Complex generic is a generic with multifaceted dynamic constituent, multifaceted preparation, multifaceted path of administration, or multifaceted medicine device mixture. For over three decades, generics have been a desirable and cost-effective production route. Since the pharmaceutical cluster aimed at cheap generic medicines takes active development, biopharmaceutical firms remain focusing on multifaceted medicine, which provides extra worth towards affected person via speaking extra unmet requirements besides allow them to attain pharmaceutical souk variation and advanced using. The complexity of chemisimilar and biosimilar products has limited the replacement of unwieldy large-scale clinical trial data with simpler, single-dose, pharmacokinetic and pharmacodynamic comparisons between test and reference. This limitation should be recognized by defining the sensitivity of any test used to detect differences in dose.
Introduction
A generic medicine is a medicine that can be arranged as an auxiliary for the inventor creation for the bioequivalence takes stood verified. The authorized who description of generic medicine: “identical multisource therapeutic creation” [1]. To be “generic”, a therapeutic creation must preliminarily show the similar qualitative and quantitative constituents of dynamic ingredient and the equal dosage form as that of the original remedy. Complex generics present additional challenges. Biopharmaceutical firms that select the correct associates toward assist through invention, conception and governing policies will secure substantial benefits. The guidelines fundamental the valuation of excellence of generic medicines, besides their bioequivalence to product title, take not altered meaning fully under the Hatch-Waxman Act in 1984. So far, the goods produced in the industry are meaning fully extra difficult nowadays, which might suggest that II bioequivalent goods remain distinguishable, respectively. Multifaceted generic production necessities have a higher degree of competences than simple generics developed in an environment of increasing expenses and amplified analysis over worldwide advance then industrial processes.
Review
Complex Generics
Complex generics is defined as common medications for which it is particularly difficult to establish therapeutic equivalence as described in Orange Book. Multifaceted generics are a generic that might take a complex dynamic component, complex preparation, complex path of distribution, or complex medicine device mixtures [1, 2]. To be «complex generic», a therapeutic ingredient essential primarily show not individual similar qualitative and quantitative constituent of dynamic ingredients and the similar dosage form as that of the original ingredient but similarly lots of several extra parameters are measured to evidence «repetitiveness», such as physical classification like particle size distribution, medicine set-up, particle morphology, physical state of entrapment, drug release, viscosity, globule size, zeta potential, excipient description etc. Generic mixture of drug substance remains generally characterized in GDUFA II direction and denote to oral goods with more than one dynamic, multifaceted preparations, such as liposomes, suspensions, emulsions, and gels, complex paths of direction, also as locally acting goods (topical creams and gels, nasal spray, and inhalation goods), drug mixtures with devices, long-acting injectables [3].
The advancement of generic [505(j)] and distinguished [505(b)(2)] LAI goods support to deliver affected person and health care workers with more action choices and to decrease general health care expenses, including those related with drug goods direction and affected person obedience. In this review, we analyse the scenery of LAI goods and categorize the most mutual procedural tasks that probable generic goods applicants’ appearance [4]. We focus on five preparation skills that explanation for ~90 % of permitted LAI goods, with those qualified for generic goods registering over the succeeding five ages, to demonstrate technology-specific tasks.
Complex Generic Combination Drug Products
The multifaceted generic goods might take a multifaceted dynamic component, preparation, route of distribution, or be a medication-device mixture. Since the goods include generic components, numerous firms are revolving their attention to multifaceted generic medicines, which bring additional problems to affected person including supplementary requirements through encouraging such medicine struggle and persistent entrée [5].
Complicated mixture of generic component goods can be abundant with stimulating components, and need abundant information to influence the governing activities of comparison of the two medicinal goods. The endorsement necessities need considerable earlier than for biosimilars and for generic goods. Here, the procedure cast-off to making the good has an affected influence on the excellence and presentation of the good. The subsequent governing procedure for the generic constituents is ample nearer to that for biosimilars, as understood in the governing direction for glatiramer acetate ANDAs.
