When taking probiotics, probiotic bacteria do not always successfully take root in the human body, and prebiotics are used to stimulate their growth and metabolic activity. A wide range of biologically active substances of natural origin used as prebiotics is of special interest and positively impacts the probiotic bacteria. In this regard, priority is currently given to this problem in the leading scientific centers around the world. The research in the field of microbiology is acquiring special relevance: obtaining dairy products for healthy nutrition and preparations of biologically active living cultures of probiotic bacteria of a new generation with prebiotics. This article presents the study on the influence of flax seeds on the reproduction rate of lactic acid bacilli when used in the experiment. The modern microbiological methods were engaged in conducting the given experiment. MALDI-TOF MS was used to identify lactic acid bacilli. It has been found that the whole flaxseeds added into a fermentation medium for probiotic cultures stimulate cell proliferation of lactic acid bacilli Lh13, Lpl-5. This indicates some application potential for using a probiotic with some prebiotic added in the form of whole flax seeds.
Introduction
For microbiological industry, it is paramount to expand a range of stock culture and improve the technology for obtaining and accumulating it. Some features of this process are focused on achieving the highest rate of biomass growth and producing probiotic cultures with high biological activity [1]. The biomass of probiotic crops accumulates in a nutrient medium containing sources of carbon, nitrogen, minerals, and vitamins. As the production of probiotic cultures grows bigger, the search for alternative raw materials to prepare nutrient media takes the leading place. The use of such raw materials, rich in carbohydrates, proteins and minerals, dietary fiber, allows for reducing costs and increasing the profitability of the enterprise [2–4].
The bacteria of the Lactobacillus, Lacticaseibacillus, Lactiplantibacillus, Limosilactobacillus genus are particularly susceptible to complex microorganisms [5]. The lactobacillus requires the substrates, which are a source of nutrition, do not grow on simple nutrient media, and need amino acids, vitamins, fatty acids, carbohydrates and nucleic acid derivatives to be introduced into them (individual for each type of lactobacillus) [6]. In addition to amino acids and vitamins, there are a number of other organic compounds, as well as the plant substrates required for the growth of lactobacillus or stimulating growth.
In the process of growth and reproduction, lactobacilli in their nutrient medium need stimulants of various natures. The use of plant-based and natural biologically active additives (BAAs) is known for its stimulating nature in the growth properties of lactic acid bacteria and is well-described in the scientific literature. A suspension obtained from soybean grains, walnut flour, Laetiporus sulphureus Ls 1–06 mycelium biomass obtained by various types of cultivation, including liquid phase cultivation in the stationary phase can be considered as good examples [7–11].
Burmasova et al. [12] tried to obtain probiotic drugs with natural antioxidants. They studied the effect of low concentrations of Chaga mushroom’s melanins on the growth of lactobacilli. The experiment showed that the action of melanins manifests itself in the exponential phase. Melanins at the concentrations of 10–10 g/cm3 and 10–5 g/cm3 contribute to the formation of more lactobacilli compared to the control group with a higher result of seeding, which is confirmed by the data of the mentioned study.
The Spanish researchers Pozuelo et al. investigated the ability of grape antioxidant dietary fiber to affect the microflora in the cecum in the experimental rats. Grape antioxidant dietary fiber is rich in polyphenol amines. The results showed that the consumption of grape antioxidant dietary fiber stimulates the growth of lactobacilli and affects the composition of bifidobacterial species, but only to an insignificant extent. It was also found that grape antioxidant dietary fiber has a stimulating effect on Lactobacillus reuteri and Lactobacillus acidophilus in vitro [13].
Warnakulasuriya et al. [14] studied the fermentation of dietary rice fiber with the joint cultivation of probiotic cultures of lactobacillus and bifidobacteria. The combined cultures grew on fiber from both rice varieties (LD356-RR1, red in color, brown rice, peel-free, and AT353-RR2, red in color, unpolished) achieving high rates of cell biomass accumulation. The study revealed some synergistic relations between these bacterial cultures. In this study, the high number of cells in the starter cultures provided high initial levels of cell count in the medium: 108–109 CFU/ml. Adding the fibrous substrate to the culture medium had a marked effect on the culture growth and viability, especially at 24-hour incubation, and in all culture combinations. This indicates the ability of co-cultures to use rice fiber as a growth-stimulating substrate. However, some joint strains of lacto-and bifido-bacterium cultures have shown a preference for glucose over fiber as a substrate. The microorganisms constituting the combinations reached the die-off phase after a short stationary phase, between 24 and 48 hours. The pH dropped down rapidly in all media reaching the lowest level after 6 hours of cultivation.
