The present article considered a question about the self-treatment capacity of lakes, rivers and reservoirs in Akmola region based on oxygen indices. Furthermore, this study shows the influence of individual hydrochemical indicators (sulfates, chlorides, magnesium, ammonium salt, nitrogen nitrite, fluorides, common iron, zinc, manganese, copper) on self-treatment capacity of water bodies. The self-treatment capacity of reservoirs in Akmola region was assessed based on oxygen parameters such as the amount of dissolved oxygen in water and the biological oxygen demand (BOD5). Rivers and lakes were divided into six classes of self-treatment ability. In addition, we studied the associatedhydro-chemical components that can have impact on the purification potential of water in lakes and rivers. The results of research showed that the increased content of ammonium salt, nitrogen nitrite, total iron and copper suppress the self-treatmentcapacity of water. The majority of studied reservoirs by annual average indices have low self-treatment capacity. It was determined that the excess of the individual hydro-chemical components and the number of exceeding components with synergistic effect could have effect on the self-treatment capacity of water.
Introduction
The water resources of arid and steppe zones of the Republic of Kazakhstan are at high risk of extinction or degradation due to the number of climatic and anthropogenic factors. Rational use and restoration of most water bodies is an important task of the country. In this regard, knowledge and use of mechanisms of potential of water bodies in natural self-treatmentplay a significant role.
Most of the scientists believe that the support of balance between the photosynthetic activity of aquatic organisms and the processes of mineralization of organic substances formed in water reservoirs is the main mechanisms in the process of natural self-treatment of water bodies [1].
The indirect result of the photosynthetic activity of aquatic plants in water bodies can be the total oxygen content in water. At the same time, the destruction of organic substances can be assessed by the biological oxygen demand (BOD1, BOD5). The total amount of oxygen in the water is formed from dissolved atmospheric oxygen. It also can be formed as the product of photosynthesis by phytoplankton, phytobenthos, periphyton and macrophytes. The part of the oxygen is consumed on breath of hydrobionts and on participation in the chemical decomposition of inorganic and organic substances. Therefore, the total oxygen level in the water is always higher than BOD1, and in some cases BOD5. The ratio of these indicators can be taken as a criterion for assessment of the self-treatment capacity in the reservoir: if the intensity of photosynthesis in the reservoir is high, the water will be saturated with oxygen more actively, and the processes of destruction of impurities in the water will be faster [2].
The purpose of the present study was to conduct a comparative analysis of the self-treatment ability of the reservoirs in Akmola region based on oxygen indices and on the effect of individual hydrochemical components.
As the objects of the study we used the mean results of hydro-chemical studies in 2017 of the following rivers and lakes in Akmola region: Yesil river, Akbulak river, Sarybulak river, Bettibulak river, Zhabay river, Kypchakty river, Shagalaly river, Nura river, Nura-Yesil channel, Sultangeldy lake, Kopa lake, Zerendi lake, Burabay lake, Ulken Shabakty lake, Shchuchinsk lake, Kishi Shabakty lake, Karasie lake, Sulukol lake, Katarkol lake, Tekekol lake, Maybalyk lake and Lebyazhie lake. In total, we considered 8 rivers, 13 lakes, one reservoir and 1 channel.
Materials and methods
We formed the indicators of the amount of dissolved oxygen (R) and BOD5 in the studied lakes. Furthermore, we estimated the ratio of these indicators as characteristics of photosynthetic activity in the reservoir to its destructive ability: R/BOD5. If the ratio is high, the ability of self-treatment in water bodies will be high. But, when the ratio is low, the ability of the reservoir for self-treatment will also be low.
The lowest value of the ratio R/BOD5 was 2 in Maybalyk lake and the highest one was 13.3 in the Bettibulak river [3] (Table 1, Fig. 1).
