In this paper, the results of study of the cellulose biodegradation using cellulolytic microorganisms are given. Aspergillus awamori VUDT-2, Bacillus subtilis 82 and consortium consisted of named strains were used in the enzymatic hydrolysis. Morphological features of these microorganisms were described using optical microscopy and by inoculation of suspensions on meat peptone agar. Initially, cellulose-degrading activity of microorganisms was tested in the medium containing reagent grade cellulose. The cultivation was carried out on a shaker-incubator in 750 mL Erlenmeyer flasks at a temperature of 30 °C and a stirring speed of 200 rpm for 6 days. As a result, the consortium showed better cellulolytic activity and 49.2 % of total cellulose was degraded in the end of the experiment. Then, the growth conditions of the consortium were optimized in terms of temperature, stirring speed and the initial pH of the medium. For further experiments, the dried distillers grains with solubles (DDGS) after alcohol production was used as a source of cellulose-containing waste. The content of the cellulose in the DDGS is 20.78 %. Under the conditions of temperature 30 °C and the stirring speed at 200 rpm the biodegradation level of cellulose using consortium of cellulolytic microorganisms was 70.24 % after 6 days.
Introduction
From the all biopolymers with carbohydrate structure, cellulose is the most common organic substance in the world. Different natural materials such as cotton and wood are mainly constituted by cellulose. Annually, there are more than 75 billion tonnes of this compound produced worldwide [1].
The applications of cellulose-based materials have been described in uncountable number of research papers in the fields of manufacturing new materials and nano-composites [2–4], biomedical [5, 6], environmental [7, 8], bio energy and bio ethanol production [9–11] and many other. Another application is that cellu- lose-containing materials can be used as a source for hydrolytic production of carbohydrates.
The molecular structure of cellulose is a linear polymer chain, consisted of an hydroglucose units which are linked with β-1,4-glucoside bonds. The number of monomeric units in the macromolecules of cellulose can be more than many thousands [12]. Nowadays, cellulose conversion to carbohydrates is mainly conducted using acid and enzymatic hydrolysis. Generally, acid hydrolysis is carried out using sulfuric acid or hydrochloric acid, while enzymatic hydrolysis is occurred by catalyzing with cellulolytic enzymes [13].
To this time, there are many microorganisms that have been described to catalyze the hydrolysis of polysaccharides. Namely, they are strains of Bacillus pumilus, Pseudomonas sp., Trichoderma reesei, Tricho- derma harzianum, Penicilliume chinulatum, Aspergillus phoenicis, Phanerochaete chrysosporium, Aspergillus niger and other. All of them are considered as producers of cellulases [14].
Various types of cellulose-containing waste after pulp and paper industry as well as secondary products from sawmilling and wood processing, waste from agricultural crops and a countless number of wild plants can be used as a raw material for the hydrolytic production of carbohydrates. The cost, size of reserves and the possibility of their transportation to the area of hydrolytic production are the main criteria for the use of certain materials.
Thus, it should be noted that if even a small part of the listed sources is converted into useful products, it will give a very tangible and important renewable source of carbohydrates as well as starting compounds for microbiological synthesis.
At the present work, we carried out the enzymatic hydrolysis of cellulose using bacterial consortium, which is consisted of strains Aspergillus awamori VUDT-2 and Bacillus subtilis 82. The fraction of dried distiller grains with soluble (DDGS) is used as a source of cellulose-containing waste.
Materials and methods
The microbial cultures used in this study are Aspergillus awamori VUDT-2 and Bacillus subtilis 82, which are deposited in the official collection of the Branch of RSE “National Center for Biotechnology” under the Science Committee of Ministry of Education and Science of the Republic of Kazakhstan.
Bacillus subtilis 82 on Petri dishes with MPA forms rod-shaped colonies of white or yellowish color with a smooth or scalloped edge and a powdery surface. It is a gram-positive, aerobic bacterium, which is represented by single or associated cells. Spores are oval and located eccentrically, mostly closer to the center. According to the registration document, the optimum temperature for bacterial growth is 30 °C.
