SUMMARY
Intraoperative neuromonitoring represents an opportunity to observe the state of the the nervous system structures during SurgicaIinterventionandtodefinei Teduceand Preventneurologicaldeficitspriortothesurgeryjntraoperativeneuromonitoring remains important, as the surgeon has no neuronavigation information during the operation to remove tumors/ Moreover, constantly changing structures can reduce the quality of the manipulations performed. Therefore, the neurophysiologist who also participates during the operations on the spinal cord "со-work" with the neurosurgeon closely in order to establish the physiological limits of resection, to prevent mechanical damage to the spinal cord and its ganglia: overstretch, rupture, trauma, detection of damage to the vessels in proper time and thereby preventing potential iatrogenic lesions.
41 operations were performed using Nim Eclipse intraoperative neuromonitoring by Medtronic at the Almaty City Clinical Hospital No. 7, This article describes neuromonitoring methods and good results confirming an obtained effectiveness. A total intravenous anesthesia without myorelaxants was performed according to the international standards Ofanesthesiologists. A close interaction of neurophysiologists, neurosurgeons, anesthesiologists is required in order to improve postoperative results and reduce the risks of neurologic lesions:
Reducing the frequency of intraoperative complications is one of the topical issues of modem neurosurgery. Modem neurosurgical technical equipment helps to prevent and detect abnormalities of the nervous system during the operation, to predict potential complications. Intraoperative neuromonitoring (hereinafter referred to as “I0NM”) allows real-time evaluation of the integrity of Stmctures at different levels of the National Assembly. This is the gold standard for operations in various areas of the spine: with degenerative diseases, with deformities, removal of spinal cord tumors or trauma complication.
In this article, we performed an analysis of modem representations and possibilities of IONM use according to the literature as well as we took our own data taken during spinal surgical interventions at the City Clinical Hospital No. 7 in Almaty.
The purpose of the study: to study results obtained from applying intraoperative neuromonitoring in the last three years during the spinal cord operations at the City Clinical Hospital No. 7 in Almaty.
Material and methods: since October 2014, the use of IONM on the Nim Eclipse device of Medtronic (USA) was introduced at the City Clinical Hospital No. 7. This allowed recording of all necessary neurophysiological parameters for various types of operations.
The choice of exact IONM modalities was made by a neurophysiologist, according to the protocol of preoperative access planning as well as with consideration of the scope of surgical intervention done by the neurosurgeon.
Given the sensitivity of the conduct of the I0NM, the anesthetic aid did not use long-acting muscle relaxants, the main stage of anesthesia was maintained on a combination of propofol with fentanyl. According to the international standards of The LAFS (Los Angeles Anesthesia Friendliness Scale) for anesthesiology, patients undergoing IONM undergo total intravenous anesthesia without the use of muscle relaxants.
According to the analysis of the results, 41 surgical interventions with the use of IONM were performed. In particular, installation of stabilizing Stmctures in degenerative-dystrophic changes of the spinal column was as follows - 13; removal of extra- and intramedullary neoplasms - 28 in total. The age of patients varied from 18 to 77 years. Each patient before the operation performed neurophysiological monitoring, analysis of the patient’s neurological status, MRI data, and then on intraoperative monitoring regimes with an individual approach in each case. The choice of the modality of IONM in operations on the spinal cord was caused by potentials and myography. Continuous monitoring of the conductivity of the spinal cord, spinal roots and peripheral nerves was performed.
