SUMMARY
Aim of research is practical application of the given classification of a pathology closely related kidneys allotransplantate and revealing the value of a complex of morphological researches in dynamics. Materials and Methods: was performed retrospective descriptive study, the depth of 5 years from January 2012 till February 2016. Sample solid. Exclusion criteria cadaver kidney transplantation. The work was conducted in the archive of pathomorphologic laboratory of National Scientific Surgery Center named after A.N.Syzganov. Results and discussion: it was about 48 casesof kidney transplantation. 64,58% (n-31) cases is related transplantations. According to Banff-07 classification they have been divided into groups by investigated signs. Diagnostic categories of Banff-07 classification renal allograft biopsias was following: the norm 51,61 % (n-16), antibody associated rejection 3,22% (n-1), boundary changes 9,67% (n-3), acute rejection 9,67% (n-3), chronic nephropathy of transplantate 25,80 % (n-8), with inherent morphological signs.
Conclusion: future advances in analysis of renal allograft biopsies can already be predicted, and the classification and grading of acute∕active rejection will continue to evolve. The significance of specific morphologic findings-including glomerulitis, arteriolitis, and infiltrates with unusual cellular features-for acute and chronic allograft dysfunction and outcome will continue to be investigated. Emphasis in biopsy assessment will shift from diagnosis to prediction of later allograft function and outcome, potentially enabling early intervention. Results of research have shown that lifetime morphological research of kidney transplant biopsies allows to give full representation about dynamics of a current, a stage of pathological process, a qualitative and quantitative estimation.
According to the statement of Arthur Cohen, which is one of famous morphologist in the field of nephrology: the kidneys is mysterious and frightening organ for many medical students and young doctors, it is impossible so to probe or see kidneys simply, The anatomy and physiology, seems so difficult and difficult, clinical features of nephritic frustration are indirect and are not easily distinguished, and there are a secret language is necessary, used by nephritic pathologists. The anatomy of kidneys is a difficult and have structures with special type of blood supply by arteries and arterioles by the type of an ended vessels, If occurs occlusion, the kidney tissue, blood supplied them is exposed to an ischemia, and result of which is necrosis. Almost all grafts have these displays in various degree [1].
One of prominent aspects is clinical diagnostics and timeliness of carrying out punction biopsy for recognition and probably, prevention of development of certain symptomatics. At the elementary level, pathologists should see in kidneys bioptates, at least one of these infringements connected with nephritic insufficiency, proteinuria and hematuria. Biopsy ofkidney allograft should become ordinaryas only it works properly, in an equivalent to nephritic insufficiency [1].
Kidney transplant is the most common type of organ transplant and one of the most effective treatments for patients with end-stage renal disease. Management of patients with kidney transplant is a difficult clinical challenge and is often associated with a number of problems. Renal biopsy remains the gold standard by which essential diagnostic and prognostic information is obtained after kidney transplantation [1]. Biopsy methodologies have been devised to assess the acceptability of an organ before transplantation and to assess and predict renal allograft performance after implantation [2, 3].
Optimal method of treatment of patients with a chronic renal failure is heterotopic allotransplantation of a donor kidney and the result from such treatment directly depends on quality of receiving and the quality of the received donor kidney [1,2]. Living and cadaveric donors can be a source of donor kidneys for transplantation to recipients. In case of an organ donorship from the living donor quality of the received donor kidneys and respectively result of transplantation are more optimal in comparison with transplantation when receiving a donor kidney from the cadaver, such difference of results is explained by various duration and intensity of influence of the damaging factors which are followed throughout time as at their receiving, preservation, and at their transplantation. Intensity of ischemic damage of a parenchyma of donor kidneys can have various expression, and its expression depends on aggression of a vasoconstriction of the dosage of cardiotonic support depending on level, which is directly dictated by an initial condition of a hemodynamics of a donor's organism [3].
Standardization of renal allograft biopsy interpretation and reporting is necessary to guide therapy in transplant patients and to establish an objective end point for clinical trials of new antirejection agents [4]. The Banff-07 Classification of Renal Allograft Pathology is an international scheme recently developed to fill this needs. The classification, which originated in a meeting held in Banff, Canada on August 2 to 4, 1991, was published in 1993, has been clinically validated in numerous studies, and is now widely used by center pathologists and in large international trials of immunosuppressive agents. Subsequent meetings have been held in Banff every two years to refine the classification. For National Institutes of Health clinical trials, a modification of the Banff grading system, the Collaborative Clinical Trials in Transplantation classification was developed; this classification and a clinical validation study were published in late 2013 [5, 6].
