In article is presented the results of studies of protein and carbohydrate metabolism in experimental animals under the influence of copper acetate in the tolerable daily intake (TDI) doses. Positive effects of correction with the infusion of Beta vulgaris seeds were studied. It is shown that the consumption of copper acetate by the animals at the level of TDI has a negative effect on the basic biochemical parameters of protein and carbohydrate metabolism. As a result of the study, it was found that the main indicators of blood biochemistry are directly depended on the duration of the effect of copper on the body of experimental animals. Receiving a solution from the infusion of Beta vulgaris's seeds as a correction in the presence of copper acetate caused a positive effect, which is manifested by a decrease in the synthesis of metabolic wastes (urea and creatinine). Copper acetate after correcting of infusion of seeds activated the processes of protein synthesis and glycolysis. Apparently, the intake of copper acetate, causing toxic effects, is simultaneously a stress factor that caused a significant release of glucose into the blood, which is the manifestation of the acute phase of the stress response in the first group. A prolonged exposure to copper acetate led to depletion of energy reserves and a decrease in the deposition of glucose in the liver, which is manifested by a significant decrease in plasma glucose levels in experimental animals after 4 months of the experiment.
Introduction
Due to the wide use of metals in the production of various products for agriculture, industry, medicine and everyday life, their compounds can penetrate the ecological environment and the human body through food chains [1–3].
In various fields of technology, allows are widely used with the use of heavy metals, including those that are part of copper-oxide galvanic cells and batteries [4, 5]. The result of widespread use of copper is its accumulation in various systems of the biosphere, the inclusion of copper and its compounds in the trophic connections of organisms and, as a consequence, the accumulation of copper in the organisms of animals and humans. In turn, this leads to a disruption of biochemical processes, which result in pathological abnormalities in the body [6–8].
The intake and impact of metal on the body and its bioavailability depend not only on its chemical properties, interactions with other metals, but also on its form [9–11]. Studies by X. Wu, Z. Liu, et al. indicated that a moderately high zinc in the diet (Zn:Cu — 40:1) significantly reduces the digestibility of Cu. In addition, a moderately high Cu content in the diet increases the Cu delay, without decreasing Zn absorption, and the moderately high Zn in the diet lowers the copper concentration in the plasma and the carbon dioxide release activity [12].
After entering the blood, copper is mainly distributed between the erythrocytes and plasma. In erythrocytes 60 % of copper is in the form of copper-zinc- metallic enzyme superoxide dismutase, the remaining 40 % are weakly bound to other proteins and amino acids [13]. This, in turn, can lead to a change in the balance in many biochemical processes in the body.
In the studies of O.V. Roshchina it is shown that the accumulation of copper causes inhibition of aminotransferase activity, which indicated changes in the regulation of protein metabolism towards an increase in the content of free amino acids, including aspartic, which serves as a substrate for gluconeogenesis [14].
E.P. Medyantseva noted that with the activity of lead and cadmium ions, the activity of alkaline phosphatase involved in the transport of phosphorus through the cell membrane is reduced and which was an indicator of phosphorus-calcium metabolism [15].
Pandey et al. found a decrease in GST activity and the level of reduced glutathione in the tissues of the spotted snakehead (Channa punctata) in the experiment on the exposure to a mixture of metals CuCl2 (50 μgZl), CdCl2, FeCl2 and NiCl2 [16].
The active effect of heavy metals, including copper on the biochemical indicators of blood and on the body as a whole, requires the development and study of corrective measures.
Recently, many scientific studies have appeared in which the authors use nanoparticles as correctors [17, 18]. So, in the studies of A. Slobodskov it is shown that copper nanoparticles reduce the level of bilirubin in the blood and increase the level of aspartate aminotransferase and alkaline phosphatase [17].
The second direction in development of tools for correcting the intake of copper in the body can serve the infusions of plant objects that have high resistance to these metals. In particular, M.R. Khanturin, R.R. Beisenova found that in acute intoxication with salts of zinc and copper, the extract of Sanguisórba officinalis normalized the parameters of peripheral blood [19].
One of the drugs that can be used to reduce the toxic effects of metals on the body was the infusion of Beta vulgaris's seeds. However, in the literature, we did not find any references to the use of extracts of this plant as a corrector when heavy metals enter the body.
Purpose of the study.
The purpose of this study was to study the effect of the infusion of Beet vulgaris's seeds on the biochemical indices of rat blood after exposure to copper acetate at the maximum permissible concentrations.