From morphological perspective, Aspergillus awamori VUDT-2 is a highly branched mycelium, divided by partitions. Conidia formed by micromycetes, covered with spikes. Conidia are round shape of brown color with a dark brown shade. On the medium with MPA, Aspergillus awamori VUDT-2 forms rounded colonies with a diameter of 18–20 mm with a smooth white edge and the surface is folded to the edges of the colony. The color is dark brown with an olive tint and exudate on the surface. According to the registration document, the optimum temperature for growth of Aspergillus awamori VUDT-2 is 30 ºC.
The samples of the cellulose-containing waste were obtained from malt distilling plant “Alfa Organic” LLP in Stepnogorsk. The content of cellulose in the DDGS is 20.78 %.
Pure cellulose and anthrone reagent were purchased from Sigma-Aldrich Co. Unless otherwise stated, all reagents were of analytical grade.
The pH was determined by the analyzer “Mettler Toledo Seven Multi S47-K”.
The concentrations of cellulose and reducing sugars were determined by anthrone method. Firstly, the sample with cellulose was digested with concentrated sulfuric acid. Then, anthrone reagent was added and the green color mixture was measured spectrophotometrically on Bio Mate 3S (Thermo Scientific, USA) at wavelength λ = 620 nm [15].
Method of serial dilutions was used to count the number of the colony forming units.
Cell counting of the aerobic fungi Aspergillus awamori VUDT-2 was carried out by the method described in [16], an approximate method of serial dilutions with the calculation of the power of the number of microorganisms.
The morphological, cultural properties of microorganisms were studied by inoculating suspensions on MPA medium with incubation at 30 °C for 48 hours. Microscopic investigations were performed on an Olim- pus 56BX microscope.
The following nutrient media were used in the experiments:
–Meat peptone broth (MPB), g/L: peptone — 10, glucose — 5, meat extract — 5, NaCl — 5, pH 7.2– 7.4.
–Meat peptone agar (MPA), g/L: peptone — 10, glucose — 5, meat extract — 5, NaCl — 5, agar — 20, pH 7.4–7.6.
– Cellulose containing medium (CCM), g/L: cellulose — 100, peptone — 5, yeast extract — 2, MgSO4 — 1, KH2PO4 — 1, pH 6.2.
–DDGS medium, g/L: DDGS —150, MgSO4 — 1, KH2PO4 — 1, pH 6.
Results
Natural cellulose is an insoluble, semi crystalline polymer (Fig. 1). Polymer alignment formed its crystalline sections and they held together by hydrogen bonding and Van der Waals interactions [17].
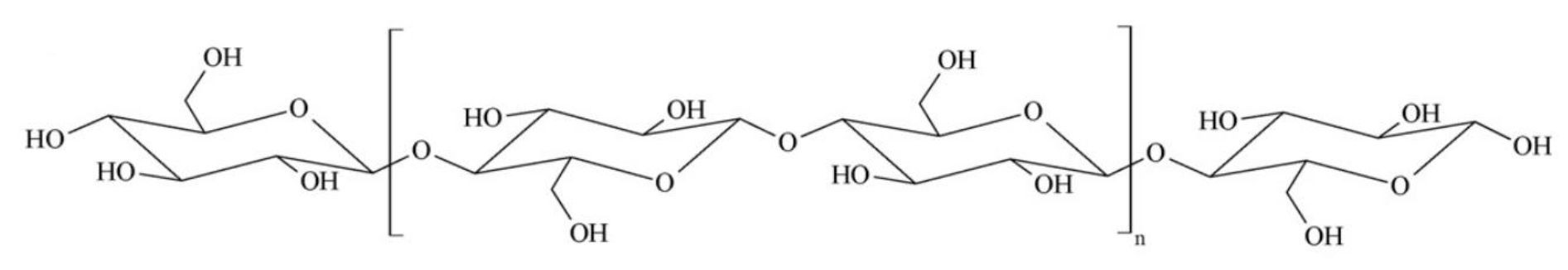
Figure 1. Chemical structure of cellulose
In nature, aerobic and anaerobic cellulolytic microorganisms degrade a major part of cellulose materials. Enzymatic degradation of cellulose occurs in several steps. Firstly, cellobiohydrolases acts on the reducing
81
and non-reducing ends and endoglucanases hydrolyze internal bonds, then b-glucosidase converts the cellobiose to glucose [18].