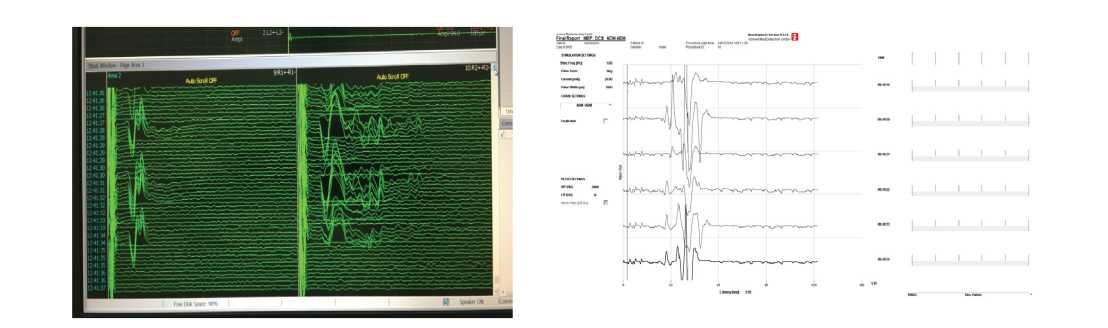
A peripheral receptors response from SSEP (somatosensory evoked potentials) stimulated by electrical impulse was obtained by excitation of the brain sensory cortex (Figure 1). Reducing the amplitude and increasing the latency of the SSVP by 50% was an alarm signal for damage to the Stmctures of the spinal cord. In our case, there was no significant reduction in the indices. Rhythmic stimulation of the SSEP was carried out from each point ⅛e initial values and then continuously until the necessary alternately to the main stage of the operation to determine manipulations were completed.
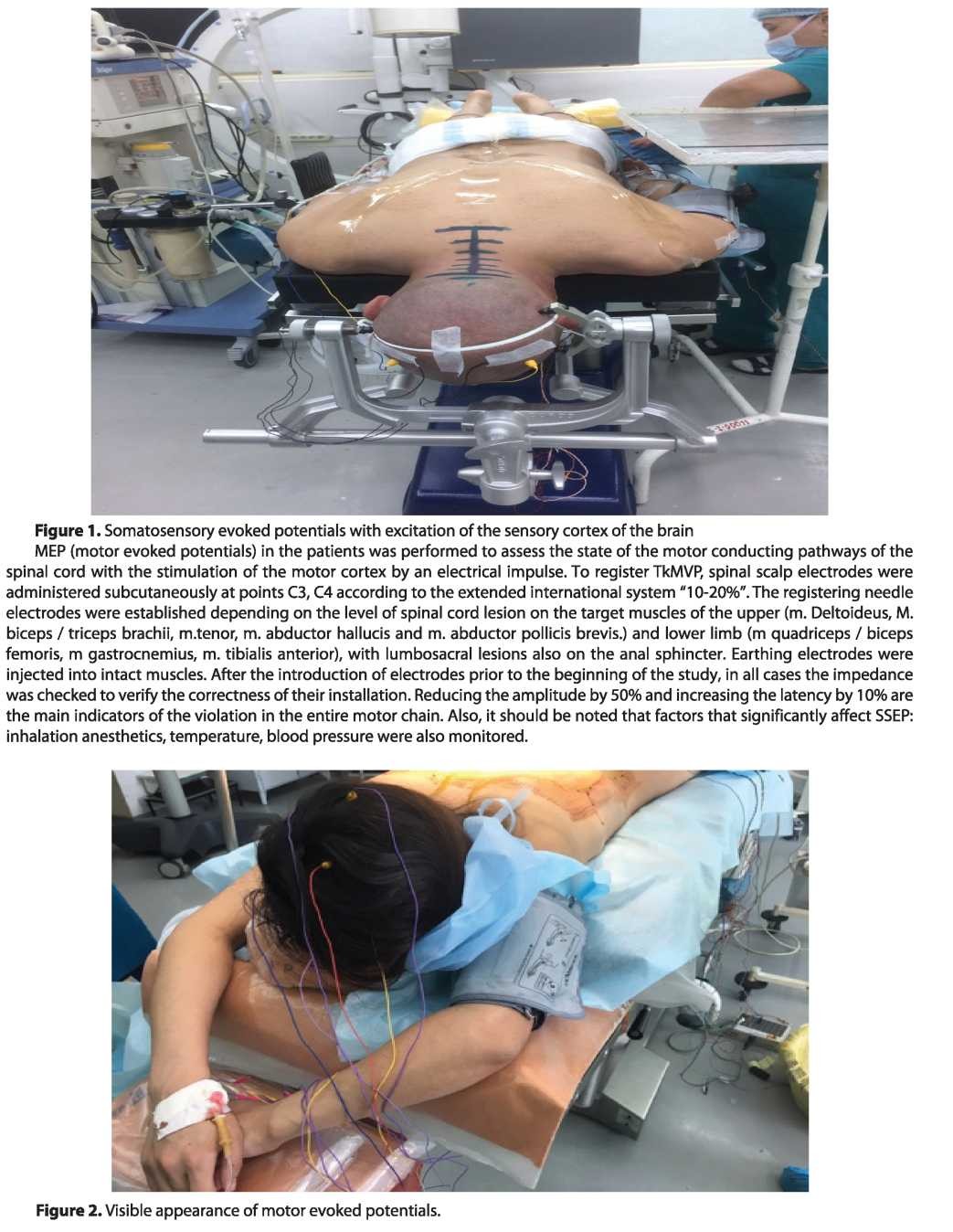
Figure 3. Stages of a persistent fixation.