Chemisimilars
When the pharmacokinetic profile correlates with the dose, such comparisons show assay sensitivity and readily detect differences in dose. This approach has also been applied to certain chemical products, termed ‘chemisimilars’, which have variable or complex synthesis of the active substance, or complex formulation, or a complex delivery device [6]. Once demonstrating batch-to-batch steadiness or correspondence of generics shows that artificial organic medicine is multifaceted. This rises through precise release preparations, mutable or multifaceted mixture, or substances wherever the configurations of sub ingredient are not well distinct. The equivalents with biosimilars are understandable, and sometimes cross applications are mandatory, where comparative information is jointed with stand-alone effectiveness and protection experimental information. Many such goods are replacements to the modernizing goods without existence interchangeable. Chemisimilar is a beneficial term occasionally cast-off by EU officials to define these multifaceted organic generics [7].
These include semi-synthetic antibiotics, approximately liposomal preparations and some slow-release trans-cutaneous coverings.
When proving batch-to-batch consistency, or equivalence of generics, some innovator synthetic chemical drugs are complex. This arises with controlled release formulations, variable or complex synthesis, or chemicals where the composition of subcomponents are not well defined. The parallels with biosimilars are obvious, and sometimes hybrid applications are required, where comparative data are combined with stand-alone efficacy and safety clinical data [8]. Many such products are alternatives to the innovator products without being substitutable.
Biosimilar
Biosimilar medicinal products are large, complex molecules that deliver active components to a binding site. A test protein may have the same primary, secondary and tertiary structure as a reference protein. When the pharmacokinetic profile correlates with the dose, such comparisons show assay sensitivity and readily detect differences in dose. For large biological molecules, different manufactured batches cannot be validated using pharmacokinetic data alone [9]. For these biosimilars, there is a three-stage assessment of pharmaceutical quality; laboratory testing and clinical data. Biosimilar therapeutic components are huge, composite particles that bring movement to an obligatory location. A test protein might take the similar primary, secondary and tertiary structure as a situation protein. So far movement at, or transport to, the obligatory location may be pretentious by additional organic variances, such as glycosylation, N-sylation, phosphorylation, deaminization, corrosion or PEGylation. Dissimilar obligatory locations might be responsible for efficiency, opposing actions, and breakdown and elimination paths. This complication is a challenge for biosimilar guideline. Different binding sites may be responsible for efficacy, adverse events, and metabolism and excretion pathways. This complexity is a challenge (or difficult) for biosimilar regulation. Small differences in manufacture or formulation can have serious clinical consequences, as documented with some bio similar epoetins and insulin.
Minor variances in production or preparation can have thoughtful experimental significances, as recognized with approximately biosimilar epoetin and Humulin. The equilibrium among responsive to testing and experimental tests that demonstration no variance in the approved sign remains to be challenging for biosimilar guideline. The current standard on biosimilar interferon b (IFN-b) for multiple sclerosis (MS) states to dose- responsive to models. The financial impact of generic drug competition can be dramatic, but significant differences in regulatory and development processes between generics and biosimilars limit such comparisons and likely present significant challenges for biosimilar approval and adoption in the US market [10]. However, a value-based care environment and their cost-savings potential make biosimilars an attractive option for the therapeutic arsenal. Oncologists’ understanding of biosimilars is critical to moving forward.
Hatch-Waxman Act
The way of the Hatch-Waxman Act in 1984 completed it easy for makers of generic medicines to arrive the U.S. medical market [11]. Earlier in 1984, generics makers remained obligatory to prove the security and efficiency of their goods using experimental trials. Conferring to Mossinghoff (1999), previous to Hatch-Waxman act, added 150 components occurred in the pharmaceutical usage which required neither patent security nor generic entrance. Hatch-Waxman Act removed the obligation for distinct experimental trials for generic producer. The Act also aided modernizer goods by encompassing the life of therapeutic patents which obligated long period on their “patent timepieces” coming up for fda endorsement [12].
Grabowski and Vernon (1996) file the rise in generic goods operation subsequent the way of HatchWaxman Act in 1984. They invention that generic provision in the primary 1980s be around 10 out of a hundred, but enlarged to 40 out of a hundred in the middle-1990s. Berndt and Aitken (2010) display that meanwhile then generic treatment shares take remained cumulative intensely. Among 1999 and 2004, the share production of generic goods increased from 49.7 out of a hundred to 74.5 out of a hundred. This quick generic entrance is allied through consistently quick failures in generics values. Rizzo and Zeckhauser (2005) asked that this deterioration in values of generics was allied through the deterioration in the usual value of product tag goods. Lately, the values of approximately generic goods take enlarged intensely subsequent in a Household Board on Omission and Administration Improvement search. Although generic components employment has evidently enlarged subsequent Hatch-Waxman Act signifying, but convenience and affordability of medications take enhanced buyer wellbeing, around remain likewise parts of apprehension.