From the literature data [19, 20], both Aspergillus awamori and Bacillus subtilis are capable for producing cellulolytic enzymes.
At the present work, Aspergillus awamori VUDT-2 and Bacillus subtilis 82 were initially cultivated in the MPB medium. They were used to increase the reducing sugars because of their ability for bioconversion of cellulose. For this purpose, the inocula of Aspergillus awamori VUDT-2, Bacillus subtilis 82 and a consortium composed of strains of Aspergillus awamori VUDT-2 and Bacillus subtilis 82, were added in the amount of 10 % on the cellulose-containing medium.
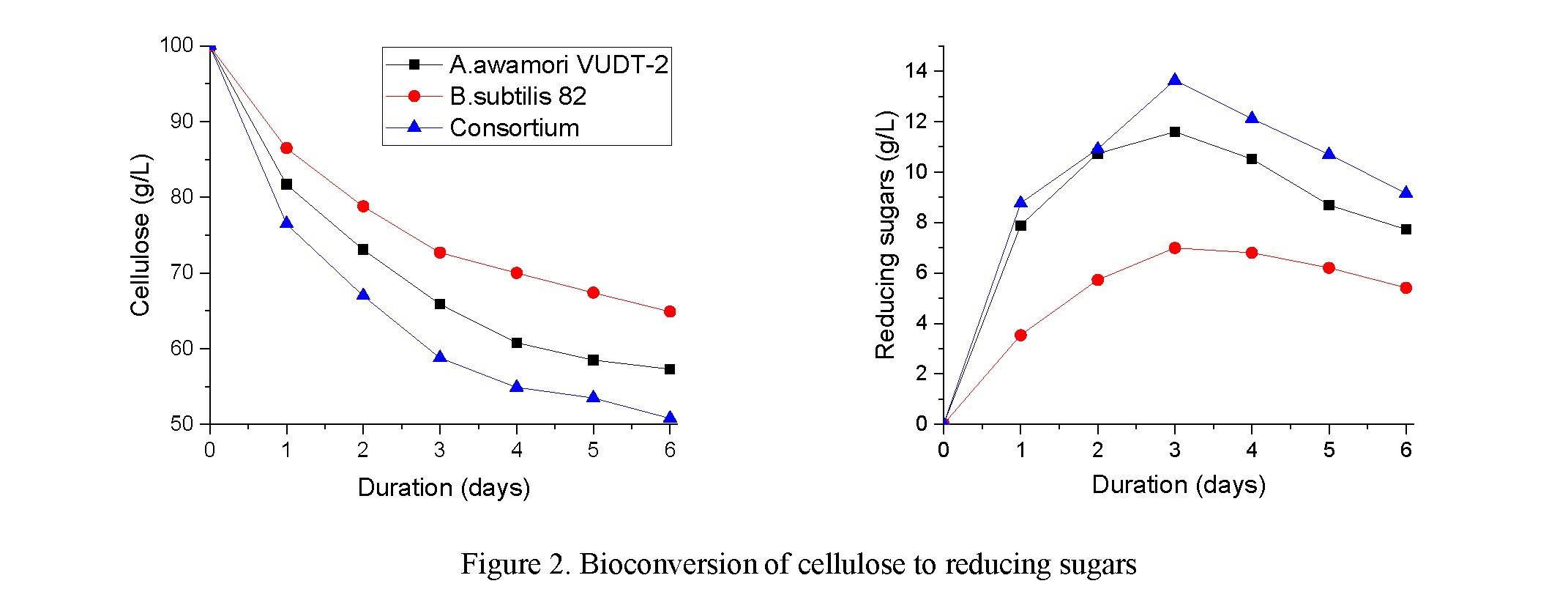
The cultivation was carried out on a shaker-incubator in 750 mL Erlenmeyer flasks at a temperature of 30 °C and a stirring speed of 200 rpm for 8 days. The results of the experiment are shown in Figure 2.
