Summary
In the this work has been shown the method of obtaining titanium substance (TiCl3, Ti2(SO4)3, TiO2, Ti(OH)4, (NH4)2TiO(SO4)2) from metallic wastes by the polarization of the industrial unstationary current in water’s solution. The goal of the dissertation workis devise new electrochemical methods of the gettings titanium substance from it wastes by the polarization of unstationary current.
The process of dissolution’s metallic titanium wastes by alternating current in the water solution is researched first time. Experiments was spent in electrochemical cell, consist of three section, in the middle section is situated titanium wastes, working as bipolar electrodes.
Keywords: titanium waste, electrolysis, alternating current, polarization, dissolution.
GOAL RESEARCH
Currently, titanium is widely used in aircraft and shipbuilding, chemical and metallurgical industries, in this regard, the processing of titanium metal, a large number of waste metal. The problem of waste processing and use of titanium current, this problem received much attention since the beginning of industrial production of titanium. Recycling of waste associated with great difficulties, because titanium is poorly soluble, refractory (tplav = 17250S) and solid metal. Titanium is corrosion resistant in acidic and alkaline solutions and is soluble only in concentrated sulfuric acid at its boiling point at a very low speed. In connection with this waste of titanium virtually no processing, no use, and accumulated in the warehouses of the industrial enterprises.
One promising solution to this problem is to dissolve the waste of titanium followed by a synthesis of his connections, finding wide application in various branches of light and chemical industries. However, the existing methods for producing titanium compounds have many drawbacks and are very costly because the metal dissolution is carried out at high temperatures, in concentrated acid for a long time. It is also known that when titanium insoluble anode polarization due to the formation on the surface of the oxide films. In this context, the aim of the thesis is to study the electrochemical dissolution of metal scrap titanium polarization current and non-stationary industrial synthesis of its compounds.
BATCH EXPERIMENTS
Experiments were performed in an electrochemical cell consisting of three sections, the middle section contains titanium chips operating as a bipolar electrode. The electrodes in the two end sections used titanium plate connected to the AC source.
As the use of titanium waste titanium shavings Ust-Kamenogorsk titanium - magnesium plant with a titanium content of not less than .%98,45
When alternating current is observed polarization dissolving titanium, which allows to obtain various compounds of titanium electrochemical method. The main electrode processes occurring in the cathode halfcycle AC is a discharge of hydrogen ions and the restoration of existing on the electrode surface oxide film:
2H++2e->TH2 (2)
TiO2+4H+ + 4e->Ti+2H2O Е0=-0,90В (3)
In the anode half cycle AC mostly are dissolved titanium in the form of trivalent ions:
Ti - 3e- > Ti3+ Е0 = -1,21 В (4)
RESULTS AND DISCUSSION
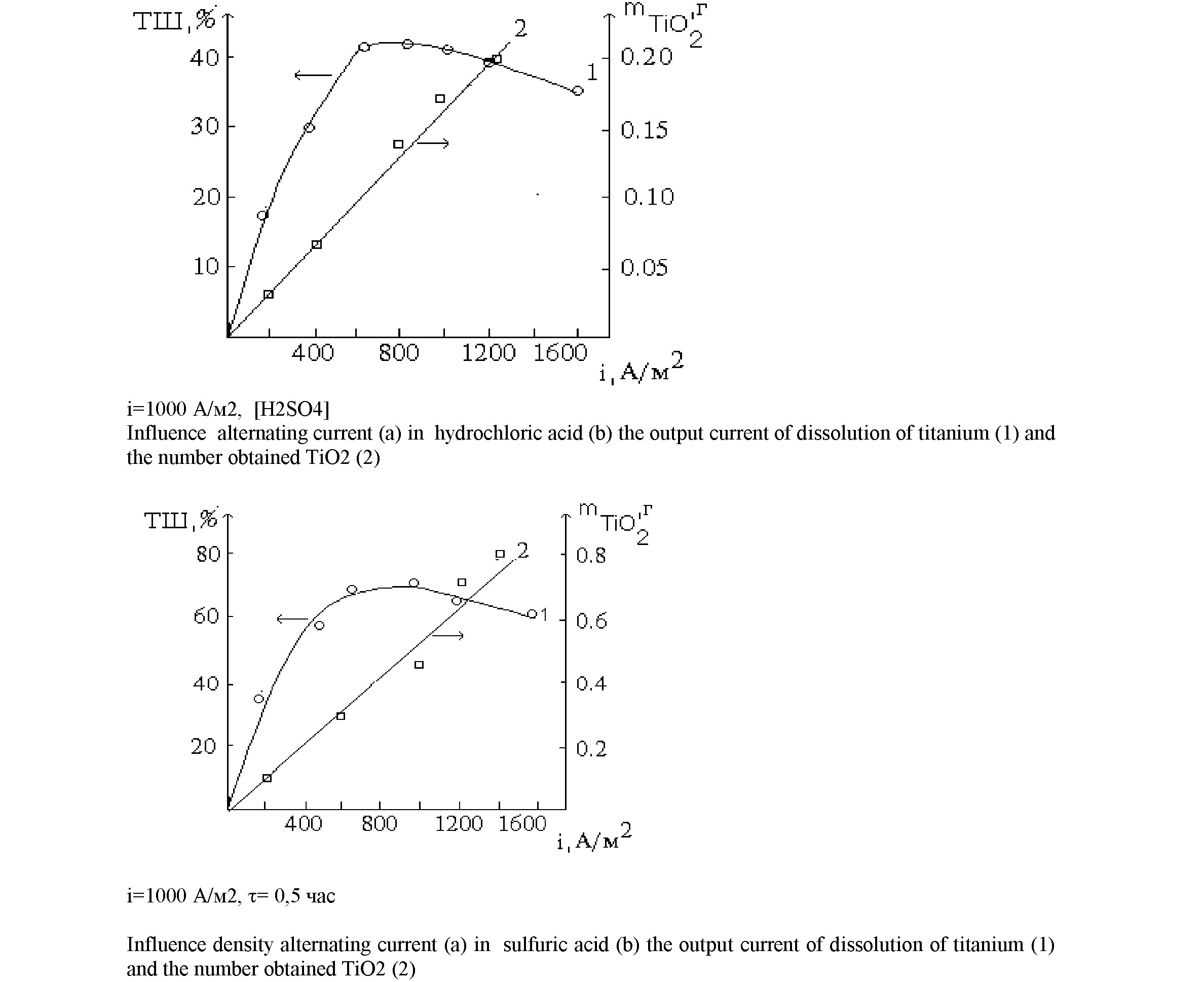
As shown by the results of studies in sulfuric acid solution with increasing current density (900 A / m2), the current efficiency of dissolution of titanium at first increases and then decreases (Figure 3). This phenomenon can be explained by the fact that at high current density in the anodic half cycle on the surface of metal oxide films are formed:
Ti+2H2O=TiO2 +4H++4e- E=-0,90 B
The sulfuric acid and hydrochloric acid solution by dissolving titanium into the solution becomes purple, which indicates the formation of sulfate and chloride, titanium (III). To the obtained salts of titanium (III) was added an alkali, the precipitate thus formed was separated, dried (100 0C), calcined (600 - 8000S) and titanium dioxide were obtained in two modifikatsiyah- rutile and anatase:
Ti2(SO4)3+NH4OH>Ti(OH)3^ + (NH4)2SO4 (6)
O 2
Ti(OH)3 > Ti(OH)4 (7)
600-700С _
Ti(OH)4 > TiO2 +2Н2О (8)
With increasing acid concentration (1-9 M) output current linearly increases titanium dissolution. Apparently, an increase of the current efficiency due to the dissolution of titanium increase as a chemical dissolution in strongly electronegative metal environments (Figure 4):
Ti+6H+>2Ti3++3H2(9)
The surface chemistry, oxidation, and dissolution kinetics of titanium were measured to establish the mechanisms of passive dissolution in physiological environments. Titanium thin films were immersed in 8.0 mM ethylenediamine- tetraacetic acid in simulated interstitial electrolyte (EDTA/SIE) and maintained at 370C, 10% O2, 5% CO2 and 7.2 pH for periods of time up to 3200 h (133 days). Two immersion schemes were employed: the integral sequentially determined the titanium released into a solution of accumulated dissolution products; and the differential continuously replenished the test solution. The solutions were analyzed for titanium by electrothermal atomic absorption spectrometry (EAAS), and the sample surfaces were analyzed by Auger electron spectroscopy (AES) and x-ray photoelectron spectroscopy (XPS) to determine oxide composition, stoichiometry, and thickness. Prior to immersion two types of hydroxyl (OH) groups were distinguished on the TiO2 surface. Upon immersion, the chemistry of the surface changed as a function of immersion: the presence of OH groups increased and P (nonelemental) was detected at the
surface. The dissolution kinetics obeyed a two-phase logarithmic model, where the transition between phases occurred simultaneously with the adsorption of the P-containing species. The dissolution kinetics depended on surface reactions, electric field strength, and molecular diffusion. These mechanisms explain the observed dependence of dissolution kinetics on the properties of the surface oxide and solution ligands.