As an outcome of Hatch-Waxman Act, the FD&C Act necessitates that not used one of the subsequent IV documentations be complete when documenting an ANDA or 505(b) (2) resolutions:
Para I: That such patent data takes not remained not used.
Para II: That such patent takes perished.
Para III: The generic medication motivation not go on the usual way till the patent ending time permits.
Para IV: The patent in query is inacceptable, impossible to enforce, or will not infringe through the making, use, or auction of the generic components.
GDUFA Act
The Generic Medication Handler Payment Revisions/Act (GDUFA) is a regulation enacted through the government of the United States of America (USA), on 9th July 2012, beneath the Food and Drug Administration Safety and Innovation Act (FDASIA) near quicken the admittance of harmless and active generic medicines to the civic and decrease the inclusive price for commercial use [13].
The act is a result of the increasing challenges in the Regulatory landscape which needs to be re-authorized every five years. According to the law, all the generic drugs manufacturers, including manufacturers of active ingredients for generic drugs, are required to submit a user fee to the FDA.
It is rule intended towards rapidity admittance to harmless and active medicines to the civic, besides decrease charges to manufacturing. Until October 2012, it was the individual obligatory handler payments for organizations succumbing New Drug Application (NDA).
By means of October 1, 2012, beneath GDUFA, entirely companies that making humanoid generic medicine goods and dynamic constituents for humanoid generic medicine goods, that are circulated in U.S. Business are topic to FDA handler payments [14].
GDUFA charges remain rise the capability of the Activity to accomplish perilous program purposes and to decrease charges seeing the decrease appraisal timelines.
Challenges
Developing multifaceted generic goods is stimulating as therapeutic correspondence, but might not inter- prete to a cathartically corresponding preparation.
The major task includes those places advancing through the modernizers of these goods, counting patent tasks and resident appeals. Added in that one of the stimulating features is the key variance in suggestion standards among numerous universal activities; which makes it difficult to develop the same product for distribution in different parts of the world.
In addition to the tasks inside the commerce, multifaceted generic associated suggestions to existing the substantial task to the OGD.
The GDUFA (Generic Drug User Fee Act), dissimilar its novel medicine complement PDUFA (Prescription Drug User Fee Act), does not take the establishment for steady pre-ANDA conferences or for suggestion of INDs.
The tasks associated to governing suggestions for multifaceted generics can be overwhelmed or curtailed by empathetic the substitute suggestion procedures needed for these medicines and subsequent over consequently.
One all-time major challenge is to counter anti-generic campaigns attributed to be advocated by patient and physician groups, but suspected to be at least in part funded by innovator companies. One effective way by which generics have learnt to compete with innovator companies is through diligent mergers and acquisitions.
One all-time major challenge is to counter anti-generic campaigns attributed to be advocated by patient and physician groups, but suspected to be at least in part funded by innovator companies. Another challenging situation is attempt by innovators to delay or stop generic entry by filing citizen’s petitions with the FDA.
Opportunities
Generics firms too frequently variation in the excipients in a preparation since the constructer of the pioneer medicine takes a formulation patent; the generics firm is observing to evade this patent. A generics firm’s capability to shift excipients in oral goods frequently boundaries the worth of preparation patents for oral goods.
The problem for the pioneer medicine firm is classically wide preparation privileges can be change on inaccuracy situation and some preparation privileges are frequently circumvent able.
Based on the FDA direction on liposomal doxorubicin, it is currently deceptive that, at a least, the generic liposome would to take to compare with the pioneer preparation in state of medicine, interior atmosphere, element/bubble morphology, element/bubble size distribution, polymer alignment, electrical exterior latent and medicine removed [15].
A challenge to evade the entitlement by replacing a dissimilar polymer or a little dropping the attention of a specific polymer would not be permitted for a multifaceted generic.
The mixture of uniform an actual arrangement or procedure patent entitlement by the FDA’s overall necessities for parenteral medicine goods and the discriminating necessities for unconventional or multifaceted transport systems can outcome in the important and elongated dated.