Materials and methods
The studies were conducted in the laboratory of ecological and genetic studies of Biological and Geographical Faculty of academician Ye.A. Buketov Karaganda State University. During the research, the ethical principles set out in the European Community Directive (2010/63/EC) and the requirements of the World Society for the Protection of Animals (WSPA) were observed.
The experiments were carried out on 40 white random-bred male rats. The animals were of the same age and were kept in standard vivarium conditions with free access to food and water. The feed ration of the tested animals consisted of solid grain feed, vegetable supplements and water in a ratio of 10×30×20 g.
1 group — experimental group — a solution of copper acetate was added to the diet of the group for 1 month (n = 10). The second group, an experimental group, added a solution of copper acetate to the diet of rats for 4 months (n = 10). 3 group — experimental group — a solution of copper acetate was added to ration of rats for 4 months with correction by infusion of Beta vulgaris's seeds (n = 10). Fourth, control group, (n = 10) with standard feeding ration.
As a source of copper, a solution of copper acetate was used, which was prepared taking into account the volume of animal feed, water consumed and the total weight of the animals. The average consumed concentration of copper by the experimental animals was 0.01 g/kg, which is the maximum allowable concentration of consumption [20, 21]. A 60 % solution of acetate was added to the water, and 40 % to the feed, thus, complex penetration of copper into the body of the experimental animals was achieved 4. The duration of the experiment was four months. Weighing of animals was carried out once a month. At the end of the experiment, the control weighing was conducted, followed by decapitation and blood sampling for biochemical analysis. To determine biochemical parameters of blood, an automatic biochemical analyzer Sysmex KX-21 manufactured by RocheDiagnostics (Switzerland) was used.
The statistical processing of the results was carried out using the Microsoft Excel 2007 application package. Differences in the dynamics of biochemical parameters were calculated by comparing the mean values using the N.A. Plokhinsky table.
Results and discussion
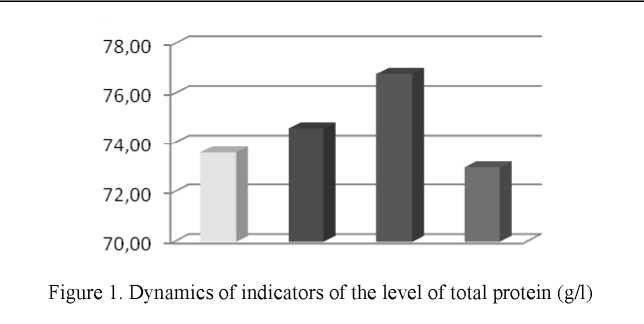
As a result of the study of biochemical parameters of blood, dynamic changes were established. The level of protein in the blood of rats of the first group was 73.6 ± 7.17 g/l, while in the second group the protein level tended to increase and amounted to 74.6 ± 4.54 g/l, which was higher in comparison with the control and the first group, although the differences were not significant (Fig. 1).
Comparison of the level of total protein between the second and fourth group in the fourth month of the experiment showed that in the fourth group this indicator was 73.0 ± 5.44 g/l, which is higher than the level of the total protein of the second group by 2.2 %
The application of beetroot's seed infusion led to an increase in the total protein index. Thus, the level of total protein in the third group was 76.8 ± 4.04 g/l, which was 5.2 % higher than in the control and 2.9 % higher than in the second group. The calculation of the significance of the differences between the second, third and fourth groups showed absent of no significant deviations.
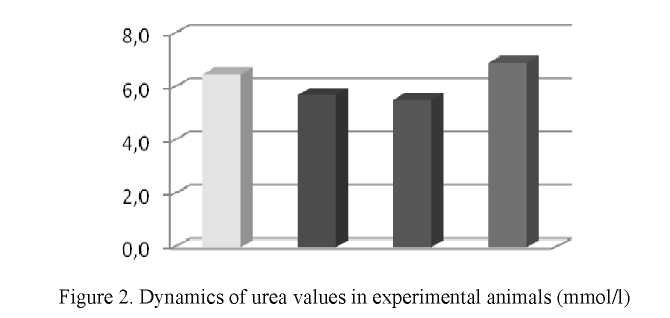
A comparative analysis of urea levels in the blood revealed that in the fourth month of the experiment (group 2) the urea level decreased by 12 % (5.7 ± 0.4 mmol/l) compared to the first month of the experiment (6.5 ± 0, 82 mmol/l) (1 group) (Fig. 2). When comparing the urea level of the first and second groups with the control group (6.92 ± 0.45 mmol/l) (group 4), it can be noted that in the second group the urea level was lower by 17.6 % and had a significant difference from of the control values (p ≤ 0.01).
