Summary
Citric acid is one of the most common products which have a never ending demand in the global market. It plays a pivotal role in food and beverage industries and pharmaceutical, chemical cosmetic, and other industries for applications such as acidulation, antioxidant, flavor, enhancement, preservation, and plasticization and as a synergistic agent. Citric acid fermentation is one of the primitive fermentations but still its production is increasing with passage of time. In 2007, its global production has exceeded 1.6 million tons.
Key words: Aspergillus niger, citric acid, surface cultivation, submerged cultivation .
Introduction
This report provides a review about the biotechnological production of citric acid starting from the physicochemical properties and industrial applications, mainly in the food and pharmaceutical sectors. Several factors affecting citric acid fermentation are discussed, including carbon source, nitrogen and phosphate limitations, pH of culture medium, aeration, trace elements and morphology of the fungus. Special attention is paid to the fundamentals of biochemistry and accumulation of citric acid. Technologies employed at industrial scale such as surface or submerged cultures, mainly employing Aspergillus niger, and processes carried out with Yarrowia lipolytica, as well as the technology for recovering the product are also described. Finally, this review summarizes the use of orange peels and other by-products as feedstocks for the bioproduction of citric acid.
Purpose : to clarify the current situation of the citric acid market and identify the positive factors of using alternative methods of biotechnological production of this product.
Fungi, in particular Aspergilli, are well known for their potential to overproduce a variety of organic acids. These microorganisms have an intrinsic ability to accumulate these substances and it is generally believed that this provides the fungi with an ecological advantage, since they grow rather well at pH 3 to 5, while some species even tolerate pH values as low as 1.5.
Aspergillus niger- aerobic organism , so to create the best conditions of aeration we use Erlenmeyer flasks.
- All the necessary salts and sucrose contribute to the flask with thoroughly cleaned pipettes.
- Flasks with medium inoculated with the fungus Aspergillus niger, closed with cotton plugs.
- affix the label indicating the appropriate variants.
For greater convenience we have made the fungus( Aspergillus Niger) reseeding.

Organic acid production can be stimulated and in a number of cases conditions have been found that result in almost quantitative conversion of carbon substrate into acid. This is exploited in large-scale production of a number of organic acids like citric-, gluconic- and itaconic acid. Both in production volume as well as in knowledge available, citrate is by far the major organic acid. Citric acid (2-hydroxy-propane- 1,2,3-tricarboxylic acid) is a true bulk product with an estimated global production of over 900 thousand tons in the year 2000. Till the beginning of the 20th century, it was exclusively extracted from lemons. Since the global market was dominated by an Italian cartel, other means of production were sought. Chemical synthesis was possible, but not suitable due to expensive raw materials and a complicated process with low yield. The discovery of citrate accumulation by Aspergillus niger led to a rapid development of a fermentation process, which only a decade later accounted for a large part of the global production.
The application of citric acid is based on three of its properties: (1) acidity and buffer capacity, (2) taste and flavour, and (3) chelation of metal ions. Because of its three acid groups with pKa values of 3.1, 4.7 and 6.4, citrate is able to produce a very low pH in solution, but is also useful as a buffer over a broad range
82