The necessity for translucent and consistent governing appeal for complex generics
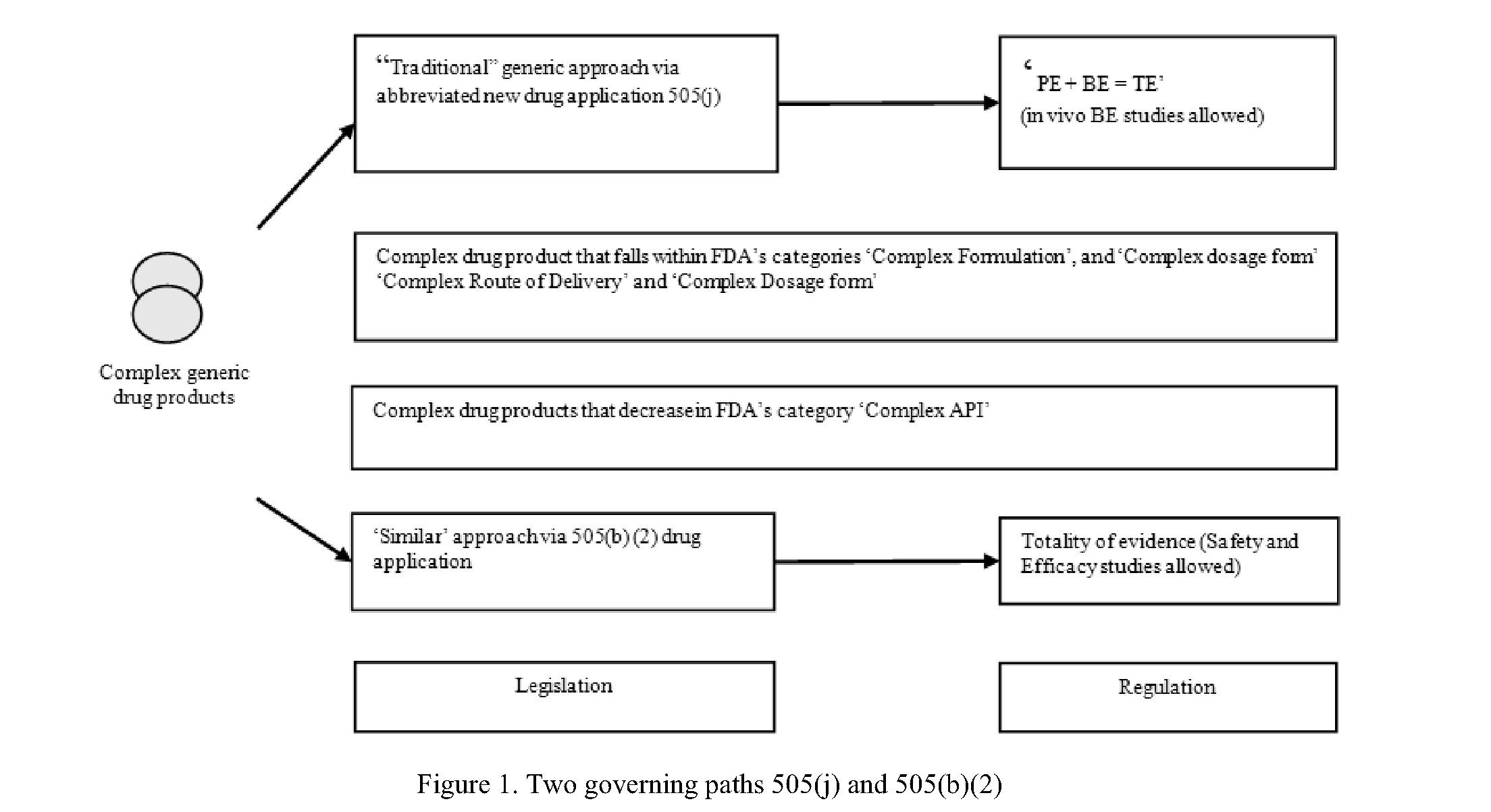
In current ages, the worldwide systematic and governing civic takes in progress to distinguish the important systematic tasks that occur for complex generic medicine goods, concerning their valuation of therapeutic equivalence. Though, hard work to bring into line governing procedures is still continuing. In the United States, the quantity of suggestion obligatory for the endorsement of a multifaceted generic till seems to frequently depend on a case-by-case appeal by the FDA. To reproduce on recent governing repetition and to deliver realistic recommendations for using tasks within prevailing governing outlines, this paper designates the opinions of the NBCD Occupied Collection on how to type greatest usage of prevailing abbreviated approval paths of 505(j) and 505(b)(2) for approving complex generics.