During the experiment, in all cases, a decrease in the amount of cellulose and an increase in concentration of reducing substances were observed. This indicates the growth of Aspergillus awamori VUDT-2 and Bacillus subtilis 82. The number of microorganisms on the 6th day of the experiment increased to 109 CFU/ml and began to decrease on the 7th day of the experiment to 108 CFU/ml and on day 8 was 107 CFU/ml. Therefore, the optimal cultivation period was 6 days.
As it can be seen from Figure 2, in the case of the microorganism Aspergillus awamori VUDT-2, the cellulose content decreased to 58.0 g/L on day 6, and for the microorganism Bacillus subtilis 82, the decrease in cellulose content reached 65.5 g/l on day 6. When consortium was utilized, the cellulose content decreased to 50.8 g/L on day 6 and remained constant until the end of the experiment. The content of reducing sugars on the 3rd day increased and reached 7.0 g/l, 11.5 g/l and 13.63 g/l for Aspergillus awamori VUDT-2, Bacillus subtilis 82 and a consortium of microorganisms, respectively. However, during the experiment, the content of reducing substances was significantly reduced due to consumption by microorganisms, accordingly, the titer of cells decreased.
Therefore, the optimal option for biodegradation of cellulose is a consortium of microorganisms consisting of Aspergillus awamori VUDT-2 from Bacillus subtilis 82.
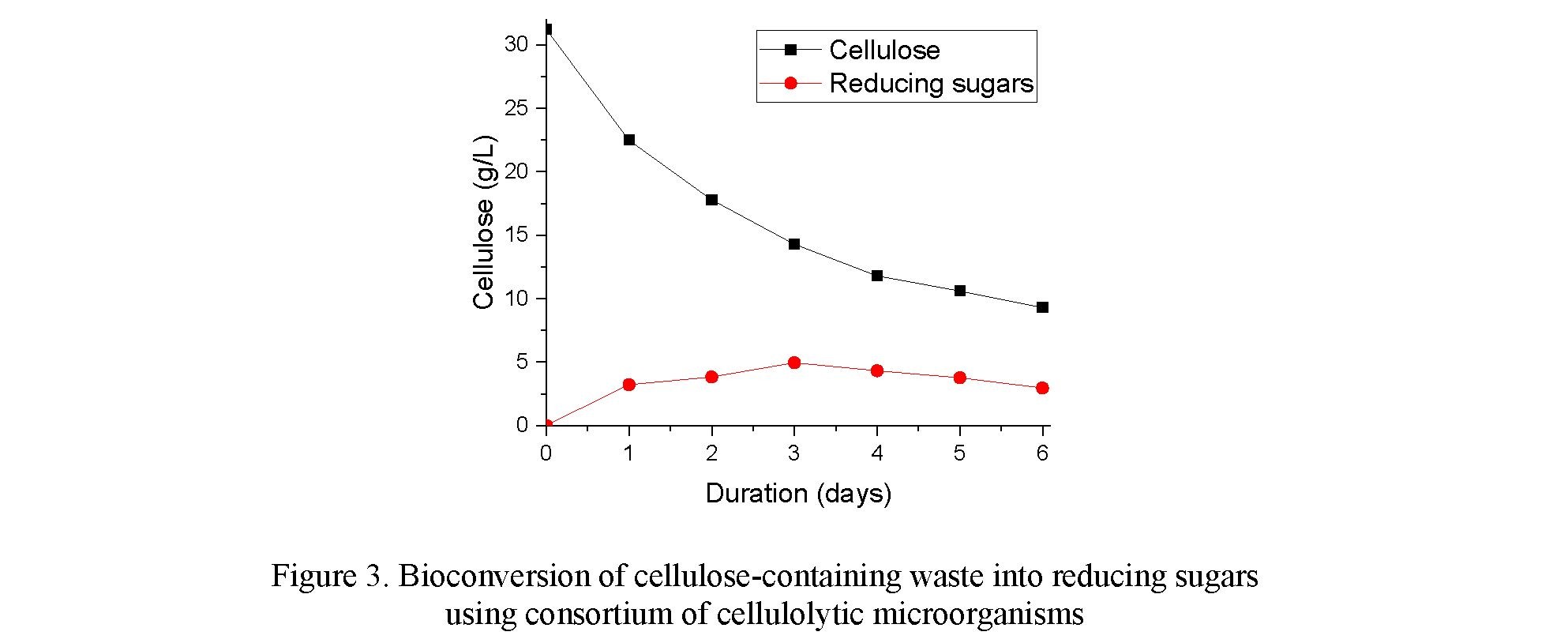
A study of the bioconversion of the cellulose-containing waste with a cellulose content of 20.78 % was conducted. For this, a consortium of microorganisms Aspergillus awamori VUDT-2 and Bacillus subtilis 82 was inoculated into medium with DDGS. The cultivation was carried out on a shaker incubator in 750 mL Erlenmeyer flasks at a temperature of 30 °C and a stirring speed of 200 rpm. The results of the experiment are presented in Figure 3.