Table 1 Average annual oxygen indicators of surface waters of Akmola region
|
No. |
Surface waters of Akmola region |
Amount of dissolved oxygen (R), mg/dm3 |
BOD5, mg/dm3 |
R∕BOD5 |
|
~¡~ |
Yesil river |
11.2 |
1.65 |
6.8 |
|
2 |
Akbulak river |
10.03 |
1.89 |
5.3 |
|
3 |
Sarybulak river |
8.45 |
3.73 |
2.3 |
|
4 |
Bettibulak river |
9.65 |
0.73 |
13.3 |
|
5 |
Zhabay river |
8.3 |
2.37 |
3.5 |
|
6 |
Kipshakty river |
6.76 |
2.58 |
2.7 |
|
7 |
Shagalaly river |
9.3 |
2.07 |
4.5 |
|
8 |
Nura river |
10.88 |
2.33 |
4.7 |
|
9 |
Nura-Yesil Channel |
11.13 |
2.19 |
4.7 |
|
10 |
Sultangeldy lake |
10.49 |
2.26 |
4.7 |
|
11 |
Vyacheslav reservoir |
11.92 |
1.64 |
7.3 |
|
12 |
Kopa lake |
9.19 |
4.04 |
2.3 |
|
13 |
Zerendi lake |
9.83 |
1.63 |
6 |
|
14 |
Burabay lake |
8.46 |
1.1 |
7.7 |
|
15 |
Ulken Shabakty lake |
8.72 |
0.84 |
10.4 |
|
16 |
Shchuchinsk lake |
9.06 |
0.97 |
9.4 |
|
17 |
Kishi Shabakty lake |
8.29 |
1.00 |
8.3 |
|
18 |
Karasie lake |
6.28 |
1.21 |
5.2 |
|
19 |
Sulukol lake |
6.02 |
2.48 |
2.5 |
|
20 |
Katarkol lake |
6.03 |
2.82 |
2.2 |
|
21 |
Tekekol lake |
7.59 |
1.11 |
6.9 |
|
22 |
Maybalyk lake |
5.47 |
2.65 |
2.0 |
|
23 |
Lebyazhie lake |
3.44 |
1.47 |
2.3 |
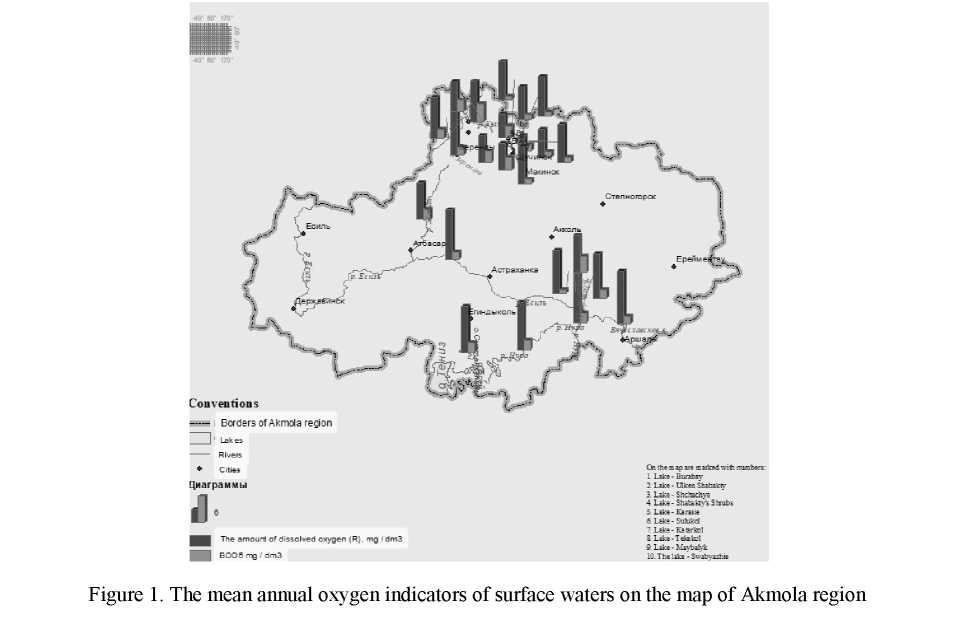
The variation number was made for the convenience of the approximation and it was divided into 6 classes (Table 2). The size of the variation class was 1.8.