Investigation of the effect on the frequency of the AC current output dissolving titanium showed that with increasing frequency of the alternating current of 50 Hz output current at first increases and then decreases. At high frequencies of alternating current (50-100 Hz), the recovery of the oxide film on the surface of the titanium for a short time in the cathodic half-hindered, so a decrease in value of the output current.
During the study of electrochemical polarization behavior of titanium at AC in acidic solutions, there is dissolution of titanium after electrolysis. Therefore, we investigated the chemical dissolution of titanium after prior activation of an alternating current. Since the dissolution of titanium was performed in a reducing hydrogen atmosphere, the oxide film on the metal surface is not formed and there is a chemical dissolution of metal.
As a titanium oxide layer is always formed in air, freshlypolished titanium dipped in a corrosive medium takes at least 24h to attain a reproducible value of the stationary state current and OCP. The stable value expected i.e —0.66 V is obtained in strong acids after dissolution of the air-formed oxide layer . The 132
evolution of the rest potential of titanium in acidic media has been studied by several authors. Its value strongly depends on th e concentration of the acid. In concentrated solutions, titanium rapidly attains the active state and corrosion occurs rapidly. K. Azumi et al. [21] have studied the behaviour of titanium electrodes during long-term immersion experiments. The corrosion resistance was evaluated by monitoring the OCP of the electrodes, some samples being previously polarized at 10 V in order to form an anodic oxide film. A potential shift in the positive direction usually means the growth of the oxidefi lm. Such a phenomenon was observed even in deaerated neutral solutions. Thomas and Nobe have studied the corrosion of Ti in 0.5 M H2SO 4. They have found that the OCP of the metal was in the passive range (400-700 mV vs SCE). However, if the electrode was first pretreated in 1M HF, a rapid decrease of the OCP was observed and the titanium dissolution occurred as the active state was reached (corrosion potential: E cor = -680 mV vs SCE). That rapid decrease of the OCP is called “self-activation”. The same behaviour was observed by Brauer and Nann who noticed that the time necessary for the appearance of self-activation was decreasing with increasing molar concentration of H2SO4 CONCLUSIONS
The titanium electrode is passivated by anodic polarization before being in contact with the corrosive solution of H2SO4, the self activation does not appear rapidly. Blackwood et al. concluded that that phenomenon was related to the regular dissolution of the oxide film. It was shown that thinning of the oxide film in 3 M H2SO4 at OCP occurs by uniform dissolution at constant rate rather than by localized attacks . These results were obtained by in situ ellipsometry.