According to the data presented in Figure 3, the cellulose content of the medium with DDGS decreased from 31.25 g/L to 9.3 g/L within 6 days. The content of reducing sugars increased in the first 3 days and reached 4.94 g/L, however, after that the content of reducing substances decreased due to the consumption of reducing sugars by microorganisms. On the 6th day of the experiment, the cell titer was 108 CFU/mL. Further incubation led to a decrease in cell titer to 106 CFU/ml, which indicates a decrease in carbohydrate sources.
As a result of the bioconversion of a sample of cellulose-containing wastes using a consortium of cellulolytic microorganisms, the cellulose level decreased by 70.24 % over 6 days and did not change significantly on days 7 and 8.
Thus, the consortium of microorganisms Aspergillus awamori VUDT-2 and Bacillus subtilis 82 is demonstrated high capability for the biodegradation of cellulose in waste from alcohol production.
Conclusion
As a result of growth of microorganisms Aspergillus awamori VUDT-2 and Bacillus subtilis 82, as well as a consortium on CCM, the cellulose content decreased to 58.0 g/L, 65.5 g/L and 50.8 g/L, respectively. The optimal incubation time is 6 days. Using a consortium of microorganisms Aspergillus awamori VUDT-2 and Bacillus subtilis 82, it was possible to lower the cellulose content in waste products of alcohol production to 70.24 % within 6 days. Furthermore, experiments will be conducted to reveal the mechanism of biodegradation and to study the cellulolytic enzymes of these strains.
References
1 Habibi Y. Cellulose nanocrystals: chemistry, self-assembly, and applications / Y. Habibi, L.A. Lucia, O.J. Rojas // Chemical Reviews. — 2018. — Vol. 110, No. 6. — P. 3479–3500.
2 Amaral H.R. Production of high-purity cellulose, cellulose acetate and cellulose-silica composite from babassu coconut shells / H.R. Amaral, D.F. Cipriano, M.S. Santos, M.A. Schettino, J.V.T. Ferreti, C.S. Meirelles, V.S. Pereira, A.G. Cunha, F.G. Emmerich, J.C.C. Freitas // Carbohydrate Polymers. — 2019. — Vol. 210. — P. 127–134.
3 Ferrer A. Nanocellulose in packaging: Advances in barrier layer technologies / A. Ferrer, L. Pal, M. Hubbe // Industrial Crops and Products. — 2017. — Vol. 65. — P. 574–582.