A comparison of urea levels in the second and third groups showed that the level of urea in the second group is 3.2 % higher than that in the third group (5.5 ± 1.15 mmol/l). The difference in urea levels between the third and fourth group was 20.2 %. Calculation of the significance of differences in urea levels between the third and second, and also the third and fourth groups of experimental animals, did not reveal significant differences.
It should be noted that the use of the infusion of Beta vulgaris's seeds not only affects the significant decrease in urea levels in the blood of animals, but also causes a more individual reaction of the body to the complex effect of copper acetate and seed infusion, as evidenced by the protein and urea values.
Thus, the use of the infusion of Beta vulgaris seeds after exposure to the copper acetate leads to a significant increase in the level of total protein in the blood and a decrease in the level of urea. Reduction of the level of urea in the blood of experimental rats may be a result of a decrease in the amount of ammonia released due to a decrease in the activity of processes of protein and amino acids destruction in the liver [22]. In addition, a decrease in urea levels may be due to a disruption in the formation of urea in the liver as a result of copper exposure and an increase in the activity level of reduced glutathione [16].
Analysis of the dynamics of blood creatinine levels in rats showed that in the first group the creatinine value was 46.1 ± 2.62 mmol/l, and in the second group — 52.6 ± 3.57 mmol/l. Thus, the amount of creatinine in the blood of the experimental rats of the second group increased (p < 0.05) compared to the first group (Fig. 3).
Reduced level of creatinine in the first group of rats may be due to a disruption of the skeletal and cardiac muscles work, since copper, being a natural zinc antagonist, when used in high amounts, can inhibit muscle contraction processes in which zinc plays an important role [23, 24].
In the fourth group of experimental animals, the level of creatinine in the blood was 82.0 ± 5.22 mmol/l. Calculation of the relative difference in the absolute values of the creatinine level in the blood showed that the creatinine value in the fourth group is higher by 35.8 % than in the second group of animals (p ≤ 0.001).
A comparative analysis of the level of creatinine between the third and second groups showed that in the third group the introduction of beetroot seed infusion increased the creatinine level to
- ± 6.95 mmol/l, which is 37.6 % more than in the second group (p < 0.01).
A comparison of the absolute values of the level of creatinine between the third and fourth groups revealed that the relative difference in the level of these indicators was 11.7 %.
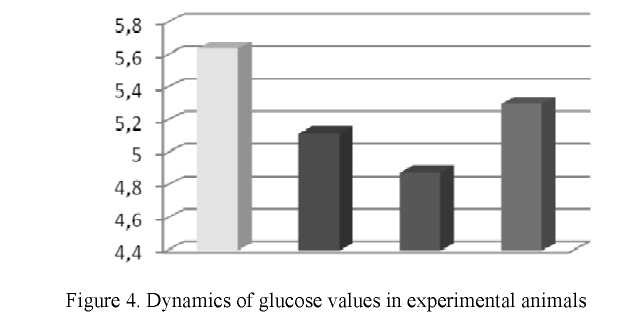
Evaluation of the blood glucose level of the experimental rats revealed that the glucose level in the second group tended to decrease. In the first group, the blood glucose level was 5.64 ± 0.34 mmol/l. In the second group, the glucose level in the blood decreased by 9.2 % and amounted to 5.12 ± 0.57 mmol/l (Fig. 4).
In the animals of the control group, the blood glucose level for the fourth month of the experiment was
- ± 0.49 mmol/l. Analysis of glucose levels revealed that in the second group, the glucose level was 3.4 % lower than in the fourth group. The glucose level in the third group was 7.9 % lower than in the fourth group.
Conclusion
Thus, as a result of the studies, it was found that biochemical blood values are directly dependent on the duration of exposure to copper of the body of experimental animals. Consumption of copper at a concentration of 0.01 g/kg of weight (TDI) significantly affects the indices of urea and creatinine towards their decrease, which indicates a change in metabolic processes, accompanied by disorders of protein metabolism. Apparently, the use of copper acetate, which has a toxic effect, is simultaneously a stress factor that caused a significant release of glucose into the blood, which is the manifestation of the acute phase of the stress response in the first group. A prolonged exposure to copper acetate leads to depletion of energy reserves and a decrease in the deposition of glucose in the liver, which is manifested by a significant decrease in plasma glucose levels in experimental animals after 4 months of the experiment. The main mechanism of replenishment of energy resources is the use of protein stored in the body, which in our case is accompanied by a decrease in protein metabolites in the blood. The solution from the infusion of Вéţа

vulgɑris's seeds as a correction, reduced the toxic effect of copper acetate and improves the processes of protein synthesis and glycolysis.