Table 2
Classes of water bodies based on self-treatment ability
|
Class |
RZBOD5 |
f |
Ability to self-treatment |
|
I |
2-3.8 |
8 |
Very weak |
|
II |
3.9-5.7 |
6 |
Weak |
|
III |
5.8-7.6 |
4 |
Average |
|
IV |
77-9.5 |
3 |
Average |
|
V |
9.6-11.4 |
1 |
Good |
|
VI |
11.5-13.3 |
1 |
High |
Results and discussions
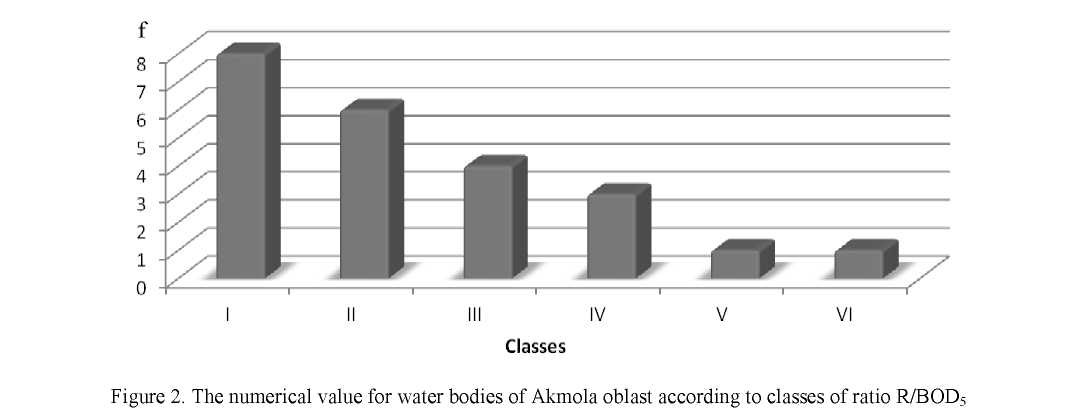
Figure 2 shows the results of the study, where we can see that based on annual indicators, the lakes with low self-treatment capacity dominate among the other studied lakes: class I (R/BOD5 ratio is 2–3.8) takes 30.5 % of all reservoirs. This class of water bodies includes Sarybulak river, Zhabay river, Kypshakty river as well as Kopa lake, Sulukol lake, Katarkol lake, Maibalyk lake and Lebyazhie lake.
Based of self-treatment capacity 26 % of the studied water bodies belongs to class II: Akbulak river, Shagalaly river, Nura river, Nura-Yesil Channel, Sultangeldy lake and Karasie lake.
17.4 % of water bodies were referred to Class III (R/BOD5 of 5.8 to 7.6): Yesil river, Vyacheslav reservoir and Tekekol lake.
Class IV (R/BOD5 77–9.5) contains 13 % of all lakes and rivers: Burabay lake, Shchuchinsk lake, Kishi Shabakty lake.
Both class V (R/BOD5 9.6–11.4) with Ulken Shabakty lake and class VI (R/BOD5 1.5–13.3) with Bettybulak river contain 4.3 % of all water bodies.
Table 3 provides information about the pollutants that effect on the photosynthetic activity of hydrophytes and depress the activity of saprobionts in the IV, V and VI classes of reservoirs with low selftreatment capacity.
As we know, threshold limit value (TLV) in natural waters can be accessed from the point of view of human toxicology. Nevertheless, the indicators toxic threshold of the same substances on biocenosis have still not been applied. Usually toxic threshold is determined for each species individually. Taking into account this fact, we conditionally applied the TLV for control the excess content of certain elements [4].
It can be noted from the data above, the reservoirs with low self-treatment capacity show high content of ammonium salt, fluorides, iron, zinc, manganese, copper in water.