There are two theories of the rejection development: -cellular theory (author is Peter Medawar): the process of graft rejection is immunologically dependent. The cells (easy to see on histological slides) were recognized and identified as a cause of tissue damage; humoral theory (author is Paul Terasaki): evidence that also in later periods after transplantation antibodies must participate in graft damage. Renal transplant biopsies uses for diagnosis of acute changes in renal function (acute rejection, drug toxic effects, infections), diagnosis of transplant-related changes in pancreas transplant, diagnosis of declining renal function over prolonged time (chronic rejection, recurrent diseases) and etc.
Approaches to renal transplant biopsy interpretation includes next positions: evaluation of all tissue components (glomeruli, interstitium, tubules, arteries and arterioles) and determination if changes are acute and/or chronic. There are standards for adequate renal biopsy: the cortex mandatory must present, at least 7 glomeruli, at least 2 arterial cross sections, not exclusively subcapsular, two cores are better than one for greater sensitivity. Tissue processing of renal transplant biopsies: light microscopy, immunofluorescence, immunoperoxidase C4d (may be limited) and electron microscopy if it is indicated.
The Banff classification characterizes five categories of renal allograft pathology: antibodymediated rejection; suspicious of acute rejection; acute rejection; chronic sclerosing allograft nephropathy; and other changes not considered due to rejection [7].
Aim of research is practical application of the given classification of a pathology closely related kidneys allotransplant and revealing the value of a complex of morphological researches in dynamics.
Materials and Methods: was performed retrospective descriptive study, the depth of 5 years from January 2012 till February 2016. Samplesolid. Exclusion criteria cadaver kidney transplantation. The work was conducted in the archive of pathomorphologic laboratory of National Scientific Surgery Center named after AN Syzganov-The age of the donor ranged from 17 till 57 years.
Antiischemic actions provided by pharmacological protection of an organ which will be extract-
Вестник АГИУВ № 3, 2016
ed according to the general demands of the maintaining the potential donor. At conditioning the hemodynamics at the potential organ donor was corrected by the cardiotonic medicine «Dofaminum» and monitoring the blood pressure level demanded various dosage of cardiotonic. It is known that Dofaminum in a dose to 10 mkg∕kg∕min stimulates postsynaptic beta 1-adrenoreceptors that causes positive inotropic effect and augmentation of minute volume of a blood, and also provides intensifying of a circulation in kidneys, the glomerular filtration and is stimulates removing of sodium ions and a diuresis. In high doses more than 10 mkg∕kg∕min. or more prevail stimulation of alfal-adrenoreceptors, there is a narrowing of renal vessels that in turn reduces earlier enlarged renal blood flow and a diuresis. For definition of a target dosage of Dofaminum counted intensity of its introduction to an organism of the donor, thus calculation was made on substance of the corresponding 1 kg of body weight of the donor and on rate of his introduction.
Extraction of donor kidneys at potential donors was made according to the standard scheme J.R.Ackermann. However the sequence of composition of solutions for primary perfusion of a kidney after their extraction was various. Options of sequences of solutions were such: either Custodiol solution, or a normal saline solution the incorporating Heparin divorced in the ratio 1:100 that is 1,0 ml of Solution of the Heparin on 100,0 ml of NaCl of 0,9 %, temperature of solution 14-16 C and after that further washing of kidneys with Custodiol.
Hypothermal pharmacocooling conservation of a donor kidney after its extraction from the potential organ donor, provides its initial washing from a blood the preserving solutions of relatives on ionic structure to intracellular liquid Collins’s solutions, Saks, SIUT and AO-2, Ross and Marshall, Bredshnayder. Despite the above-mentioned actions, measures referred on conservation of a donor organ have the specific features as the task of an organ washing from a blood lays down on the Conservating solutions having excess viscosity. As solution with the smallest viscosity in the studied work to conservation of a donor kidney and washing of a lumen of vessels of the finest order from a blood and bloody clots was applied the sequence of solutions the Heparin divorced in a normal saline solution in the ratio of 1:100 that is 1,0 ml of Solution of the Heparin on 100,0 ml of NaCl of 0,9% in the temperature range 14-16 C and after that further washing of kidneys Custodiol solution.