References
- Shimada, H., Nagano, M., Funakoshi, T., & Kojima, S. (1996). Pulmonary toxicity of systemic terbium chloride in mice. J. Toxicol. Environ. Health, 48, 1, 81–92.
- Lu, R., & Ni, J.Z. (2002). Mechanism of rare earth effect on liver. J. Chin. Rare Earth Soc., 20, 3, 193–198.
- Bogart, J.A., Lewis, A.J., & Schelter, E.J. (2015). DFT Study of the Active Site of the XoxF-Type Natural, CeriumDependent Methanol Dehydrogenase Enzyme. Chemistry. A European Journal, 21, 1743.
- Wieser, M.E., Holden, N., Coplen, T.B., B0hlke, J. K., Berglund, M., & Brand, W.A. et al. (2013). Atomic weights of the elements 2011 (IUPAC Technical Report). Pure and Applied Chemistry, 85, 5, 1047–1078.
- Nikolskii, B.P., et al. (1966). Spravochnik khimika [Reference book of the chemist]. (2nd ed.). Moscow: Khimiia [in Russian].
- Fedorova, N.N., Zhuravleva, H.F., & Zemkov, H.V. (2004). Razvitie patolohii vnutrennikh orhanov karpa v zavisimosti ot kontsentratsii sulfata medi [Development of pathology of internal organs of carp depending on the concentration of copper sulphate]. Vestnik Astrakhanskoho hosudarstvennoho universiteta — Bulletin of Astrakhan State University, 2(21), 156–161 [in Russian].
- Faizulin, A.I., & Zaripova, F.F. (2013). Vliianie tekhnohennoho zahriazneniia tiazhelymi metallami i urbanizatsii na morfofiziolohicheskie pokazateli populiatsii ozernoi liahushki Rana Ridibunda [Influence of technogenic pollution of heavy metals and urbanization on morphophysiological indices of the population of the lake frog Rana Ridibunda]. Vestnik Tomskoho hosudarstvennoho universiteta — Bulletin of Tomsk State University, 18(3), 834–837 [in Russian].
- Tinkov, A.A., & Nikonorov, A.A. (2013). Vliianie peroralnoho postupleniia solei zheleza i medi na initsiatsiiu adipoheneza i ateroheneza u krys linii Wistar [Effect of oral intake of iron and copper salts on the initiation of adipogenesis and atherogenesis in rats of the Wistar line]. Vestnik Orenburhskoho hosudarstvennoho universiteta — Bulletin of Orenburg State University, 12, 161, 173–177 [in Russian].
- Miinner. K., Simon, O., & Schlegel, P. (2006). Effects of different iron, manganese, zinc and copper sources (sulfates, chelates, glycinates) on their bioavailability in early weaned piglets. Tagung Schweine- und Geflügelemährung (28-30 November 2006): UniversitätHalle-Wittenberg, 27-35.
- Świątkiewicz, S., Koreleski, J., & Hong, D.Q. (2001). The bioavailability of zinc from inorganic and organic sources in broiler chickens as affected by addition of phytase. Journal of Animal and Feed Sciences, 10, 2, 317-328.
- Andersen, O. (2004). Chemical and biological considerations in the treatment of metal intoxications by chelating agents. Mini Reviews in Medicinal Chemistry, 4, 1, 11-21.
- Wu, X., Liu, Z., Guo, J., Wan, C., Zhang, T., & Cui, H., et al. Influence of dietary zinc and copper on apparent mineral retention and serum biochemical indicators in young mal mink. Biological Trace Element Research, 165, 1, 59-66.
- Mason, K.E. (1979). A conspectus of research on copper metabolism and requirements of man. The Journal of nutrition, 109, 1979-2066.
- Roshchina, O.V. (2010). Vliianie prirodnykh i antropohennykh faktorov na aktivnost fermentov syvorotki krovi chernomorskikh ryb (na primere morskoho ersha) [Influence of natural and anthropogenic factors on the activity of serum enzymes in the blood of Black Sea fishes (on the example of the sea ruff)]. Extended abstract of candidate's thesis. Moscow [in Russian].