Akalin et al. [15] studied the microflora of probiotic ice cream with various enriched dietary fibers (apple, orange, oats, bamboo, and wheat) and the viability of cultures stored at -18 °C for 180 days. The experiment established a viable amount of Lactobacillus acidophilus ≥7 log CFU/g during storage with the exception of samples having orange and bamboo fiber in them. It was also found that the amount of Bifidobacterium lactis in these samples was>6 log CFU/g up to 150 days of storage.
Thus, a comparative literature review of pro- and pre-biotics, which can be used to cultivate lactic acid bacteria, demonstrates that the success of growing these microorganisms depends on the quality and composition of the medium, as well as the specifics of the stimulating components.
Polysaccharides of flaxseed are referred to as mucus-like high-molecular polysaccharides, in other words, mucus soluble in water. Along with a high content of polyunsaturated fatty acids (PUFAs) and protein, flax seeds are characterized by the presence of a significant amount of soluble dietary fiber based on mucus polysaccharides. Flaxseed mucus also attracts attention as a source of biologically active oligosaccharides [16].
Linumus itatissimum L. or flaxseed is an important functional food ingredient as it is rich in α-linolenic acid (ALA, omega-3 fatty acid), lignans and fiber, which are potentially beneficial to human health. Due to the high content of polysaccharides, the decoction of flax seeds has an enveloping and bactericidal effect. The F, A, E, B vitamins and flax seeds are excellent external sources of important vitamin F, which is actively involved in fat and cholesterol metabolism. The five main nutritional components of flaxseed are fiber, protein, lignans, oil, and vitamins, which are determined by the methods of gas chromatography and high-performance liquid chromatography (HPLC) [17–18].
Coarse flaxseed is rich in biologically active compounds, and its extracts have a strong anti-radical activity [19]. It contains the largest amount of secoisolariciresinol diglucoside (SDG) of plant lignan [20].
Numerous studies have shown that the consumption of flaxseed leads to an improvement in the metabolism of glucose or lipids [21, 22].
The current study on flax seeds is at the center of increased interest in the field of research on the health benefits associated with its individual components and their biological activity.
The given work aims to optimize the intensity of reproduction of lactobacillus cells by rational cultivation with raw extract of flaxseeds added in various concentrations.
Experimental
The lactobacillus isolates of L. helveticus-13(Lh-13) and L. plantarum-5 (Lpl-5) isolated from national lactic acid products, which are produced by artisanal traditional method in different districts of the Karaganda region were used for the experiment. The obtained isolates were identified by MALDI BioTyper [23].
To assess the growth of Lh-13, Lpl-5 lactobacillus isolates with added raw extract flaxseeds (whole flaxseed used from Zerde Phyto-tea), a growth curve was used. Further, a sterile enzymatic medium was used for probiotic cultures [24] to cultivate the Lh-13, Lpl-5 lactobacillus isolates with added raw extract offlaxseeds. For the control group, 50 μl of inoculate containing 105 CFU/ml of Lh-13, Lpl-5isolates was added to the enzymatic medium for probiotic cultures with no raw extract of flaxseeds.
In the experimental groups, the raw extract of lax seeds was used at the concentrations of 5%, 10%, 15% in the enzymatic medium for probiotic cultures (100 ml) with 50 μl of inoculated suspension containing 105 CFU/ml of Lh-13, Lpl-5 lactobacillus isolates. After suspension, it was cultured at 37ºC under anaerobic conditions.
The growth rate was determined by the increase in optical density. Throughout the culture cycle, the samples were well mixed in a magnetic stirrer at regular intervals. The fermented liquid was taken from the samples under study, and the optical density of the suspension was measured by the TecanEVolizer100 (Tecan) laboratory robot at the wavelength of 490 nm. The trials were carried out three times with the results expressed in the form of an arithmetic mean.
The control microscopy was performed in the control and experimental groups during cultivation. The samples were taken for microscopy every 0, 8, 16, 24, 36, 48, 72 hours. The Gram stain method was used.