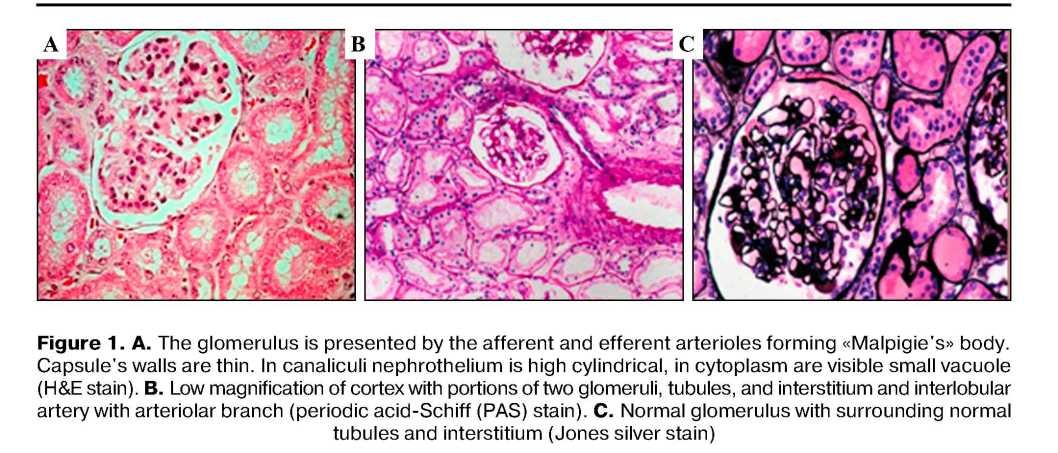
The main clinical criteria of quality of the carried-out actions at stages of extraction the donor kidneys referred on their «protection» namely at a stage of conditioning and a stage of their conservation were the beginning of excretory function after immediate transplantation and further conservation of these parameters with long-term adequate functioning. The condition of a nephrosites, convoluted tubules, vascular glomulus, degree of a dystrophia and a condition of a canaliculus of a kidney, existence of contents and fullness of a vascular glomulus of a kidney were the main morphological data of quality of extraction of donor kidneys, these data were regarded at 400 and 1000 multiple augmentation when coloring by a hematoxylin and eosine and the basal fuchsine of an azur 2 methylene blue terms of opportunity to trace such data were at the time of conservation of a donor kidney, for the 5th and 21st days after transplantation of this kidney.
The pathogenesis of acute antibody-mediated rejection includes: subsequent complement activation and adhesion of cells in result of necrosis of endothelial-cells, produced by platelet deposition and coagulation. It is denotes polymorphonuclear cells. Usually mononuclear cells adherence to the endothelium of the glomeruli is prodused, which process is accompanied by C4d deposition in the glomeruli and peritubular capillaries, in the peritubular capillaries between ghost outlines of the tubules.
Chronic allograft rejection is immune injury of the graft, developed due to a failure to maintain sufficient immunosuppression which controlled by residual antigraft antibodies and lymphocytes. Which is include a progressive decline in renal function, infiltration and invasion of the renal parenchyma by T-cells, and persistent infiltration of the interstitial space by T-cells and macrophages, proliferation of smooth-muscles and hyperplasia vessel walls, with formation a neointima; focal destruction and necrosis of internal elastic lamina, result of which is total vascular occlusion.
Acute antibody-mediated rejection is divided on: acute antibody-mediated rejection (type I: expression of positive C4d, minimal signs of inflammation, type II: expression of positive C4d, leukocytic infiltration, type III: expression of positive C4d, transmural arteritis), chronic active antibody-mediated rejection.
The pathogenesis of second theory mainly includes processing of endogenous and exogenous antigens by class I and class II of major histocompatibility complex pathways. The first one is endogenous antigens are digested by proteosomes into peptides and are leads into class I major histocompatibility complex. Exogenous antigensare degraded by endosomes and are leads into class II major histocompatibility complex. Assembly of the major histocompatibility complex within the cell'sendoplasmic reticulum leads for its transport through the Golgi apparatus and provides its expression on the cellsurface in connection with peptide, where the major histocompatibility complex -peptide complex connects with CD8+ CD4+ T lymphocytes. Next stage is activation and differentiation of T-lymphocytes from dendritic-cell maturation till activation and proliferation of effector T-cells with its active products.
T-cell rejection is divided on: acute T-cell rejection (Type IA: moderate intimal arteriitis;severe intimal arteriitis and moderate tubulitis; type IB: severe tubulitis. Type IIA: mild-moderate intimal arteritis. Type IIB: severe intimal arteritis. Type III: transmural arteritis) Clironic active T-cell rejection.