- Medyantseva, E.P., Vertlib, M.H., & Budnikov, H.K. (1997). Iony metallov kak effektory fermentov [Metal ions as the effectors of enzymes]. Uspekhi khimii — The successes of chemistry, 67, 3, 252-260 [in Russian].
- Pandey, S., Parvez, S., & Ansari, R.A. et al. (2008). Effects of exposure to multiple trace metals on biochemical, histological and ultrastructural features of gills of a freshwater fish, Channa punctata Bloch. Chem. Biol. Interact., 174, 3, 183-192.
- Slobodskov, A.A. (2015). Vliianie vnutrimyshechnoho vvedeniia nanorazmernykh chastits medi na biokhimicheskie pokazateli krovi samok krys pri hestatsii [Influence of intramuscular introduction of nanosized copper particles on biochemical parameters of blood of female rats during gestation]. Nauchnoe obozrenie. Biolohicheskie nauki — Scientific Review. Biological Sciences, 1, 148-149 [in Russian].
- Notova, S.V., Timasheva, A.B., Lebedev, S.V., Sizova, Ye.A., & Miroshnikov, S.V. (2013). Elementnyi status i biokhimicheskii sostav krovi laboratornykh zhivotnykh pri vnutrimyshechnom vvedenii asparhinata i nanochastits medi [Elemental status and biochemical composition of blood of laboratory animals with intramuscular injection of aspartate and copper nanoparticles]. Vestnik Orenburhskoho hosudarstvennoho universiteta — Bulletin of Orenburg State University, 161, 12, 159-163 [in Russian].
- Khanturin, M.R., Beisenova, R.R., Taikina, S.S., & Asankhan, A. (2013). Izmeneniia tsitolohicheskikh pokazatelei krovi pri ostroi intoksikatsii tsinkom i mediu i na fone krovokhlebki lekarstvennoi [Changes in the cytological parameters of blood in acute intoxication with zinc and copper and against the background of a blood grooming drug]. Vestnik Karahandinskoho universiteta, Seriia Biolohiia. Meditsina. Heohrafiia — Bulletin of Karaganda University. Series Biology. Medicine. Geography, 70, 2, 40-45 [in Russian].
- Prikaz Ministra natsionalnoi ekonomiki Respubliki Kazakhstan ot 16 marta 2015 hoda. № 209 «Ob utverzhdenii Sanitarnykh pravil: «Sanitarno-epidemiolohicheskie trebovaniia k vodoistochnikam, mestam vodozabora dlia khoziaistvenno-pitevykh tselei, khoziaistvenno-pitevomu vodosnabzheniiu i mestam kulturno-bytovoho vodopolzovaniia i bezopasnosti vodnykh obektov»» [Order of the Minister of National Economy of the Republic of Kazakhstan of March 16, 2015 № 209 «On approval of the Sanitary Rules: «Sanitary and epidemiological requirements for water sources, water intake points for household and drinking purposes, domestic and drinking water supply and places of cultural and domestic water use and safety of water objects»»]. (2015, 16 March) Ministerstvo iustitsii Respubliki Kazakhstan — Ministry of Justice of the Republic of Kazakhstan, 10774 [in Russian].
- Voda pitevaia. Hihienicheskie trebovaniia i kontrol za kachestvom [Drinking water. Hygienic requirements and quality control]. (1985). HOST 2874–82. Moscow: Izdatelstvo standartov [in Russian].
- Marshall, J. (2000). Klinicheskaia biokhimiia [Clinical Biochemistry]. Moscow; Saint Petersburg: Binom, Nevskii dialekt [in Russian].
- Corny F., Lafargue P., & Guezennec C.Y. (1990). Zinc metabolism in the athlete: Influence of training nutrition and other factors. Sports Medicine, 31, 98, 577-582.
- Skalnyi, A.A., Tinkov, A.A., Medvedeva, Yu.S., & Alchinova, I.B. (2015). Vliianie vvedeniia tsinka na eho soderzhanie v tkaniakh laboratornykh zhivotnykh i aktivnost antioksidantnykh fermentov v syvorotke krovi pri fizicheskoi nahruzke [Influence of the introduction of zinc on its content in the tissues of laboratory animals and the activity of antioxidant enzymes in the blood serum under physical load]. Kazanskii meditsinskii zhurnal — Kazan Medical Journal, 96, 5, 862-866 [in Russian].