Direct stimulation by electric impulse or direct mechanical action.
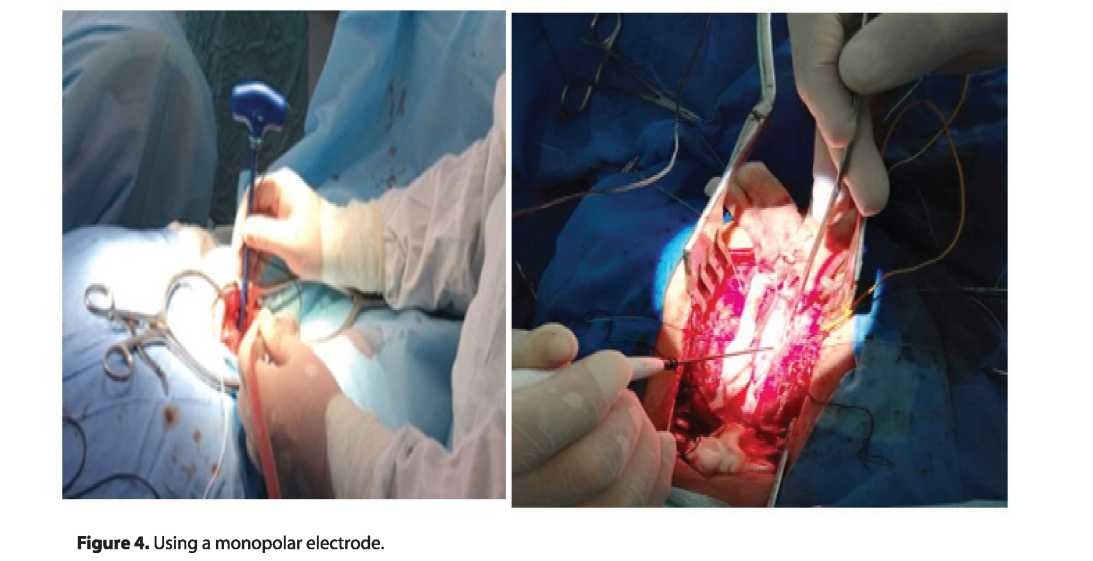
The advantage of the motor response in peripheral stimulation is that it comes with less latency (faster) than with the registration of profit centers. Registration is performed between two needle electrodes mounted on the target muscles, depending on the level of damage. A neurosurgeon using a bipolar or a monopolar probe can accurately determine the roots of the spinal cord and assess the correct location of the screw in the body of the vertebra according to international visualization standards developed by neurophysiologists (Figure 3). If the implanted screw was completely positioned in the bone, it was furthershield ed and the electrical stimulation of the screw does not activate the nerve. But if the medial wall of the vertebral body was damaged, a conductor with low resistance between the screw and surrounding tissues is formed. And when the current was applied to the screw, the nerve root was stimulated, the corresponding muscles contracted, and the MVP was recorded.
In this case, a stimulating bipolar and (or) monopolar electrode was used, the current strength was from 7 to 15 mA (Figure 4). Trigger EMG was also used to assess the correct position of transpedicular screws when installing stabilizing structures. In this case, the stimulus strength was 3-5 mA, the duration was 200 ms.
