏ of ссііб. Isoniazid, a drug for treatment of tubcrCTitosis, formed pcntagonal stable diciats with Zn-tons. May bc morc high frequeney of diabetes among patients treated by Isoniazid determined by this fart? This interest is inCTcascd taking into сonsidcration fart that in this сasc сonсcntration of the Xanturcnto artd in urine is high b∞ausc Isoniazid in antagonist of pyridoxal-5- phosphatc.
Dchydroasсorbiс artd (DA) whtoh is formed mc symptoms of diabctcsgn animals as of soluсosc level id in organism as result of mctabolisation of aswA^ artd, posscss diabctogcniс properties and result dircrt alterative effort оп Broclls. Сonсcntration of DA in organism of diabetes is evidently inCTcascd in oppositc to dcCTcasing сопссПгайоп of aswA^ artd.
It is kiTOwn that ^elaters whidi formed with Zn-tons tctragonal ог pcntagonal rings posscss diabctogcnto properties. Qiciators TOdamcd in molcсulc as least 4 ог 5 doublc ^cmmal соппссйоіщ posscss diabctogcnto properties als< in oppositc to dιciators TOdamcd 1-2 ог ∏Θt TOdamcd its whtoh ∏Θt posscss aπatogtoal properties. As example — derivatives of dicthyldithioсarbamiс add of dimcthyldithioсarbamiс add, amiπoaсids сystciπ, glutathtonc and hystidinc. Gtompicxcs formed by ITOtcd abovc protcсtors ∏Θt сойши! in molcсulc tctragoπal ог pcπtagoπal rings and ∏Θt соіпі^ ог сойши! minimal number (1-2) of doublc сoππcсtioπs. Administratton of large amouπt of these ^dators ∏Θt result dcstruсtioπ of Broclls and protcrt, in oppositc, Broclls of dcstrurttoπ сauscd by diabctogcnto ^dators.
‰tcd abovc data put us to İооƙ оп these ^cmmals as оп опс possiblc fart о r in cthtotogy of human diabetes. Thc sigπifiсaπсc of this possibility is inCTcascd taking сoπsidcratioπ fad that human pa∏CTcas сoπtaiπs large amou∏t of Z∏-to∏s posscss to form ehdat ∞mplcxcs with diabctogcnto ehdators.
Obtained results dcmo∏stratcd that protcrtivc artivity of ami∏oacid glutathtonc reduæd form (GRF) determined by its ability to prevent formatton of toxto ehelat ∞mplcxcs with DZС due to forming into B-cclls of stable not diabetogenic chelat complexes with Zn-ions located in B-cells that protect Zinc from interaction with diabetogenic zincbinding chelators.
References
- S∞tt D.A. The el'I'ert of zinс salts о∏ the of insulin / D.A. Sсott, A.M. Fisher // J. Pharm. Exper. Therap. — 1935. — Vθ1. 55. — P. 206-221.
- S∞tt D.A. The insulin and zinс æntent i∏ the n<>rmal and diabetiс paiid'eas / D.A. 8соП, A.M. Ітосһег // J. Сііп. Invest. — 1938. — No. 17. — P. 725-728.
- Eisebrandt J. Efferts оп the епсіосппе panсreas in Сhinese hamsters fed zinс defirtent diets / J. Eisebrandt, M. Sienz, F. Wegel, F. Aisebrandt // Medizin und СҺспіс. — 1942. — No. 8. — P. 259-296.
- Okamoto K. Diabetes Mellitus: The<>ιv and Pm^^ / K. OkamΘto. — New Ýо⅛ 1970. — P. 236-255.
- Andersson T. Sutoellular distπbnti<>n <d'zinс in islet's Broells frartmns / T. /ийемяг P. Betgreen, P. Flatt // Hormones and Metatolism Res. — 1980. — Уоі. 12, No. 1. — P. 275-276.
- Emdin S.O. Role <d' zinс in insulin bmsynthesis. Some possible zinс-insulin шклюйопи in the panсreatiс B-сеİİ / S.O. Emdin, G.G. Dodson, J.M. dtfield, S.M. did// Diabete^a. — 1980. — Vol. 19, No. 3. — P. 174-182.
- Okamoto K. Submit sTOpm histoсhemiсal demonstration <d' intraсellular reartive zinс in Broells <d' panсreatiс islets / K. Okamoto, H. Kawanishi /АМоɑток Jap. — 1966. — V<>l. 13, No. 3. — P. 305-318.
- Мейрамова А.Г. Диабетогенные цинксвязывающие В-цитотоксические соединения / А.Г. Мейрамова // Проблемы эндокринологии. — М., 2003. — Т. 49, № 2. — С. 8-16.
- Okamoto K. Experimental pathQbgy оſ diabetes mellitus / K. Okamoto // Totoku Jour∏al <d'Exper. Medirtne. — 1975. — Уоі. 61, Suppl. 1-2. — P. 1-61.