Current governing rehearsal for complex generics
For the generic medicine goods to be official and estimated as therapeutically equivalence (TE), it needs to be pharmaceutically equivalent (PE) as well as bioequivalent (BE) to the Reference Listed Drug (RLD). This approach is appropriate for “humble” minor particle generics, but comes difficult with tasks for pe and/or BE with complex generics.
A pragmatic and science-based governing approach for complex generic
Since current effort by the FDA on decreasing the sprints of complex generic medicine advance, the FDA has printed their description of “complex generic drug products.” Complex drug products are defined as goods with:
^ a complex dynamic component (e.g., peptides, polymeric composites, complex combinations of APIs, and obviously obtained components);
133
^ a complex preparation (e.g., liposomes and colloids);
^ a complex route of distribution (e.g., locally acting drugs, such as dermatologic goods and complex ophthalmological goods and otic dosage forms that are framed as suspensions, emulsions, or gels);
^ a complex dosage form (e.g., transdermal, metered dose bronchodilators, and extended release injecta- bles).
505(j) Guideline
“An anda is an application succumbed and official beneath section 505(j) of the fd&c Act for a medicine good that is a replacement of an earlier official medicine good. An ANDA depend on FDA’s verdict that the earlier agreed drug goods, i.e., the Reference Listed Drug (RLD), is harmless and active. An ANDA usually essential encompass data to display that the planned generic product1 is the similar by means of the RLD with admiration to the dynamic constituent(s), situations of usage, way of administration, dosage form, asset, and cataloguing (by certain allowable variances) and 2 is bioequivalent to the RLD. An ANDA do not be succumbed if experimental surveys are obligatory to found the protection and efficiency of the planned medicine goods” (Fig. 1).
505(b)(2) Guideline
“A 505(b)(2) application is an nda succumbed beneath section 505(b)(1) and official beneath section 505(c) of the FD&C Act that covers complete information of surveys of protection and efficiency, wherever at least approximately of the data obligatory for endorsement arises from revisions not directed by or for a correct of location otherwise usage”.
How to agreement with dynamics in a defined governing outline
Rendering to the International Conference on Harmonization (ICH), a CQA is a guide for physical, chemical, biological, or microbiological assets or characteristic, which are fundamental inside a suitable boundary, series, or circulation to confirm the wanted goods excellence. For multifaceted medicine goods encompassing multifaceted APIs, there is a pure necessity to rise the information on individuals’ goods features that take an influence on the excellence and the experimental outline of the goods. Subsequent to CQAs, a improved empathetic of additional related features is wanted to rise the strength of valuations for complex drug goods.
They have faith in numerous situations aimed at a vigorous appeal through 505(j) and 505(b)(2) occur (individually of such 4 situations are additional reflected arranged beneath):
- Empathetic of all related critical quality attributes (CQAs);
- A confident quantity of elasticity in the governing approaches for multifaceted generic medicine goods;
- An inclusive and widely obtainable list of all multifaceted goods per GDUFA II Obligation Letter;
Серия «Биология. Медицина. География» No. 1(105)/2022
- Supplementary valuation of latent variances and fences through affection to Book programs or successive practice in experimental exercise.
Conclusions
To conclude, the guidelines fundamental the valuation of excellence of generic medicines, and their bioequivalence to product title goods, have not altered meaningfully despite the adopted act of the Hatch-Waxman Act in 1984. So far, various products are much more complex, which may indicate that 2 bioequivalent goods are not indistinguishable for individual addition. As long as suggestion from medicines to delicate epilepsy, misery and other situations this paper express that converting from a modernizer product to a generic might outcome in opposing significances for affected person. Moreover, generic and product title medicines cause additional constituents and uniform last goods after external the U.S. specifically from countries like India and China which have unfortunate governing omission. This would influence excellence in US market.
Future of Complex Generics
The upcoming of generics is in growth of complex generics, biologics or biosimilars for which here is an enlarged selectness dated of 12 ages in favour of the modernizer in dissimilarity to the 5-year New Chemical Entity (NCE) selectness. The legislation governing biosimilars is not the Hatch Waxman Act, but the 2010 Biosimilar Price Competition and Innovation Act (BPCIA) with a different set of regulatory exclusivity provisions. It is extremely serious for pharma firms to interchange on after ordinary generics to complex generics, specific goods, biosimilars and advanced goods, which motivate upcoming therapeutic development.