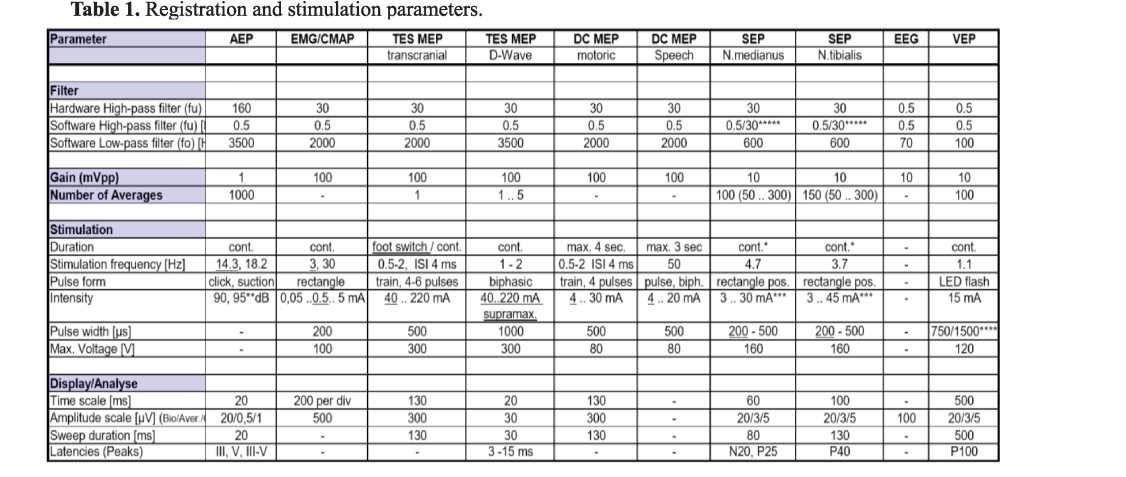
The parameters of registration and stimulation of intraoperative neurophysiological neuromonitoring were performed according to international standards, reflected in Table 1.
, ORCID 0000-0002-3190-2395
Results of the study and their discussion:
During intraoperative neuromonitoring, minor changes in bioelectrical indices were noted, all stages of operative intervention were performed according to preoperative planning. During the operation, correction of the surgeon’s manipulation was performed with:
- a stable decrease in the amplitude of evoked potentials below the baseline by 50%, an increase in latency of 10% (compulsory comparison of data with anesthesia, hypothermia, arterial pressure);
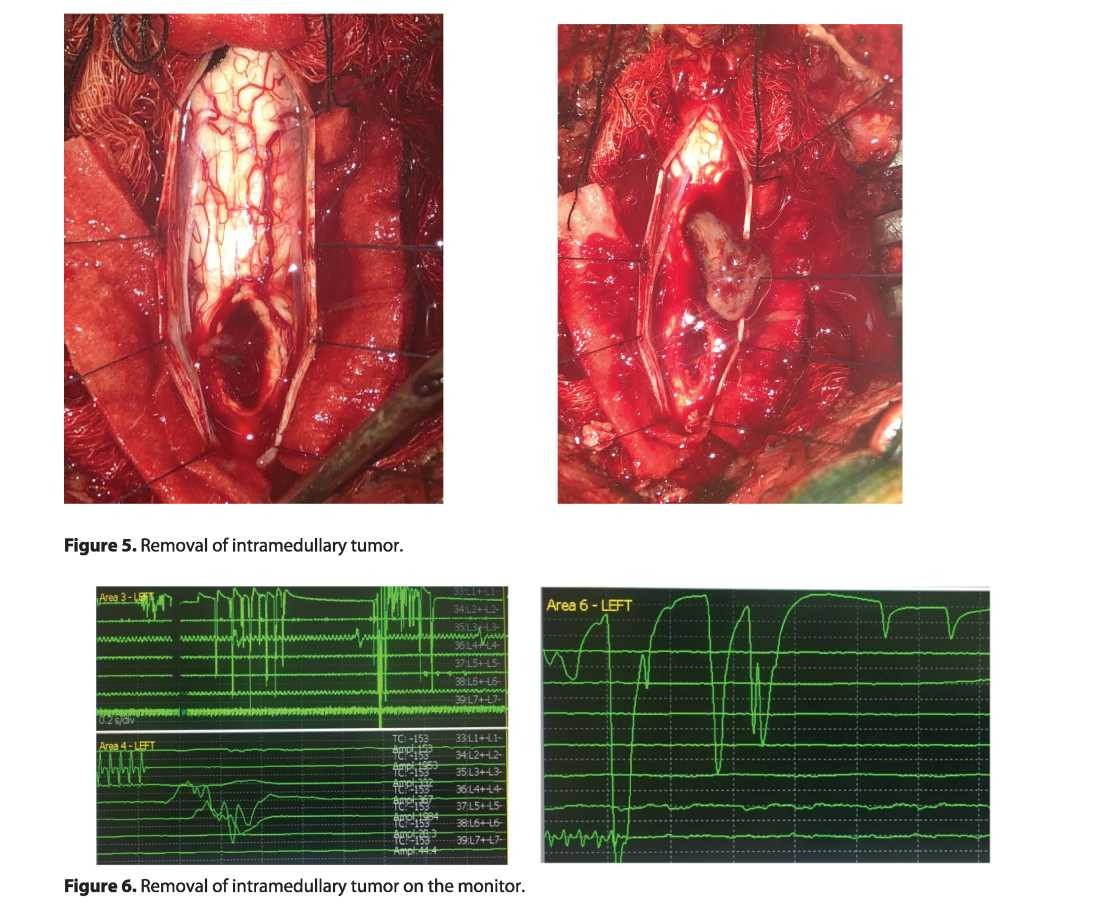
- a stable М-response with stimulation of a transpedicular screw with a current strength of 3-4 mA (oscillations of SSVP and TcMVP up to 30% were considered acceptable). In the case of irritation of motor fibers, M-responses to EMG were obtained, disappearing after the termination of the manipulation. In 5 patients with degenerative-dystrophic changes of the spine, the amplitude of the SSVP was below 0.9 mV during the entire intraoperative period (while maintaining normal latency). Motor disorders of the conductor type developed in one case after surgery for the removal of intramedullary tumors with complete regression during 10-15 days of conservative therapy.
According to the literature, there are reports of false positive and false negative results of neuromonitoring, on average. These data represented approximately 4%, the most frequent causes of which are anesthesia, temperature in the operating room and blood pressure indicators. [14].
IONM is an additional effective method of intraoperative diagnostics, which allows to reduce the risks of postoperative complications and improve the results of surgical manipulations.
Conclusions: Intraoperative neuromonitoring:
- allows to monitor the functional consistency of motor portions of the conducting pathways of the spinal cord, peripheral structures, and helps to reduce the degree of motor and sensory disorders during the operation.