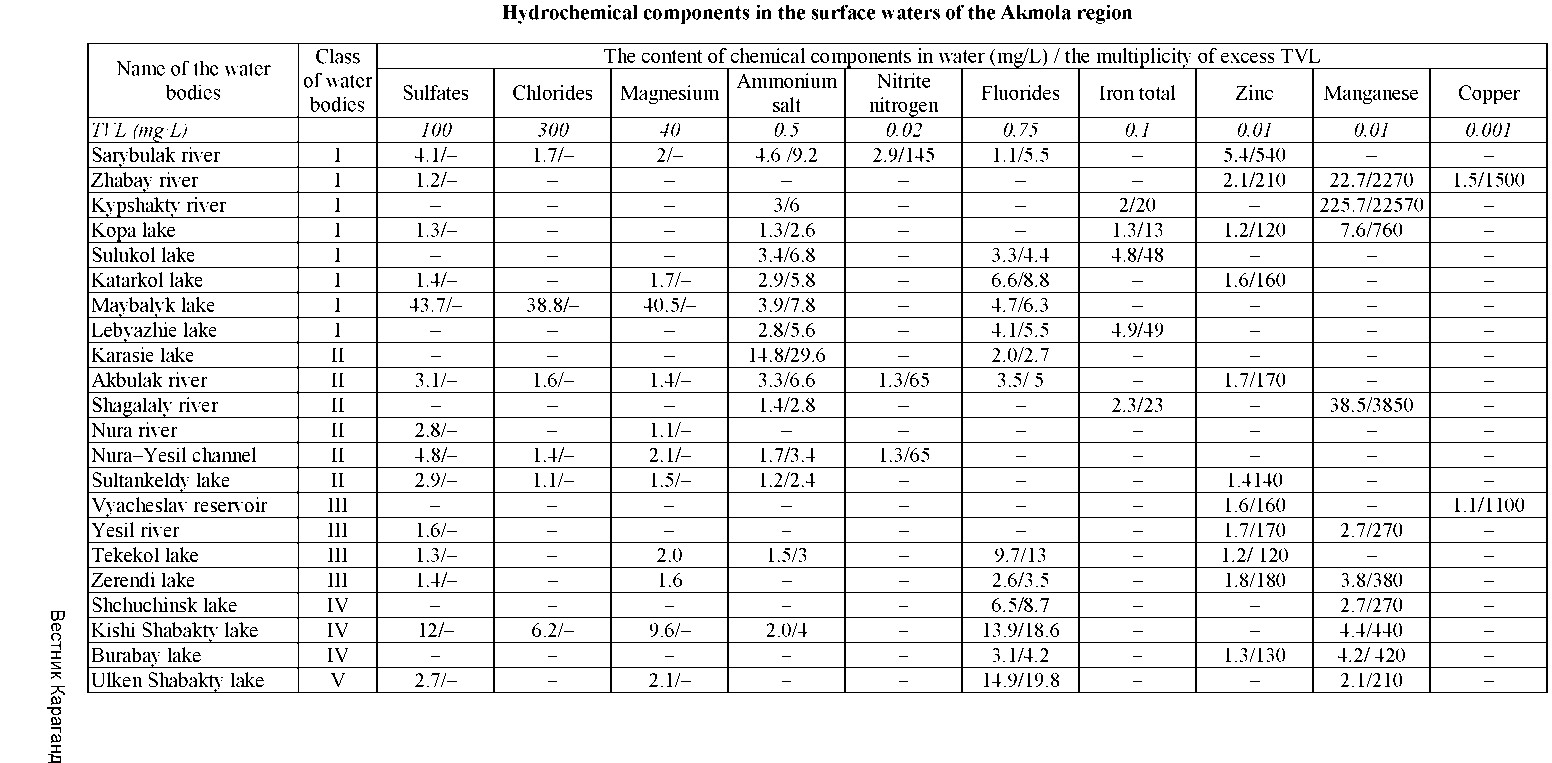
In the reservoirs of the I class the content is exceeded by 3-4 components as ammonium salt (9.2 TVL), nitrite nitrogen (145 TVL), fluorides (5.5 TVL), zinc (540 TVL) in the Sarybulak river. In the Zhabay river was the mean excessive concentrations of zinc (210 TVL), manganese (2270 TVL) copper (1500 TVL). There was excess of ammonium salt (6 TVL) iron total (20 TVL), manganese (22570 TVL) in Kypshakty river. In Kopa lake ammonium salt (2.6 TVL), iron total (13 TVL), zinc (120 TVL), manganese (760 MPC) were exceed, while in Sulukol lake there was excess of ammonium salt (6.8 TVL), fluorides (4.4 TVL), iron total (48 TVL). Katarkol lake demonstrated the high content of ammonium salt (5.8 TVL), fluorides (8.8 TVL), zinc (160 TVL). The multiplicity of excess ammonium salt (7.8 TVL), fluorides (6.3 TVL) were recorded in Maybalyk lake. There was excessive content of ammonium salt (5.6 TVL), fluorides (5.5 TVL), iron total (49 TVL) in Lebyazhielake.
The reservoirs of the II class also showed high concentrations of the studied components, they contained from 2 to 4 elements. For instance, in the Akbulak river there was the excess of TVL by 4 components, however, this excess was relatively low in comparison with those in reservoirs of the I class: Karasie lakehad excess of ammonium salt (29.6 TVL), fluorides (2.7 TVL). Akbulak river showed high content of ammonium salt (6.6 TVL), nitrite nitrogen (65 MPC), fluorides (5TVL) and zinc (170 TVL). There was excess of ammonium salt (2.8 TVL), iron total (23 TVL) and manganese (3850 TVL) Shagalaly river. Nura- Yesil channel had high content of ammonium salt (3.4 TVL) and nitrite nitrogen (65 TVL). There was excess content of ammonium salt (2.4 TVL) and zinc (140 TVL) in Sultankeldy lake.