- Bate, R., Mathur, A., Lever, H.M., Thakur, D., Graedon, J., Cooperman, T., Mason, P., & Fox, E.R. (2016). Generics substitution, bioequivalence standards, and international oversight: complex issues facing the fda. Trends Pharmacol. Sci., 37 (3); 184–191. https://doi.org/10.1016/j.tips.2015.11.005
- Raw, A.S., & Wu, L. (2019). Scientific Considerations in the Approval of Complex Generics. In book The scientific considerations in the approval of complex generics. Springer, 32; 157–173. https://doi.org/10.1007/978-3-030-11751-1_9
- Klein, K., Borchard, G., Shah, V.P., Flühmann, B., McNeil, S.E., & de Vlieger, J.S. (2021). A pragmatic regulatory approach for complex generics through the us fda 505 (j) or 505 (b)(2) approval pathways. Ann N Y Acad Sci., 1502(1); 5–13. https://doi.org/10.1111/nyas.14662
- Warren, J.B. (2013). Generics, chemisimilars and biosimilars: is clinical testing fit for purpose. British Journal of clinical pharmacology, 75 (1); 7–14. https://doi.org/10.1111/j.1365–2125.2012. 04323.x
- Nambiar, S., Madurawe, R.D., Zuk, S.M., Khan, S.R., Ellison, C.D., & Faustino, P.J., et al. (2012). Product quality of parenteral vancomycin products in the United States. Antimicrobial agents and chemotherapy, 56(3); 2819–2823. https://doi.org/10.1128/AAC.05344-11
- Sterken, R.K. (2017). The meaning of generics. Philosophy Compass, 12 (8); e12431. https://doi.org/10.1111/phc3.12431
- Leslie, s.-j. (2008). Generics: Cognition and Acquisition. Philosophical review, 117 (1); 1–47.https://doi.org/10.1215/00318108-2007-023
- Bell, C., Anderson, J., Ganguly, T., Prescott, J., Capila, I., Lansing, J.C., Sachleben, R., Iyer, M., Fier, I., Roach, J., & Storey, K. (2017). Development of Glatopa® (glatiramer acetate): The first FDA-approved generic disease-modifying therapy for relapsing forms of multiple sclerosis. Journal of Pharmacy Practice, 31 (2); 89719001772584. https://doi.org/10.1177/0897190017725984
- O'Brien, M.N., Jiang, W., Wang, Y., & Loffredo, D.M. (2021). Challenges and opportunities in the development of complex generic long-acting injectable drug products. J Control Release., 336; 144–158. https://doi.org/10.1016/j.jconrel.2021.06.017
- Misra, A. (2010). Are biosimilars really generics? Expert Opin Biol Ther., 10 (4); 489–494.https://doi.org/10.1517/14712591003662615
- Boehm, G., Yao, L., Han, L., & Zheng, Q. (2013). Development of generic drug industry in the USA after the Hatch-Waxman Act of 1984. Acta Pharmaceutica Sinica, 3 (5); 297–311. https://doi.org/10.1016/j.apsb.2013.07.004
- Gupta, R., Kesselheim, A.S., Dowing, N., Greene, J., & Ross, J.S. (2016). Generic drug approvals since the 1984 HatchWaxman Act. jama Intern Med., 176 (9); 1391–1393. https://doi.org/10.1001/jamainternmed.2016.3411
- Sindkhedkar, M., Jagtap, S., Shah, C., & Palle, V.P. (2020). Pharmaceutical Research in India: Current Status and Opportunities. Proceedings of the Indian National Science Academy, 86 (2); 1015–1022. https://doi.org/10.16943/ptinsa/2020/153225
- Warren, J.B., & Oselin, K. (2011). Generic and therapeutic substitution: a viewpoint on achieving best practice in Europe. British Journal of clinical pharmacology, 72 (5); 727–730. https://doi.org/10.1111/j.1365–2125.2011. 03939.x
- Nabhan, C., Parsad, S., Mato, A.R., & Feinberg, B.A. (2018). Biosimilars in oncology in the United States: a review. JAMA oncology, 4 (2); 241–247. https://doi.org/10.1001/jamaoncol.2017.2004