Results and Discussion
Based on the obtained data, growth curves were constructed within 72 hours. When analyzing the constructed growth curves, it can be concluded that when raw extract is added to the medium, the growth of Lh-13, Lpl-5 lactobacillus isolates increases. In all experiments and in the control group, the optical density values varied.
Some significant increase in the optical density of the culture fermented liquid was recorded in 8 hours of cultivation in the experimental groups. Nevertheless, the control groups showed an increase in cells only after 8 hour.
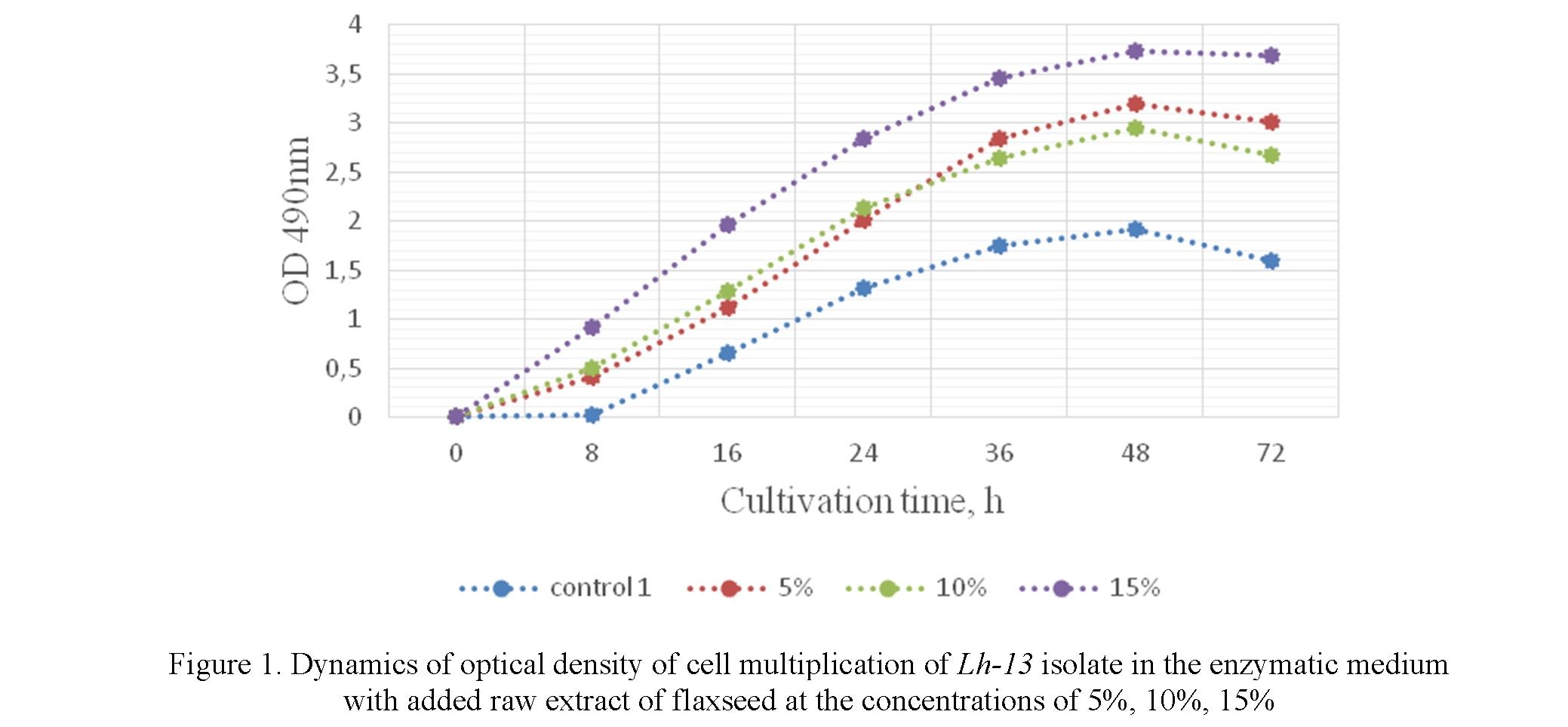
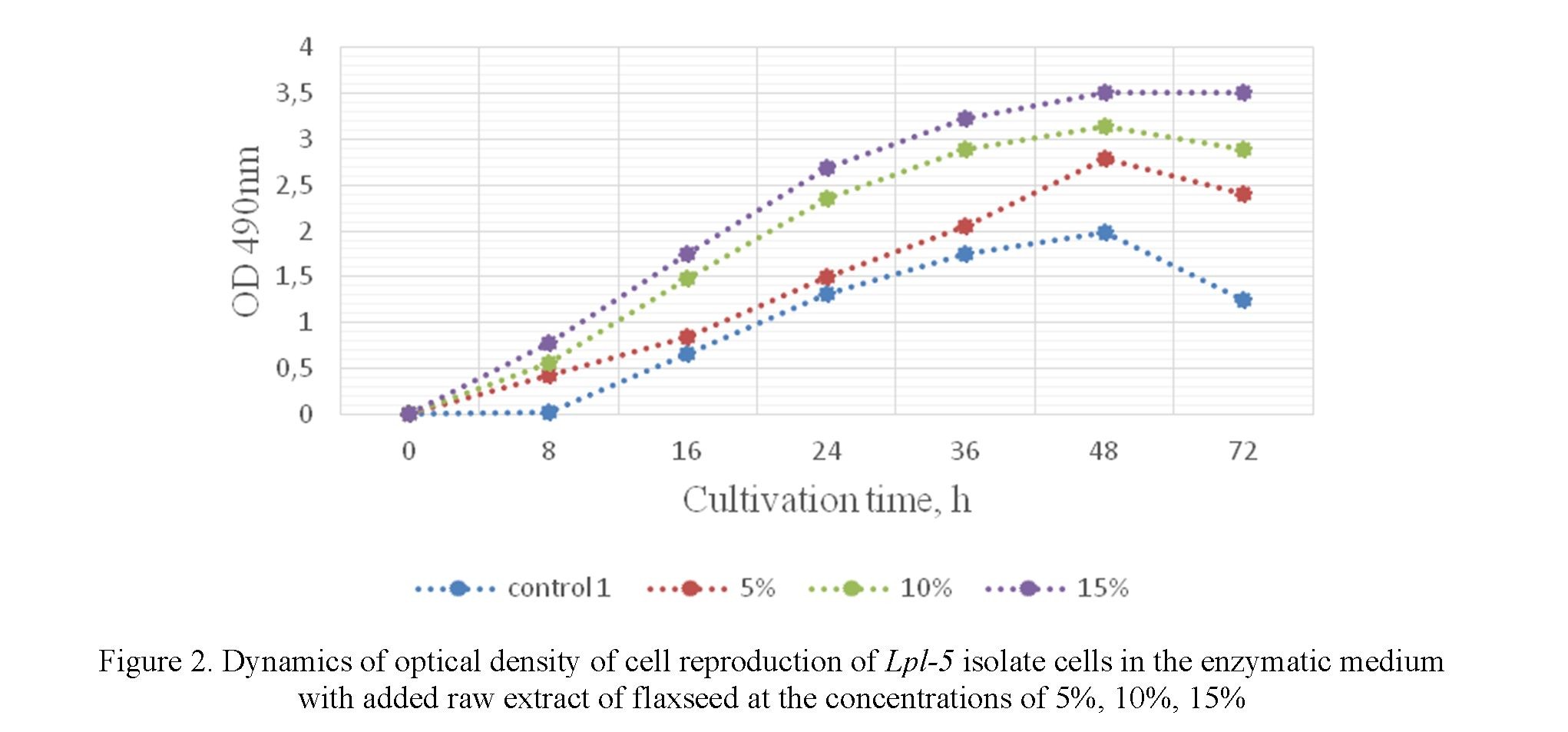
After 8 hours of cultivation, growth is accelerated due to the faster growth of Lh-13, Lpl-5 isolates and the consumption of nutrients from the culture fermented liquid (Fig. 1, 2).
After 16, 24, 36 hours of cultivation with 5%, 10%, 15% of raw extract of the flaxseed added to it, the reproduction rate of cell increases. In 48 hours of cultivation, the biomass of the cells reached its maximum in each experimental group in all concentrations.
In the experimental groups with the addition of 5 %, 10 % raw extract of flaxseeds after 48 hours, one could observe the onset of the growth inhibition. Meanwhile, the growth of lactobacillus is constant in case of the addition of 15% raw flaxseed extract with the optical density being slightly varied. In the control groups, a decrease in values is well observed during cultivation of 48 hours with some decrease in the biomass. There was an imbalance and rapid death of cell biomass observed.
