Abstract
Acute myocardial infarction is a frequent and disabling disease. Ischemic postconditioning, consisting of repeated brief cycles of ischemia-reperfusion performed immediately after reperfusion following a prolonged ischemic insult, dramatically reduces infarct size in experimental models. A recent clinical study demonstrated that repeated brief episodes of inflation-deflation of the angioplasty balloon performed immediately after re-opening of the culprit coronary artery reduced infarct size by 36% in patients with current acute myocardial infarction. This proof-of-concept study identified lethal reperfusion injury as a new target for future pharmacological treatments. Future therapeutic strategies for acute myocardial infarction should include mimetics of postconditioning.
Key words: Ischemia, myocardial infarction, postconditioning, reperfusion
Miokardni ishemik postkonditsionirlashning protektiv xususiyatlari O'tkir miokard infarkti tez uchraydigan jiddiy kasallik. Ishemik postkonditsionirlash ya‘ni qisqa fursatli ishemiya-reperfuziya epizodlari eksperimental tajriba modellarida infarkt hajmini kamaytirishini ko‘rsatdi. Shu bilan birga so'nggi klinik tadqiqot natijalariga ko’ra, infarktga olib kelgan arteriyaning angioplastika ballonlash jarayonida takroriy va qisqa inflyatsiya-deflyatsiyasi o'tkir miokard infarkti bilan og'rigan bemorlarda infarkt hajmini 36% ga kamaytiradi. Bu ma‘lumotlar o‘tkir miokard infarktining kelajak davolash strategiyasiga farmakologik postkonditsionirlashni olib kirish imkoniyati mavjudligini ko‘rsatadi.
Kalit so'zlar: ishemiya, miokard infarkti, postkonditsionirlash, reperfuziya
Postconditioning protects the human heart
Acute myocardial infarction is a frequent and disabling disease, with heart failure as a common outcome. Infarct size is recognized as a major determinant of myocardial functional recovery and mortality after acute myocardial infarction [1]. Limitation of infarct size appears thus to be an appropriate strategy with which to prevent postinfarction heart failure and to improve survival. Current strategies to limit infarct size include emergency re-opening of the culprit coronary artery (coronary angioplasty or thrombolysis) and prevention of re-occlusion (antiplatelet agents). It is not questionable that myocardial reperfusion improves outcomes. Treatments adjunctive to reperfusion, including в-blockers and angiotensin converting enzyme inhibitors, can ameliorate morbidity or mortality, or both, but not via a reduction in infarct size.
Zhao et al [2] first reported that, in the dog heart, three episodes of 30 s of reperfusion and 30 s of ischemia performed immediately after a prolonged 60 min ischemic insult dramatically attenuated irreversible myocardial injury. This cardioprotective effect was comparable to that observed with the powerful phenomenon of preconditioning [3]. Several other investigators have now been able to reproduce this initial experiment and found that ischemic postconditioning is also efficient in pig, rabbit, rat, and mouse hearts. This description of "postconditioning" demonstrated the existence of a lethal myocardial reperfusion injury and thereby helped us to consider potential new ways to achieve further attenuation of infarct size.
