Introduction. Electrodialysis can be applied to the continuous-flow type of operation needed in industry. Multi-membrane stacks can be built by alternately spacing anionic- and cationic-selective membranes. Among the technical problems associated with the electrodialysis process, concentration polarization is perhaps the most serious. Other problems in practical applications include membrane scaling by inorganics in feed solutions as membrane fouling by organics [1-2].
Purpose of the study. The technologies discussed in this chapter are changing relatively rapidly, and hence you need to stay in touch with vendor developments. Remember to refer to the Glossary at the end of the book if you run across any terms that are unfamiliar to you.
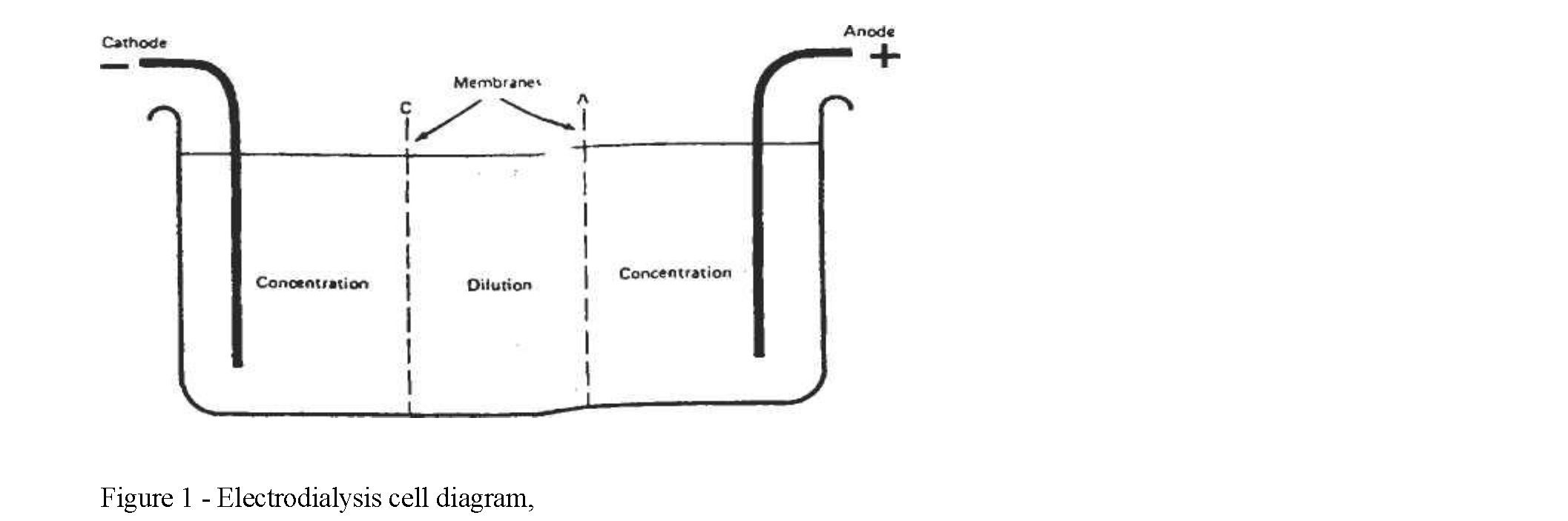
Materials and methods. Membrane processes can be operated as: Dead-end filtration; and Cross-flow filtration, Electrodialysis aparat Results and discussion. The principle behind electrodialysis is that electrical potential gradients will make charged molecules diffuse in a given medium at rates far greater than attainable by chemical potentials between two liquids as in conventional dialysis. When a DC electric current is transmitted through a saline solution, the cations migrate toward the negative terminal, or cathode, and the anions toward the positive terminal, the anode. By adjusting the potential between the terminals or plates, the electric current and, therefore, the flow of ions transported between the plates can be varied.
There are five types of membrane processes, which are commonly used in water and wastewater treatment:
- Through these processes dissolved substances and/or finely dispersed particles can be separated from liquids. All five technologies rely on membrane transport, the passage of solutes or solvents through thin, porous polymeric membranes.
- A membrane is defined as an intervening phase separating two phases forming an active or passive barrier to the transport of matter. Membrane processes can be operated as: (1) Dead-end filtration; and (2) Cross - flow filtration.
- filtration refers to filtration at one end. A problem with these systems isfrequent
- membrane clogging. Cross-flow filtration overcomes the problem of membrane clogging and is widely used in water and wastewater treatment.
The membrane itself is a polymeric coating or extrusion with inverted conicalshaped pores. Membrane filters do not plug because the pore diameter is smaller at the top, which is the point of contact with the wastewater. Material passing through the membrane passes unimpeded through the membrane structure, therefore eliminating accumulation of material within the filter. Wastewater is pumped across the membrane surface at high flow rates. This parallel fluid flow eliminates the cake-like build-up typical of conventional filters such as bags and cartridges which must be frequently replaced. Some wastewater contaminants slowly accumulate on the membrane surface, forming a thin film, during normal operating conditions. This fouling process is normal and causes the filtration rate to slowly decrease with time. When membranes no longer produce clean water at the desired rate they are cleaned in place with soap and water and returned to service. Membranes can be repeatedly cleaned for years of productive, dependable service prior to replacement[2-5].
Most of these processes are oftentimes used with chemical mechanical polishing (CMP), which is fast becoming the established technology for planarizing multilevel devices. This process requires large quantities of ultrapure water for rinsing slurry particles off the polished wafers. Treatment by this method is generally needed in order to maintain an acceptable level of total suspended solids (TSS) in industrial
wastewater effluent. With large quantities of particles in the CMP wastewater stream, crossflow filtration is the most economical method for TSS removal.
Conclusions. Nowadays, electrochemical technologies have reached such a state that they are not only comparable with other technologies in terms of cost but also are more efficient and more compact. For some situations, electrochemical technologies may be the indispensable step in treating wastewaters containing refractory pollutants.
References
- Climate Change 2001. Synthesis report.,Cambrige University Press, UK, 2003.
- G. I. Marchuk, Mathematical Modelling in Environmental Problems ,Nauka, Moscow ,1982.
- G. I. Marchuk, Adjoint Equations and Analysis of Complex Systems Kluwer Academic Publishers, Dordrecht, 1995.
- V. V. Penenko, Methods of Numerical Modelling of Atmospheric Processes, Gidrometeoizdat, Leningrad, 1981.
- V. V. Penenko and A. E. Alojan.Models and methods for environment protection problems Nauka, Novosibirsk, 1985.