4 Kumar T.S.M. All-cellulose composite films with cellulose matrix and Napier grass cellulose fibril fillers / T.S.M. Kumar, N. Rajini, K.O. Reddy, A.V. Rajulu, S. Siengchin, N. Ayrilmis // International Journal of Biological Macromolecules. — 2018. — Vol. 112. — P. 1310–1315.
5 Du H. Cellulose nanocrystals and cellulose nanofibrils based hydrogels for biomedical applications / H. Du, W. Liu, M. Zhang, C.Si, X. Zhang, B. Li // Carbohydrate Polymers. — 2019. — Vol. 209. — P. 130–144.
6 Picheth G.F. Bacterial cellulose in biomedical applications: A review / G.F. Picheth, C.L. Pirich, M.R. Sierakowski, M.A. Woehl, C.N. Sakakibara, C.F. de Souza, A.A. Martin, R. da Silva, R.A. de Freitas // International Journal of Biological Macromolecules. — 2017. — Vol. 104. — P. 97–106.
7 Rossi E. Carboxymethylated bacterial cellulose: An environmentally friendly adsorbent for lead removal from water / E. Rossi, Ú.M. Rojo, P. Cerrutti, M.L. Foresti, M.I. Errea // Journal of Environmental Chemical Engineering. — 2018. — Vol. 6, No. 6. — P. 6844–6852.
8 Cheng H. Cotton aerogels and cotton-cellulose aerogels from environmental waste for oil spillage cleanup / H. Cheng, B. Gu, M.P. Pennefather, T.X. Nguyen, N. Phan-Thien, H.M. Duong // Materials & Design. — 2017. — Vol. 130. — P. 452–458.
9 Dong C. Feasibility of high-concentration cellulosic bioethanol production from undetoxified whole Monterey pine slurry / C. Dong, Y. Wang, H. Zhang, S. Leu // Bioresource Technology. — 2018. — Vol. 250. — P. 102–109.
10 Chen M. The U.S. cellulosic biofuels industry: Expert views on commercialization drivers and barriers / M. Chen, P.M. Smith // Biomass and Bioenergy. — 2017. — Vol. 102. — P. 52–61.
11 Popovic M. Thermodynamics of hydrolysis of cellulose to glucose from 0 to 100 °C: Cellulosic biofuel applications and climate change implications / M. Popovic, B.F. Woodfield, L.D. Hansen // The Journal of Chemical Thermodynamics. — 2019. — Vol. 128. — P. 244–250.
12 Updegraff D.M. Semimicro determination of cellulose in biological materials / D.M. Updegraff // Analytical Biochemistry. — 1969. — Vol. 32, No. 3. — P. 420–424.
13 Yu X. Ultrasound-ionic liquid enhanced enzymatic and acid hydrolysis of biomass cellulose / X. Yu, X. Bao, C. Zhou, L. Zhang, A.E.A. Yagoub, H. Yang, H. Ma // Ultrasonics Sonochemistry. — 2018. — Vol. 41. — P. 410–418.
References
- Habibi, Y., Lucia, L.A. & Rojas, O.J. (2018). Cellulose nanocrystals: chemistry, self-assembly, and applications. Chemical Reviews, 110, 6, 3479–3500.
- Amaral, H.R., Cipriano, D.F., Santos, M.S., Schettino, M.A., Ferreti, J.V.T., & Meirelles, C.S., et al. (2019). Production of high-purity cellulose, cellulose acetate and cellulose-silica composite from babassu coconut shells. Carbohydrate Polymers, 210, 127– 134.
- Ferrer, A., Pal, L., & Hubbe M. (2017). Nanocellulose in packaging: Advances in barrier layer technologies. Industrial Crops and Products, 65, 574–582.
- Kumar, T.S.M., Rajini, N., Reddy, K.O., Rajulu, A.V., Siengchin, S., & Ayrilmis, N. (2018). All-cellulose composite films with cellulose matrix and Napier grass cellulose fibril fillers. International Journal of Biological Macromolecules, 112, 1310–1315.
