Extraction processes are largely used in many chemical, biotechnological and pharmaceutical industries for recovery of valuable and bioactive compounds. The hydrodynamic cavitation can be effectively used to improve the extraction rate by the increasing the mass transfer and possible rupture of cell wall due the formation of microcavities leading to higher product yields with reduced processing time and solvent consumption [3, 4]. The aim of our research was the study of hydrodynamic cavitation influence on physical and chemical properties of different extractants. Determination of refractive index, density, рН on rheological properties of extractants were carried out [10].
Keywords: cavitation, hydrodynamic cavitation, extractants, determination, physical properties.
Introduction. Bioactive compounds are largely obtained from medicinal plants. Solid-liquid extraction is used in many chemical, biochemical and pharmaceutical industries for recovery bioactive compounds [9]. Plants generally contain only a small amount of active compounds, but in most cases its high value justifies the development of the high-performance process. The need for effective extraction of bioactive compounds from plants without any loss of activity and high purity has resulted in development of newer process of extraction [1].
It is possible to influence various methods on the kinetics of the mass transfer process, but the special interest is hydrodynamic cavitation. The phenomenon of cavitation is widely used to intensify a number of technological processes: splitting of solid particles into a suspension, dissolving solid particles, extraction, etc.
Hydrodynamic cavitation describes the process of vaporisation, bubble generation and bubble implosion which occurs in a flowing liquid as a result of a decrease and subsequent increase in local pressure [2, 6]. Cavitation will only occur if the local pressure declines to some point below the saturated vapor pressure of the liquid and subsequent recovery above the vapor pressure.