of pH values (2 to 7). Citric acid has a pleasant acid taste which leaves little aftertaste. It sometimes enhances flavour, but is also able to mask sweetness, such as the aspartame taste in diet beverages. Chelation of metal ions is a very important property that has led to applications such as antioxidant and preservative. Moreover, it is a "natural" substance and fully biodegradable. Citric acid (2-hydroxy-propane-1,2,3-tricarboxylic acid) derives its name from the Latin word citrus, a tree whose fruit is like the lemon. Citric acid is a tricarboxylic acid (Figure 1) with a molecular weight of 210.14 g/mol, which contains three carboxylic functional groups with three different values of pKa (3.1, 4.7, and 6.4). It is a primary metabolic product formed in the tricarboxylic acid (or Krebs) cycle and is found in small quantities in virtually all plants and animals, being isolated from lemon juice in 1784.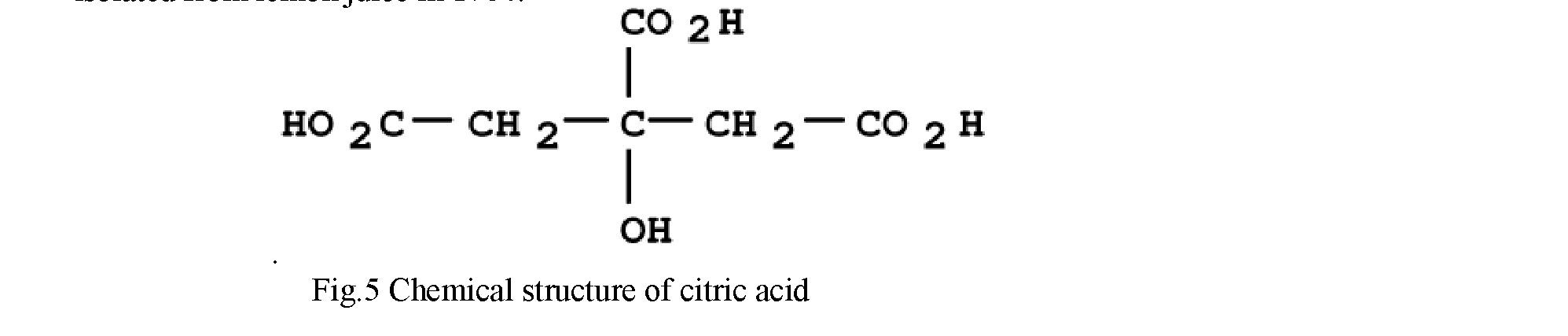
Citric acid was first commercially-produced in England around 1826 from imported Italian lemons (lemons contain 7-9% citric acid). Lemon juice remained the commercial source of citric acid until 1919, when the first industrial process using Aspergillus niger started in Belgium. Currently, the extraction of citric acid is limited to some small factories in Mexico and Africa.
Citric acid was synthesized from glycerol by Grimoux and Adams and later from symmetrical dicloroacetone. Other routes have been published from various synthetic materials since then, but chemical methods have so far proved uncompetitive.
Wehmer was the first to demonstrate that Citromyces (now Penicillium) accumulated citric acid in a medium containing sugar and inorganic salts. Since then, many organisms have been found to accumulate citric acid: A. niger, Aspergillus awamori,Aspergillus nidulans, Aspergillus fonsecaeus, Aspergillus luchensis, Aspergillus phoenicus, Aspergillus wentii, Aspergillus saitoi, Aspergillus lavus, Absidia sp.Acremonium sp., Botrytis sp., Eupenicillium sp., Mucor piriformis, Penicillium janthinellum, Penicillium restrictum, Talaromyces sp., Trichoderma viride and Ustulina vulgaris .
Currie found that some strains of A. niger were able to grow in a medium containing sugars and salts at an initial pH of 2.5-3.5. Throughout their growth, these strains excreted large amounts of citric acid, which established the basis for industrial production.
Besides fungi, it is known that several yeasts produce citric acid from n-alkanes and carbohydrates , especially species belonging to the genera Candida, Hansenula, Pichia, Debaromyces, Torula, Torulopsis, Kloekera, Saccharomyces, Zygosaccharomyces and Yarrowia. During the 60s and 70s oil was cheap, and citric acid was produced industrially from this source by Candida sp., including C. tropicalis, C. catenula, C. guilliermondii and C. intermediate .
Conclusion.Many microorganisms can be employed to produce citric acid, A. niger is still the main industrial producer. In fact, specific strains that are able to overproduce citric acid in different types of fermentation processes have been developed. The theoretical yield is 112 g of anhydrous citric acid per 100 g of sucrose. However, in practice, due to losses during trophophase, the yield of citric acid from these strains often does not exceed 70% of the theoretical yield on carbon source. Despite a long and successful history of producing citric acid, there is not unanimous explanation of the biochemical basis of the process.
According to all this information you can see that the most optimal variant and economically profitable producer of citric acid are A.Niger strains. Also biotechnological obtaining of citric acid is ecologically safe method , what is important in now days.
References:
- .Adham N.Z. Attempts at improving citric acid fermentation by Aspergillus niger in beet-molasses medium.Bioresour Technol.
- .Aghdam M.G., Taherzadeh M. Production of citric acid by solid state fermentation. J Biotechnol.2008
- .Ali S., Ashraf H., Ikram U. Enhancement in citrate production by alcoholic limitation. J. Biol. Sci.2002