 The study on the effect of the accumulated maximum cell count of Lh-13, Lpl-5 isolates with the addition of 5%, 10% concentration of raw flaxseed extract was conducted at the acidity value of the medium varied from pH 5.6; 5.8; 6.0; 6.1 and at the temperature of 37oC (8–72 h).
The study on the effect of the accumulated maximum cell count of Lh-13, Lpl-5 isolates with the addition of 5%, 10% concentration of raw flaxseed extract was conducted at the acidity value of the medium varied from pH 5.6; 5.8; 6.0; 6.1 and at the temperature of 37oC (8–72 h).
28
Вестник Карагандинского университета
29
In the medium with a 15% concentration of raw flaxseed extract added to the Lh-13, Lpl-5 isolates, the acidity activity of the medium varied from pH 5.5–6.2. In 24–48 hours, the acidity of the medium was maintained at the pH level of 5.5–5.7. In the control samples, the acidity of the medium ranged from 5.7–6.0. The analysis of the presented results shows that the highest cell content was as follows: Lh-13 isolates OD = 3.73 (48 h) and Lpl-5 OD = 3.52 (48 h).
This study is fully consistent with other researchers who have similarly reported that the use of prebiotics helps to stimulate cell growth of lactobacillus. By combining these two components in so-called synbiotic products, one can expect a positive effect on the intestinal microflora [25–27].
In our opinion, dietary fiber is an additional substrate with a certain set of substances capable of satisfying the nutritional needs of lactobacillus: vitamins, micro- and macro-elements, which are indispensable factors for the growth of lactobacillus.
In the course of research, it was found that introducing such prebiotics as raw extract of flaxseeds in the concentrations of 1–5% into the nutrient medium allows for maximizing the development of the studied Lh- 13, Lpl-5 isolates. This is expressed in an increase in the rate of cell reproduction, which is a rational management process. Whereas 5–10% of the added stimulant for 72 hours causes a decrease in the culture growth (OD). The analysis of the obtained data demonstrates that the intensity of reproduction of the studied isolates depends on the type and amount of growth stimulant introduced.
In the control and prototype samples, microscopy showed that the gram-positive rods of the cell are located: singly, in pairs and in clusters. They were immobile and typical for lactic acid bacilli, forming no spores.
According to the results of this study, raw flaxseed extract (Linum usitatissimum L.) may be a potential prebiotic to stimulate the growth of bacteria in the colon. The raw flaxseed extract is a promising nutritional basis to cultivate lactobacillus. Therefore, flaxseed mucus has prebiotic properties, which, in turn, allows us to consider the possibility of including it in functional products.
Thus, in case of correct selection of dietary fiber (raw extract of the flaxseed), an attempt was made to achieve significant results in cultivating the Lh-13, Lpl-5 lactobacillus in an experiment to increase the stimulating properties of probiotic cultures.
Conclusions
The up-to-date microbiological technologies dictate the relevance of the use of flaxseed dietary fiber. They have a unique composition and are rich in dietary fiber, which can meet the nutritional needs of lactobacillus. The use of flaxseed dietary fiber stimulates the growth of lactobacilli and will help to determine the appropriate growth-stimulating concentrations of dietary fiber by randomized selections in the experiment while improving the nutritional value and biological activity of starters to be used in the finished product. The beneficial effects of dietary fiber can be improved when combined with prebiotic cultures.
References
- Lysenko, Yu.A., Luneva, A.V., Volkova, S.A., Nikolaenko, S.N., & Petrova, V.V. (2014). Podbor optimalnoi pitatelnoi sredy dlia kultivirovaniia, kontsentrirovaniia i vysushivaniia kletok Lactobacillus acidophilus [Selection of optimal nutrient medium for cultivation, concentration and drying of Lactobacillus acidophilus cells]. Politematicheskii setevoi elektronnyi nauchnyi zhurnal Kubanskogo gosudarstvennogo agrarnogo universiteta — Polythematic Online Scientific Journal of Kuban State Agrarian University, 102 (08); 1–11 [in Russian].
- Alekseeva, T.V., Riaskina, L.O., & Rodionov, A.A. (2016). Novye sinbioticheskie pishchevye sistemy dlia profilakticheskogo pitaniia [New synbiotic food systems for preventive nutrition]. Izvestiia vuzov. Seriia Pishchevaia tekhnologiia — Bulletin of Higher Educational Institutions. Food Technology, 4 (352); 44–47 [in Russian].