Results and discussion: it was about 48 cases. 64,58 % (n-31) cases is related transplantations of a kidney. According to Banff-07 classification they have been divided into groups by investigated signs. Diagnostic categories of Banff-07 classificationrenal allograft biopsiaswas following: the norm51,61 % (n-16), antibodyassociated rejection3,22 % (n-l), boundary changes 9,67 % (n-3), acute rejection 9,67 % (n-3), chronic nephropathy of transplantate 25,80 % (n-8), with inherent morphological signs.
The antibody-associated rejection, presumably mediated by antibodies is typically characterized by the accumulation of polymorphonuclear leukocytes in glomerular and peritubular capillaries in the early period after transplantation, followed by damage to the endothelium and capillary thrombosis (fig. 2).

However, the histological diagnosis of acute ABMR is still problematic, especially in cases with few outward symptoms [8, 9]. The studies using immunofluorescence to detect immunoglobulins (IgG, IgM and IgA), complement factors (C3, C4 and Clq) and fibrin are not very accurate [10].
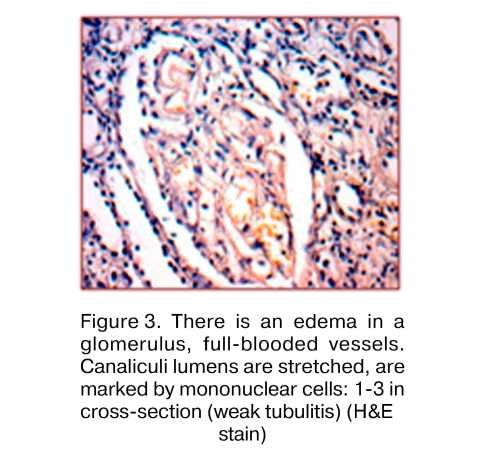
Boundary changes «suspicion5» on acute rejection [11]. This category is used when there is no intimal arteriitis, but the presence of foci of weak tubulitis.
In grafts undergoing acute rejection, T lymphocytes infiltrate and proliferate within the interstitial space, whence they invade renal tubules, causing tubulitis.Invading CD8 T lymphocytes, which have immunologic specificity for the allograft, cross the basement membrane of the tubule, where they proliferate and induce apoptosis of tubular cells [12]. Sublethally injured tubular cells can also transform from their native epithelial phenotype into primitive mesenchymal myofibroblasts, promoting interstitial fibrosis [13, 14]. Necrosis of tubular epithelial cells and basementmembrane rupture cause urinary leakage, graft dysfunction, and progressive tubular atrophy [15, 16]. The histologic characteristics of vascular rejection (also termed arteritis or endarteritis) include the infiltration of vessels by mononuclear cells, endothelial-cell apoptosis, and the synthesis of matrix proteins and collagens by intimal myofibroblasts.
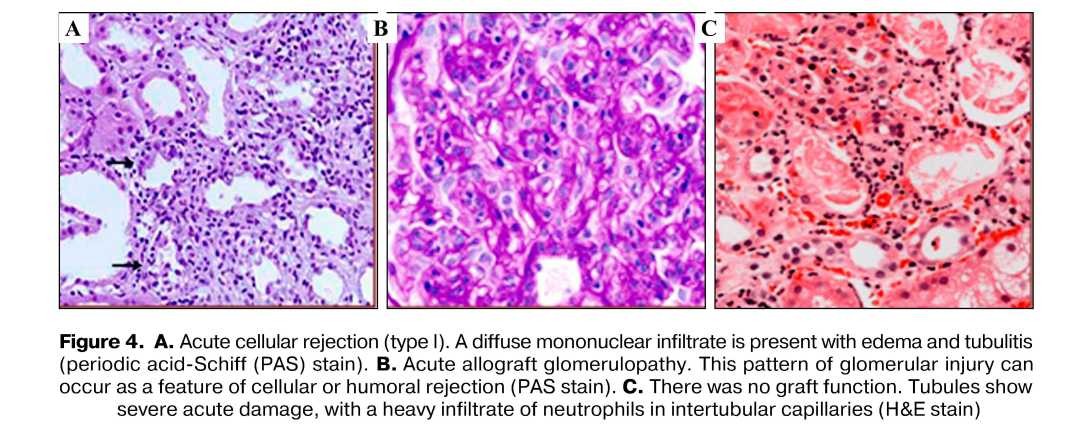
Chronic allograft rejection ongoing immune injury to the graft is due to a failure to maintain sufficient immunosuppression to control residual antigraft lymphocytes or antibodies [13]. Its features include a progressive decline in renal function, invasion of the renal parenchyma by T cells, and persistent infiltration of the interstitium by T cells and macrophages [17]. Occasionally, one also sees smooth-muscle proliferation and hyperplasia in vessels, forming a neointima; focal destruction of internal elastic lamina; and finally, vascular occlusion.