The process of bubble generation, and the subsequent growth and collapse of the cavitation bubbles, results in very high energy densities and in very high local temperatures and local pressures at the surface of the bubbles for a very short time. Controlled cavitation can be used to enhance chemical reactions or propagate certain unexpected reactions because free radicals are generated in the process due to disassociation of vapors trapped in the cavitating bubbles. All this initiates a convective mass transfer in porous particles [4, 7, 8]. Experimental. Experiments on the study of cavitation have been taking place for a long time, but still this problem remains relevant. Of particular interest is the use of hydrodynamic cavitation, in which there is virtually no significant negative effect on biologically active substances. Usually, cavitation hydrodynamic devices of dynamic or static type are used. Therefore, we carried out experimental studies on a cavitation hydrodynamic device of a dynamic type, the main element of which is a high-velocity impeller.
In the first phase of our study, we had studied the effect of hydrodynamic cavitation on various physical and chemical properties of known extractants. The objects of the research comprised the purified water, ethyl alcohol 40%, 50%, 60%, 70%, and 96% [5].
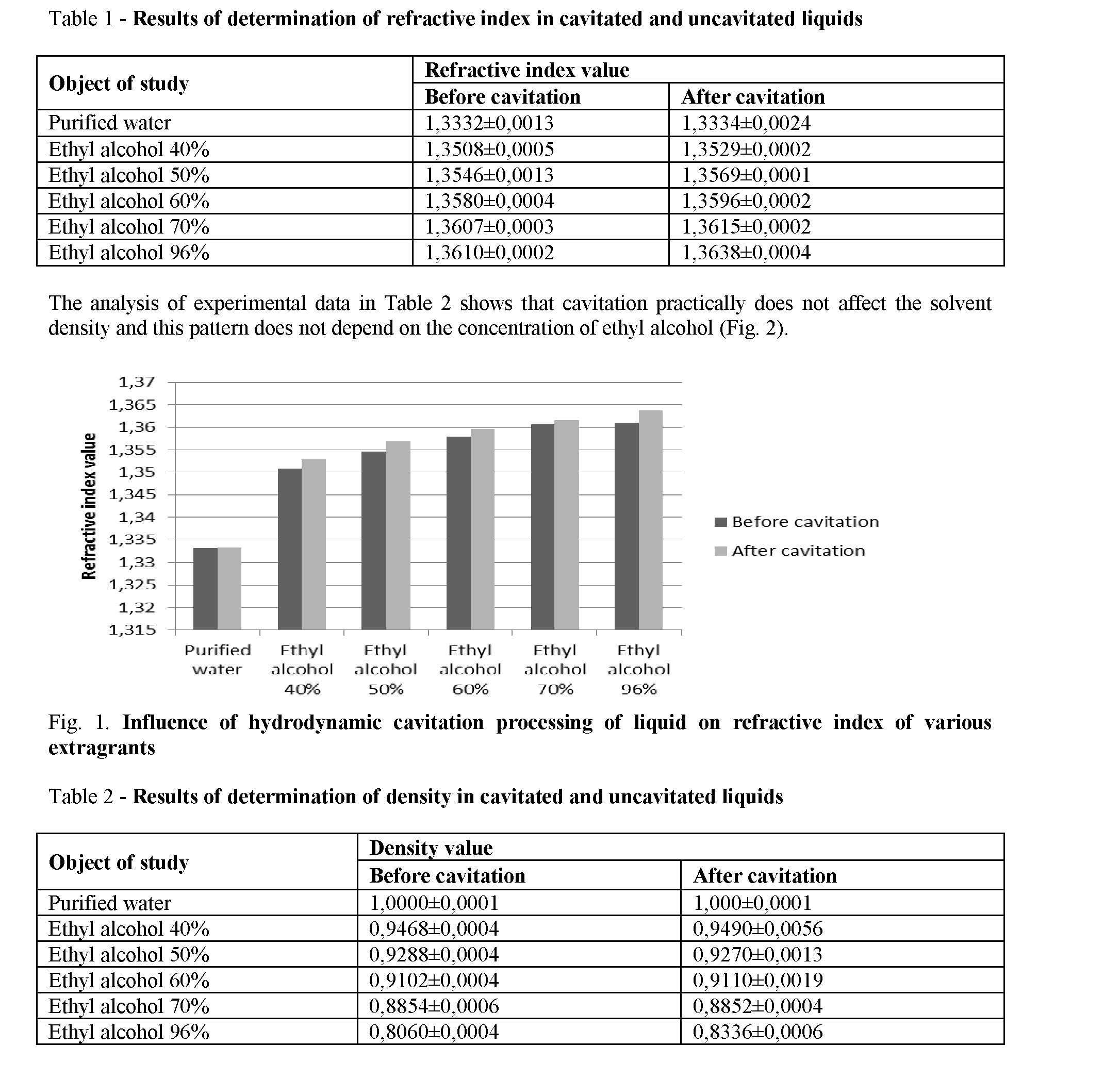
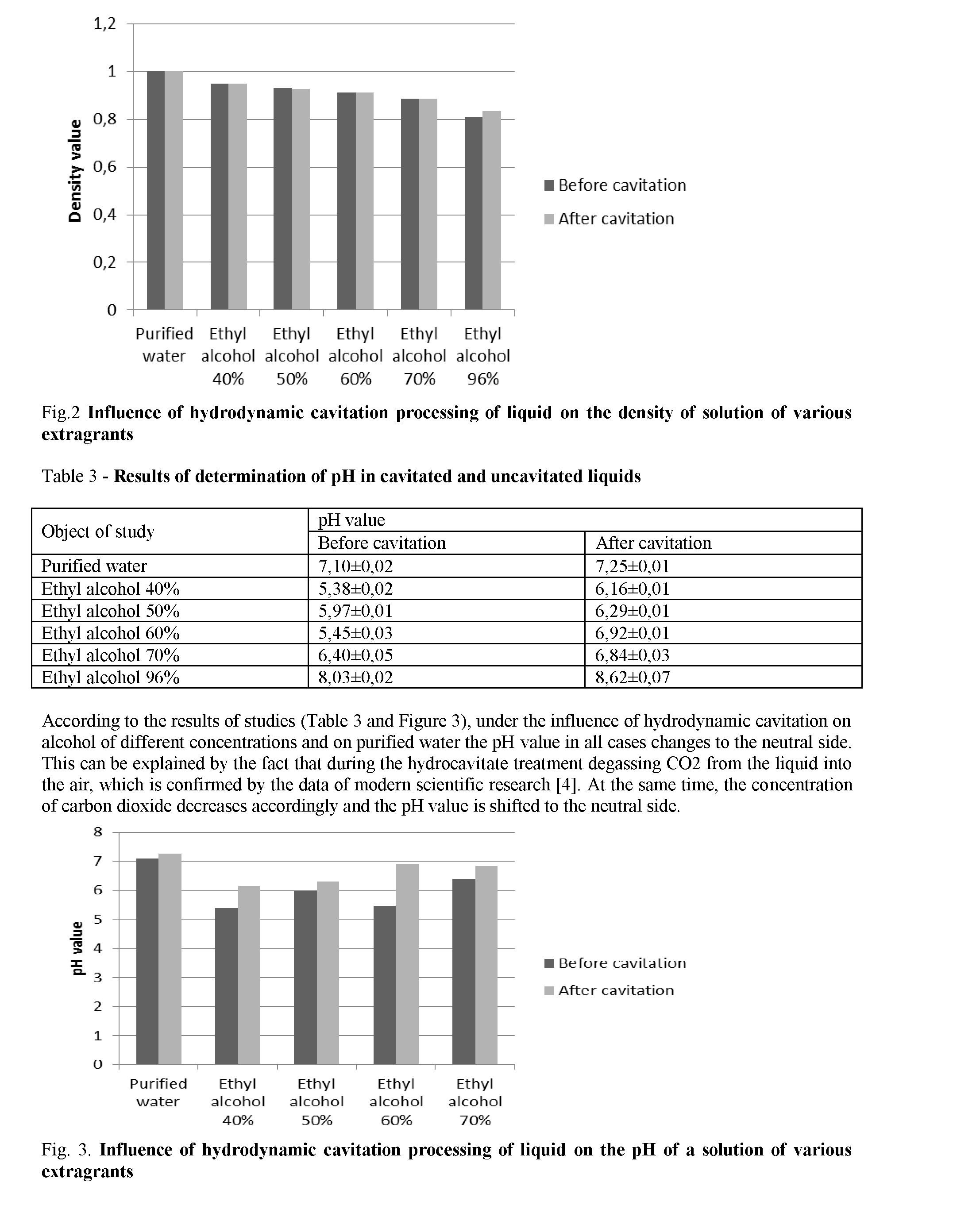
Results and discussion. Determination of density, pH and refractive index before and after cavitation was carried out of the influence of hydrodynamic cavitation on various physical and chemical parameters of purified water, ethyl alcohol 40%, 50%, 60%, 70%, and 96%. The results are shown in the tables 1-3.
Determination of the refractive index shows that there is a tendency to increase, under the influence of cavitation, changes in optical properties of liquids that do not depend on the nature of the solvent (Fig. 1).
36
37
Conclusions. Thus, the various physicochemical properties of known solvents investigated indicate that hydrodynamic cavitation is a powerful factor of influence, which reduces the strength of liquids into a rupture and promotes the formation and development of active cavitation bubbles.