- Du, H., Liu, W., Zhang, M., Si, C., Zhang, X., & Li B. (2019). Cellulose nanocrystals and cellulose nanofibrils based hydrogels for biomedical applications. Carbohydrate Polymers, 209, 130–144.
- Picheth, G.F., Pirich, C.L., Sierakowski, M.R., Woehl, M.A., Sakakibara, C.N., & de Souza, C.F., et al. (2017). Bacterial cellulose in biomedical applications: A review. International Journal of Biological Macromolecules, 2017, 104, 97–106.
- Rossi, E., Rojo, Ú.M., Cerrutti, P., Foresti, M.L. & Errea M.I. (2018). Carboxymethylated bacterial cellulose: An environmentally friendly adsorbent for lead removal from water. Journal of Environmental Chemical Engineering, 6, 6, 6844–6852.
- Cheng, H., Gu, B., Pennefather, M.P., Nguyen, T.X., Phan-Thien, N. & Duong, H.M. (2017). Cotton aerogels and cotton- cellulose aerogels from environmental waste for oil spillage cleanup. Materials & Design, 130, 452–458.
- Dong, C., Wang, Y., Zhang, H. & Leu, S. (2018). Feasibility of high-concentration cellulosic bioethanol production from undetoxified whole Monterey pine slurry. Bioresourse Technology, 250, 102–109.
- Chen, M. & Smith, P.M. (2017). The U.S. cellulosic biofuels industry: Expert views on commercialization drivers and barriers. Biomass and Bioenergy, 102, 52–61.
- Popovic, M., Woodfield, B.F. & Hansen L.D. (2019). Thermodynamics of hydrolysis of cellulose to glucose from 0 to 100 °C: Cellulosic biofuel applications and climate change implications. The Journal of Chemical Thermodynamics, 128, 244–250.
- Updegraff, D.M. (1969). Semimicro determination of cellulose in biological materials. Analytical Biochemistry, 32, 3, 420– 424.
- Yu, X., Bao, X., Zhou, C., Zhang, L., Yagoub, A.E.A., Yang, H. & Ma H. (2018). Ultrasound-ionic liquid enhanced enzymatic and acid hydrolysis of biomass cellulose. Ultrasonics Sonochemistry, 41, 410–418.
- Salihu, A., Abbas, O., Sallau, A.B., Alam, M.Z. (2015). Agricultural residues for cellulolytic enzyme production by Aspergillus niger: effects of pretreatment. 3 Biotech., 5, 6, 1101–1106.
- Viles, F.J. & Silverman, L. (1949). Determination of Starch and Cellulose with Anthrone. Analytical Chemistry, 21, 8, 950– 953.
- Netrusov, A.I., Yegorova, M.А. & Zakharchuk, L.M. (2005). Praktikum po mikrobiolohii [Practicum on microbiology]. Moscow: Akademiia [in Russian].
- Collinson, S.R. & Thielemans, W. (2010). The catalytic oxidation of biomass to new materials focusing on starch, cellulose and lignin. Coordination Chemistry Reviews, 254, 15–16, 1854–1870.
- Pérez, J., Muñoz-Dorado, J., de la Rubia, T. & Martínez, J. (2002). Biodegradation and biological treatments of cellulose, hemicellulose and lignin: an overview. International Microbiology, 5, 2, 53–63.
- Díaz, A.B., de Ory, I., Caro, I. & Blandino, A. (2012). Enhance hydrolytic enzymes production by Aspergillus awamori on supplemented grape pomace. Food and Bioproducts Processing, 90, 1, 72–78.
- Petre, M., Zarnea, G., Adrian, P. & Gheorghiu, E. (1999). Biodegradation and bioconversion of cellulose wastes using bacterial and fungal cells immobilized in radiopolymerized hydrogels. Resources, Conservation and Recycling, 27, 4, 309–332.