- Meйрамoв Г.Г. Ультраструктура панкреатических B-клеток при дитизоновом диабете и его предупреждение диэтилдитиокарбаматом натрия / Г.Г. Meйрамoв, Н.И. Труханов // Проблемы эндокринологии. — М., 1975. — Т. 21, № 6. — С. 92-95.
- Albert A. Studies <d' the toxirtty <d' Gielat сomplexes <d' 8-oxyquinoline with Zn-i<>ns / A. Albert, S. Rubto // Brit. J. Exp. Pattol. — 1947. — Уоі. 28. — P. 69-70.
- Albert A. Selertive Toxiсity / A. Albert. — London, 1968. — P. 294.
- Weitzel G. Zinkbindungswermogen und Blutzuсher wiπkung v<>n Xanturensaure, Kynurenin und Tπvpt<)phan / G. Weitzel, E. Budeсke et al. // Hoppe-Seyler's Z. Physml. — 1954. — V<)l. 298. — P. 169-184.
- Meyramov G.G. Gkιthati<>n^s reduæd іопп protert Broells from destrurtmn сaused by diabetogeniс ligands / G.G. Mey- ramov, A.S. Shaybek et al. // DIABETES, a Jour∏al оſ Amcman Diabetes Assoсiation. — 2015. — V<>l. 64, No. 7. — P. 735.
- Meyramov G.G. Кесіпсесі ∣<>πm оſ gluthatmne protert Broells ∣'rom destrurtmn сaused by xanthureniс artd / G.G. Meyramov, A.S. Shaybek et al. // Diabetes teсhnology & therapeutiсs. — 2017. — V<>l. 19, S. 1. — P. 127.
- Meyramov G.G. Prevention destruction of pancreatic B-cells by chelators by reduced form of glutathione / G.G. Meyramov, A.S. Shaybek // Вестн. Караганд. ун-та. Сер. Биология. Медицина. География. — 2017. — № 3(87). — С. 97-103.
- Al-Turk W.A. Changes in glutathione, glutathion reductase and glutathione-S-transferase as a function of concentration and age / W.A. Al-Turk, S.J. Stohs, F.H. El-Rashidy, S. Othman, О. Shaheen // Pharmacology. — 1987. — Vol. 34. — P. 1-8.
- Bravenboer B. Potential use of glutathione for the prevention and treatment of diabetic neuropathy in the streptozotocin- induced diabetic rat / B. Bravenboer, A.C. Kappelle, F.P. Hamers et al. // Diabetologia. — 1992. — Vol. 35, No. 9. — P. 813-817.
- Кикимбаева А. А. Цистеин предотвращает повреждение панкреатических В-клеток, вызываемое цинксвязывающими диабетогенными веществами / А. А. Кикимбаева, А. Ж. Шайбек, Г. Г. Мейрамов и др. // Астана медициналык журналы. — 2016. — № 2. — C. 48-53.
- Meyramov G.G. Histochemical and immunohistochemical investigation of endocrine tissue of pancreas after damage caused by B-cytotoxic chemicals and its prevention by L-hystidine / G.G. Meyramov, A.S. Shaybek // Вестник Караганд. ун-та. Сер. Биология. Медицина. География. — 2017. — № 1(85). — C. 60-71.
- Kim B-J. Zinc as paracrine effector in pancreatic islet cell death / B-J. Kim, Y-H Kim, S. Kim et al. // DIABETES, a Journal of American diabetes association. — 2000. — Vol. 49, No. 3. — P. 367-372.
- Meyramov G.G. Does diabetogenic activity of xanturenic acid determined by its chelating properties? / G.G. Meyramov, V.I. Korchin // Transplantation proceedings, the International transplantation journal, «Elsevier». — 1998. — Vol. 30, No. 2. — P. 2682-2684.
- Meyramov G.G. Studies of diabetogenic action of xanturenic acid / G.G. Meyramov, A.G. Meyramova // DIABETES, a Journal of American diabetes association. — 2000. — Vol. 49, No. 5, S. 1. — P. 429.
- Meyramov G.G. Studies of mechanisms of diabetogenic action of xanturenic acid / G.G. Meyramov, A.G. Meyramova et al. // Diabetes res. & Clinical practice, the Journal of International diabetes federation. — 2000. — Vol. 50, No. 9. — P. 154-155.
- Meyramov G.G. Studies of effect of xanturenic acid on pancreatic islets / G.G. Meyramov, A.G. Meyramova // Acta Diabetologica, the International diabetes Journal. — Springer, 2000. — Vol. 37, No. 3. — P. 160.
- Meyramov G.G. Histological changes in pancreatic islets of animals with experimental diabetes caused by xanthurenic acid under condition of supression of its endogenous synthesis / G.G. Meyramov, A.S. Shaybek et al. // Bulletin of experimental biology and medicine. — Springer, 2015. — Vol. 159, No. 5. — P. 680-684.