The use of hydrodynamic cavitation irradiation during the extraction procedure presents several advantages in terms of shortening the time of the process, decrease the volume of the extracting solvent and increasing the yield of the extraction in comparison with conventional methods.
References
- Albanese L, Ciriminna R, Meneguzzo F, Pagliaro M. Energy efficient inactivation of Saccharomyces cerevisiae via controlled hydrodynamic cavitation. Energy Sci Eng 2015; 3: 221238.
- Amin LP, Gogate PR, Burgess AE, Bremner DH. Optimization of a hydrodynamic cavitation reactor using salicylic acid dosimetry. Chem Eng J 2010; 156: 165-169.
- Bagal MV, Gogate PR. Degradation of diclofenac sodium using combined processes based on hydrodynamic cavitation and heterogeneous photocatalysis. Ultrason Sonochem 2014b: 21: 10351043.
- Brennen, Christopher. "Cavitation and Bubble Dynamics" (PDF). Oxford University Press. p. 21. Retrieved 27 February 2015.
- Derzhavna Farmakopeya Ukrayini. [State Pharmacopoeia of Ukraine]. Derzhavne pidpriemstvo Ukrayinskiy naukoviy farmakopeyniy tsentr yakosti likarskih zasobiv. Ch 2. Vol. 1. Kharkiv: Derzhavne Farmakopeya pidpriemstvo Ukrayinskiy naukoviy farmakopeyniy tsentr yakosti likarskih zasobiv( 2015).
- Franke M, Ondruschka B, Braeutigam P. Hydrodynamic-Acoustic-Cavitation for Biodiesel Synthesis. 3rd International Conference on Environment, Chemistry and Biology, 2014; 78: 23-30.
- Gogate, P.R.; Kabadi, A. M. (2009). "A review of applications of cavitation in biochemical engineering/biotechnology". Biochemical Engineering Journal. 44 (1): 60-72.
- Holzfuss J. Chemical Oscillations of Air-Seeded Bubbles in Water Driven by Ultrasound / Holzfuss J. //Rapid Communication, Phys. Rev. —2008, E 78, 025303(R). — Р. 24.
- Huang Y, Wu Y, Huang W, Yang F, Ren X. Degradation of chitosan by hydrodynamic cavitation. Polym Degrada Stab 2013; 98: 37-43.
- Terentiev, A, Kirschner, I, Uhlman, J, (2011) The Hydrodynamics of Cavitating Flows. Backbone Publishing Company, 598pp.