Conclusion: future advances in analysis of renal allograft biopsies can already be predicted, and the classification and grading of acute∕active rejection will continue to evolve. The significance of specific morphologic findings-including glomerulitis, arteriolitis, and infiltrates with unusual cellular features-for acute and chronic allograft function and outcome will continue to be investigated. Emphasis in biopsy assessment will shift from diagnosis to prediction of later allograft function and outcome, potentially enabling early intervention. Results of research have shown that lifetime morphological research of kidney transplant biopsies allows to give full representation about dynamics of a current, a stage of pathological process, a qualitative and quantitative estimation.
REFERENCES
- Hoffmann SC, Hale DA, Kleiner DE, et al. Functionally significant renal allograft rejection is defined by transcriptional criteria. Am J Transplant 2005; 5: 573-81.
- Li XC, Rothstein DM, Sayegh MH. Costimulatory pathways in transplantation: challenges and new developments. Immunol Rev 2009; 229: 271-93.
- Stratta R J, Sundberg AK Rohr MS, et al. Optimal use of older donors and recipients in kidney transplantation. Surgery 2006; 139: 324-33.
- Coniell LD, Smith RN, Colvin RB. Kidney transplantation: mechanisms of rejection and acceptance. Annu Rev Pathol. 2008; 3: 189-220.
- Furness PN, Taub N, Assmann KJ et al. International variation in histologic grading is large, and persistent feedback does not improve reproducibility. Am J Surg Pathol 2003; 27: 805-810.
- Terasaki PL Antibody response to homograft. I. Preliminary studies of the time of appearance of Iymphoagglutininsuponhomografting-AmSmg 1959; 11: 896-9. [Pubmed]
- Howie AJ. Problems with BANFF. Transplantation. 2002; 73(9): 1383-4, a comment on McCarthy GP, Roberts IS. Diagnosis of acute renal allograft rejection: evaluation of the Banff 97 Guidelines for Slide Preparation. Transplantation 2002; 73: 1518-1521
- Halloran PF. Immunosuppressive drugs for kidney Iiansplantation. N Engl J Med 2004; 351:2715-29.
- Sis B, Mengel M, Haas M, Colvin RB, Halloran PF, Racusen LC, et al. Banff '09 Meeting Report: antibody mediated graft deterioration and implementation of Banff working groups. Am J Transplant 2010; 10: 464-71. [Pubmed]
- Stegall MD, Gloor JM. Deciphering antibody-mediated rejection: new insights into mechanisms and treatment. Curr Opin Organ Transplant 2010; 15: 8-10.
- De Freitas DG, Sellare ɪ's J, Mengel M et al. The nature of biopsies with 'borderline rejection' and prospects for eliminating this category. Am J Transplant 2012; 12: 191-201.
- Bestard O, Nickel P, Cruzado JM, et al. Circulating alloreactive T cells correlate with graft function in longstanding renal Eansplant recipients. J Am Soc Nephrol 2008; 19: 1419-29.
- Bonsib SM, Abul-Ezz SR, Ahmad I, et al. Acute rejection-associated tubular basement membrane defects and chronic allograft nephropathy. Kidney Int 2000; 58: 2206-14.
- Kalluri R. EMT: when epithelial cells decide to become mesenchymal-like cells. J Clin Invest 2009;119:1417-9.
- Elshafie M, Furness PN Identification of lesions indicating rejection in kidney transplant biopsies: tubulitis is severely under-detected by Conventionalmicroscopy. Nephrol Dial Transplant 2012; 27:1252-1255.
- Robertson H., Kirby JA. Post-transplant renal tubulitis: the recruitment, differentiation and persistence of intra-epithelial T cells. Am J Transplant 2003;3: 3-10.
- Nankivell BJ, Borrows RJ, Fung CL, O'Connell PJ, Allen RD, Chapman JR. The natural history of chronic allograft nephropathy. N Engl J Med 2003; 349: 2326-33.