- Ghosh, C., Sarkar, P., Issa, R., & Haldar, J. (2019). Alternatives of conventional antibiotics in the era of antimicrobial resistance. Trends Microbiol., 27(4); 323–328.
- Ardatskaia, M.D. (2010). Klinicheskoe primenenie pishchevykh volokon: metodicheskoe posobie [Clinical use of dietary fiber. Methodical manual]. Moscow: 4TE Art [in Russian].
- Zheng, J., Wittouck, S., Salvetti, E., Franz, C.M.A.P., Harris, H.M.B., & Mattarelli, P. (2020). A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol., 70(2); 2782–2858. https://doi.org/10.1099/ijsem.0.004107.
- Bespomestnykh, K.V. (2015). Izuchenie vliianiia sostava pitatelnoi sredy na izmenenie biokhimicheskikh i morfologicheskikh svoistv shtammov laktobatsill [Study of the influence of the composition of the nutrient medium on the change in the biochemical and morphological properties of strains of lactobacilli]. Zhurnal «Nauchnoe obozrenie». Seriia: Tekhnicheskie nauki — Scientific Review Journal. Technical Sciences, 1; 77, 78 [in Russian].
- Drozdov, R.A., Kozhukhova, M.A., & Barkhatova, T.V. (2016). Optimizatsiia retseptury probioticheskogo kislomolochnogo napitka s dobavleniem pishchevykh volokon topinambura [Optimization of the formulation of a probiotic fermented milk drink with the addition of Jerusalem artichoke dietary fiber]. Polzunovskii vestnik — Polzunov Bulletin, 4 (2); 4–11 [in Russian].
- Domotenko, L.V., Shepelin, A.P., & Detushev, K.P. (2014). Sravnitelnye ispytaniia laktobakagar i MRS agara [Comparative tests of lactobacillus and MRS agar]. Kurskii nauchno-prakticheskii vestnik «Chelovek i ego zdorove» — Kursk Scientific and Practical Bulletin “Man and His Health”, 4; 5–10 [in Russian].
- Van de Casteele, S., Vanheuverzwijn, T., & Ruyssen, T. (2006). Evaluation of culture media for selective enumeration of probiotic strains of lactobacilli and bifidobacteria in combination with yoghurt or cheese starter. Int Dairy J., 16 (12); 1470–1476.
- Veliamov, M.T., Chizhaeva, A.V., Vorobev, A.L., & Dudikova, G.N. (2015). Pitatelnaia sreda dlia vyrashchivaniia probioticheskikh mikroorganizmov na osnove molochnoi syvorotki i rastitelnykh nanoporoshkov [Nutrient medium for growing probiotic microorganisms based on whey and plant nanopowders]. Innovatsionnyi patent № 29970 — Innovating Patent No. 29970 [in Russian].
- Manju, P., & Danik, M. (2012). Optimization of an effective growth medium for culturing probiotic bacteria for applications in strict vegetarian food products. Functional foods in Health and Disease, 2 (10); 369–378.
- Burmasova, M.A., Miljuhina, A.K., Mubarakshina, G.Sh., Utebaeva, A.A., & Sysoeva, M.A. (2017). Rost laktobakterii na srede s melaninami chagi [Growth of lactobacilli on a medium with chaga melanins]. Vestnik Tekhnologicheskogo universiteta — Bulletin of Technological University, 20 (8); 152–154 [in Russian].
- Pozuelo, M.J., Agis‐Torres, A., Hervert‐Hernandez, D., Lopez‐Oliva, M.E., Munoz‐Martinez, E., & Rotger, R. (2012). Grape Antioxidant Dietary Fiber Stimulates Lactobacillus Growth in Rat Cecum. Journal of Food Science., 77 (2); 59–62.
- Warnakulasuriya, M.A.D.B.F., Flint, S., Zou, M., & Brennan, C.S. (2011). The effect of rice fibre fractions on the growth of co-cultures of probiotics. Journal Food Science and Technology, 48 (1); 14–25.
- Akalin, A.S., Kesenkas, H., Dinkci, N., Unal, G., & Ozer, E. (2018). Enrichment of probiotic ice cream with different dietary fibers: Structural characteristics and culture viability. Journal of the American Dairy Science Association January, 8 (101); 37–46.