- helps to find the safest access when removing intramedullary spinal cord tumors and reduces the risk of postoperative neurologic disorders;
- Ensures control and prevention of intraoperative complications with predicting the course of the postoperative period;
- Identifies the anatomical structures within the operating field and contributes to an increase in the radical nature of the operative intervention and the safety of the submerged metal-fixed spinal column without the formation of iatrogenic neurological deficits;
- It is needed for the legal security of the operating surgeon due to the formation of the neurophysiological protocol and its application to the medical documentation.
REFERENCES
- Левина Г.Ю., Борисов A.C., Калинин A.A., Бывальцев В.А.. Анализ результатов применения интраоперационного нейромониторинга в центре нейрохирургии дорожной клинической больницы на станции Иркутск-пассажирский ОАО «РЖД». Бюллетень ВСНЦ СО РАМН, 2016, Том 1, №4 (ПО).- С. 46-50.
- Румянцев П.О. Интраоперационный нейромониторинг при операциях на голове и шее // Опухоли головы и шеи,-2012.-№ İ.-С. 32-36.
- Васяткина А.Г., Левин Е.А., Кобозев В.В., Орлов К.Ю., Опыт применения нейрофизиологического мониторинга при нейрохирургических операциях. Патология кровообращения и кардиохирургия. 2016. Т. 20. №3. С. 108-117.
- Jimenez J.C., Sani S., Braverman В., Deutsch H. Palsies of the fifth cervical nerve root after cervical decompression: prevention using continuous intraoperative electromyography monitoring. J. Neurosurg. Spine, 2005, 3 (2), 92-97.
- Kelleher M.O, Tan G., Sarjeant R,, Fehlings M.G. Predictive value of intraoperative neurophysiological monitoring during cervical spine surgery: Aprospective analysis of 1055 consecutive patients. J. Neurosurg. Spine, 2008, 8 (3), 215- 221.
- Lee J.J., Kim Y.I., Hong J.T., Sung J.H., Lee S.W., Yang S.H. Intraoperative monitoring of motor-evoked potentials for supratentorial tumor surgery. J. Korean Neurosurg. Soc., 2014, 56 (2), 98-102.