The average treatment capacity of III and IV class reservoirs was also accompanied by excess of TVL on some components. For example, Vyacheslav reservoir showed a multiplicity of excess of zinc (160 TVL) and copper (1100 TVL). In the Yesil river was excess of zinc (170 TVL) and manganese (270 TVL). Lake Tekekol had relatively high concentration of ammonium salt (3 TVL), fluoride (13 TVL) and zinc (120 TVL). Zerendi lake demonstrated high content fluorides (3.5 TVL), zinc (180 TVL) and manganese (380 TVL). The content of fluoride (8.7 TVL) and manganese (270 TVL) was also high in Shchuchinsk lake. Kishi Shabakty lake exceed the amount of ammonium salt (4 TVL), fluoride (to 18.6 TVL) and manganese (440 TVL). There was exceed concentration of fluorides (4.2 TVL), zinc (130 TVL), manganese (420 TVL) in Burabay lake.
The water bodies with high level of self-treatment capacity represent a comparative interest (V and VI classes). In comparison with I and II classes, they showed less excessive concentrations of selected elements: Ulken Shabakty lake showed the excess of fluoride (19.8 TVL) and manganese (210 TVL). There was the excess of zinc (140 TVL) and manganese (400 TVL) in the river Bettibulak.
It should be noted that the reservoirs of V and VI classes did not show contamination on ammonium salt, nitrogen nitrite, iron total and copper.
Bettibulak river from VI class with the highest self-treatment capacity demonstrated the excess of TVL only for zinc and manganese. The elements are not involved in the inhibition of the photosynthetic process and do not inhibit the activity of heterotrophic bacteria.
The observed lakes showed a low content of sulphate ions, chloride and magnesium and practically did not exceed the TVL.Therefore, their effect on the self-treatment capacity of these reservoirs was excluded.
Thus, among the many factors which can have effect on the self-treatment capacity of water bodies we can note the influence of high content of such chemical components as ammonium salt, nitrite nitrogen, iron and copper. However, the concentration sulfates, chlorides, magnesium, increased zinc and manganese in the water had no effect on the treatment capacity.
Conclusions
- From the 23 reservoirs in Akmola region 60.8 % of rivers and lakes (I-II classes) have a weak selftreatment capacity. The average (satisfactory) self-treatment capacity wasin 30.4 % of reservoirs (III-IV classes); good self-treatment capacity was in 8.6 % of reservoirs (V-VI classes).
- The excess content of individual hydro-chemical components and the amount of exceeded components with synergistic effect can influence on the self-treatment capacity of surface waters.
- The increased content of ammonium salt, nitrite nitrogen, iron and copper can lead to the inhibition of the self-treatment capacity of reservoirs.
- Sulphates, chlorides, magnesium, increased content of zinc and manganese do not have particular influence on self-treatment capacity of water.
References
- Bulon, V.V., & Nikulina, V.N. (1976). Rol fitoplanktona v protsessakh samoochishcheniia v vodotokakh [The role of phytoplankton in the processes of self-purification in watercourses] Hidrobiolohicheskie osnovy samoochishcheniia vod — Hydrobiological basis of water self-purification. Leningrad: Zoolohicheskii institut AN SSSR, 15–24 [in Russian].
- Fedorov, V.D., & Kapkova, V.I. (2006). Prakticheskaia hidrobiolohiia. Presnovodnye ekosistemy [Practical hydrobiology. Freshwater ecosystems]. Moscow: PIM [in Russian].
- Informatsionnyi biulleten o sostoianii okruzhaiushchei sredy Respubliki Kazakhstan za 2017 hod [Information bulletin on the state of the environment of the Republic of Kazakhstan for 2017]. kazhydromet.kz/ru/p/ekologia. Retrieved from https://kazhydromet.kz/ru/p/ekologia [in Russian].
- Moiseenko, T.I. (2009). Vodnaia ekotoksikolohiia [Water ecotoxicology]. Moscow: Nauka [in Russian].