- Minevich, I.Ye., Osipova, L.L., Nechiporenko, A.P., Smirnova, E.I., & Melnikova, M.I. (2018). Osobennosti protsessa ekstraktsii polisakharidov slizi iz semian lna [Features of the process of extraction of mucus polysaccharides from flax seeds]. Nauchnyi zhurnal natsionalnogo issledovatelskogo universiteta ITMO. Seriia Protsessy i apparaty pishchevykh proizvodstv — Scientific Journal of National Research University ITMO. Series Processes and Food Production Equipment, 2, 3–11 [in Russian].
- Ivanova, S., Rashevskaya, T., & Makhonina, M. (2011). Flaxseed additive application in dairy products production. Procedia. Food Sci., 1; 275–280.
- Alhassane, T., & Xu, X. M. (2010). Flaxseed lignans: source, biosynthesis, metabolism, antioxidant activity, bio-active components and health benefits. Compr. Rev. Food Sci. Food Saf., 9; 261–269.
- Tavarini, S., De Leo, M., Matteo, R., Lazzeri, L., Braca, A., & Angelini, A. (2021). Flaxseed and Camelina Meals as Potential Sources of Health-Beneficial Compounds. Plants. 10; 156. https://doi.org/10.3390/plants10010156
- Zhang, W., Wang, X.B., Liu, Y., Tian, H.M., Flickinger, B., Empie, M.W., & Sun, S.Z. (2008). Dietary Flaxseed Lignan Extract Lowers Plasma Cholesterol and Glucose Concentrations in Hypercholesterolaemic Subjects. British Journal of Nutrition, 99, 1301–1309. https://doi.org/10.1017/S0007114507871649
- Almehmadi, A., Lightowler, H., Chohan, M., & Clegg, M.E. (2021). The Effect of a Split Portion of Flaxseed on 24-h Blood Glucose Response. European Journal of Nutrition, 60; 1363–1373. https://doi.org/10.1007/s00394-020-02333-x
- El-Seedy, G.M., El-Shafey, E.S., & Elsherbiny, E.S. (2021). Fortification of Biscuit with Sidr Leaf and Flaxseed Mitigates Immunosuppression and Nephrotoxicity Induced by Cyclosporine A. Journal of Food Biochemistry, 45; 13655. https://doi.org/10.1111/jfbc.13655
- Alatoom, A.A., Cunningham, S.A., Ihde, S.M., Mandrekar, J.N., & Patel, R. (2011). Comparison of Direct Colony Method versus Extraction Method for Identification of Gram-Positive Cocci by Use of Bruker Biotyper Matrix-Assisted Laser Desorption Ionization–Time of Flight Mass Spectrometry. J. Clin Microbiol, 49(8); 2868–2873. https://doi.org/10.1128/JCM.00506-11
- Kushugulova, A.R., Saduahasova, S.A., Shahabaeva, G.S., Kozhahmetov, S.S., Tynybaeva, I.K., Nurgozhin, T.S., & Zhumadilov, Zh.Sh. (2015). Fermentatsionnaia sreda dlia probioticheskikh kultur [Fermentation medium for probiotic cultures]. Innovatsionnyi patent № 29528 — Innovative Patent No. 29528 [in Russian].
- Lee, J.K., Cho, H.R., Kim, K.Y., Lim, J.M., Jung, G.W., Sohn, J.H., & Choi, J.S. (2014). The growth — stimulating effects of fermented rice extract (FRe) on lactic acid bacteria and Bifidobacterium spp. Food Science and Technology Research., 20; 479– 483.
- Dong-Hyeon, K., Jeong, D., Oh, Y.-T., Song, K.-Y., Kim, H.-S., Chon, J.-W., Kim, H., & Seo, K.-H. (2017). Stimulating the Growth of Kefir-isolated Lactic Acid Bacteria using Addition of Crude Flaxseed (Linum usitatissimum L.) Extract. J. Milk Sci. Biotechnol, 35(2); 93–97.
- Theegala, M., Arévalo, R.A.C., Viana, V., Olson, D., & Aryana, K. (2021). Effect of Flaxseed on Bile Tolerances of Lactobacillus acidophilus, Lactobacillus bulgaricus, and Streptococcus thermophilus. Food and Nutrition Sciences, 12 (7); 670–680. https://doi.org/10.4236/fns.2021.127050