- Lieberman J.A., Lyon R., Feiner J. The efficacy of motor evoked potentials in fixed sagittal imbalance deformity correction surgery. Spine, 2008.-33 (13), E414-E424.
- 13. TamakiT., KubotaS. Historyofthe development of intraoperative Spinalcordmonitoring. Eur. SpineJ., 2007.- 16 (2), 140-146.
- Wiedemayer H., Sandalcioglu I.E., Armbruster W, Regel J., Schaefer H., Stolke D. False negative findings in intraoperative SEP monitoring: analysis of 658 consecutive neurosurgical cases and review of published reports. J. Neurol. Neurosurg. Psychiatry. 2004, (75), 280-286.
- Epstein N.E. A review of medicolegal malpractice suits involving cervical spine: What can we Ieam or change J Spinal Disord Tech. 2011. № 24. Р. 15-19.
- Deletis V., Sala F. Inttaoperative neurophysiological monitoring of the spinal cord during spinal cord and spine surgery: a review focus on the corticospinal tracts. Clin Neurophysiol. 2008. № 119 (2). P. 248-264.
- Epstein N.E. The need to add motor evoked potential monitoring to somatosensory and electromyographic monitoring in cervical spine surgery. Surg Neurol Int. 2013. № 4 (Suppl 5). P. 383-391.
- Gavaret M., Jouve J.L., PereonY. et al. Intraoperative neurophysiologic monitoring in spine surgery. Developments and state of the art in France in 2011.Orthop Traumatol Surg Res. 2013. № 99 (6 Suppl). P. 319-327.
- Kobavashi S., Matsuyama Y, Shinomiya K. et al. A new alarm point of ttanscranial electrical stimulation motor evoked potentials for intraoperative spinal cord monitoring: a prospective multicenter study from the Spinal Cord Monitoring Working Group of the Japanese Society for Spine Surgery and Related Research. J Neurosurg Spine. 2014. № 20 (1). P. 102-107.
- Morora N., Deletis V., Constantini S. et al. The role of motor evoked potentials during surgery for intramedullary spinal cord tumors. Neurosurgery. 1997. № 41 (6). P. 1327-1336
- Macdonald D.B., Skinner S., Shils J., Yingling C. Intraoperative motor evoked potential monitoring - a position statement by the American Society OfNeurophysiological Monitoring. Clin Neurophysiol. 2013. № 124 (12). P. 2291- 2316.
- Azabou E., Manel V., Andre-Obadia N. et al. Optimal parameters of ttanscranial electrical stimulation for intraoperative monitoring of motor evoked potentials of the tibialis anterior muscle during pediatric scoliosis surgery. Neurophysiol Clin. 2013. № 43 (4). P. 243- 250.
- Azabou E., Manel V., Abelin-Genevois K. et al. Predicting intraoperative feasibility of combined TES-mMEP and cSSEP monitoring during scoliosis surgery based on preoperative neurophysiological assessment. Spine J. 2013. № 13. 1464p.
- Capone F., Tamburelli F.C., Pilato F. The role of motor-evoked potentials in the management of cervical Spondylotic myelopathy. Spine J. 2013. № 13 (9). P. 1077-1079.
- Вишневский A.A., Посохина 0.B., Рыжова 0.E. и др. Возможности исследования соматосенсорных вызванных потенциалов при патологии позвоночника. Хирургия позвоночника. 2005. № 3. С. 101-110.
- Vishnevskiy А.А., Posokhina 0.V., Ryzshova О.Е. i dr. Vozmozshnosti issledovaniya Somatosensomykh vyzvannykh potencialov pri patologii pozvonochnika. Khirurgiya pozvonochnika. 2005. № 3. 101-110.
- Deiner S. Highlights of anesthetic considerations for intraoperative neuromonitoring. Eur Spine J. 2009. № 18. P. 1541-1547.
- Calancie B., Molano M.R. Alarm criteria for motor-evoked potentials: what’s wrong with the «presence-or- absence» approach? Spine. 2008. № 33 (4). P. 406-414.
- Lee J.Y., Hilibrand A.S., Lim M.R. et al. Characterization of neurophysiologic alerts during anterior cervical spine surgery. Spine (Phila Pa 1976). 2006. № 31.P. 1916-1922