Unlike preconditioning, postconditioning can be triggered at the onset of reperfusion. It can therefore be manipulated in clinical situations in which reperfusion can be controlled, for example during coronary angioplasty or cardiac surgery. In a proof-of-concept trial, Staat et al [4] aimed to determine whether postconditioning might be protective in patients with current acute myocardial infarction. In a
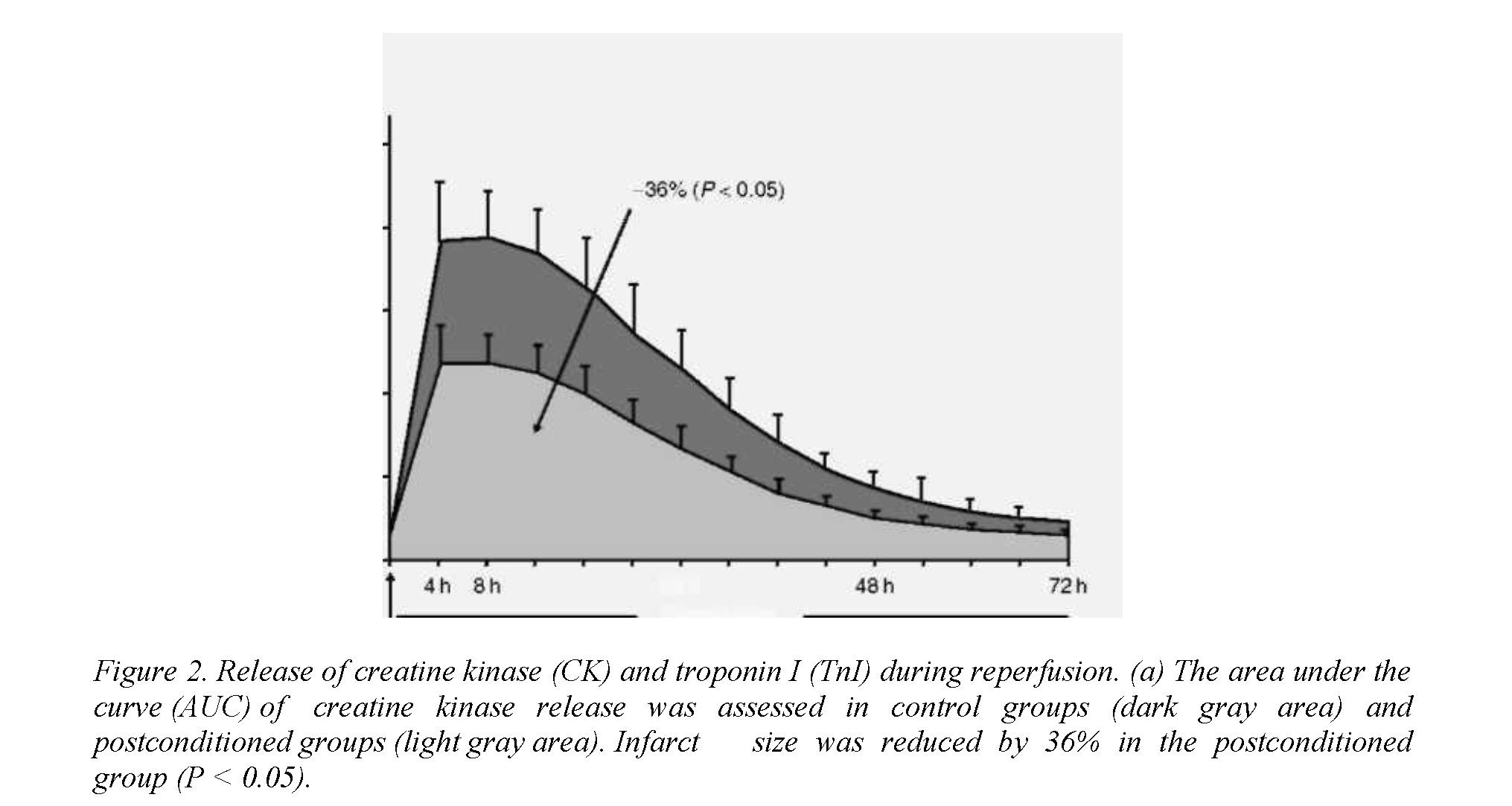
multicenter, controlled, randomized study, they enrolled patients with a first ST-segment elevation myocardial infarction, chest pain of less than 6 h duration, and a need for revascularization via angioplasty. Patients were allocated randomly to either a control or a postconditioned group. Major determinants of infarct size were assessed using methods that would be possible in an emergency clinical situation. The area at risk was estimated by left ventricular angiography. Duration of ischemia was estimated as time from the onset of chest pain to time of re-opening the coronary artery. Patients with overt collateral circulation to the region at risk were excluded from the study. After direct stenting, control patients underwent no additional interventions, whereas postconditioned patients underwent four cycles of inflation - deflation of the angioplasty balloon, starting within 1 min after the initial reperfusion. Each inflation and deflation lasted for 1 min. This procedure was simple and feasible in each patient, and was without any adverse event. Infarct size was first assessed by measuring the release of creatine kinase over the first 4 days of reperfusion. The area under the curve of creatine kinase release was significantly reduced (by 36%; P<0.05) in the postconditioned group compared with the control group. Analysis of covariance revealed that the reduction in infarct size persisted whatever the size of the area at risk (Figure
Revisiting the pathophysiology of acute myocardial infarction
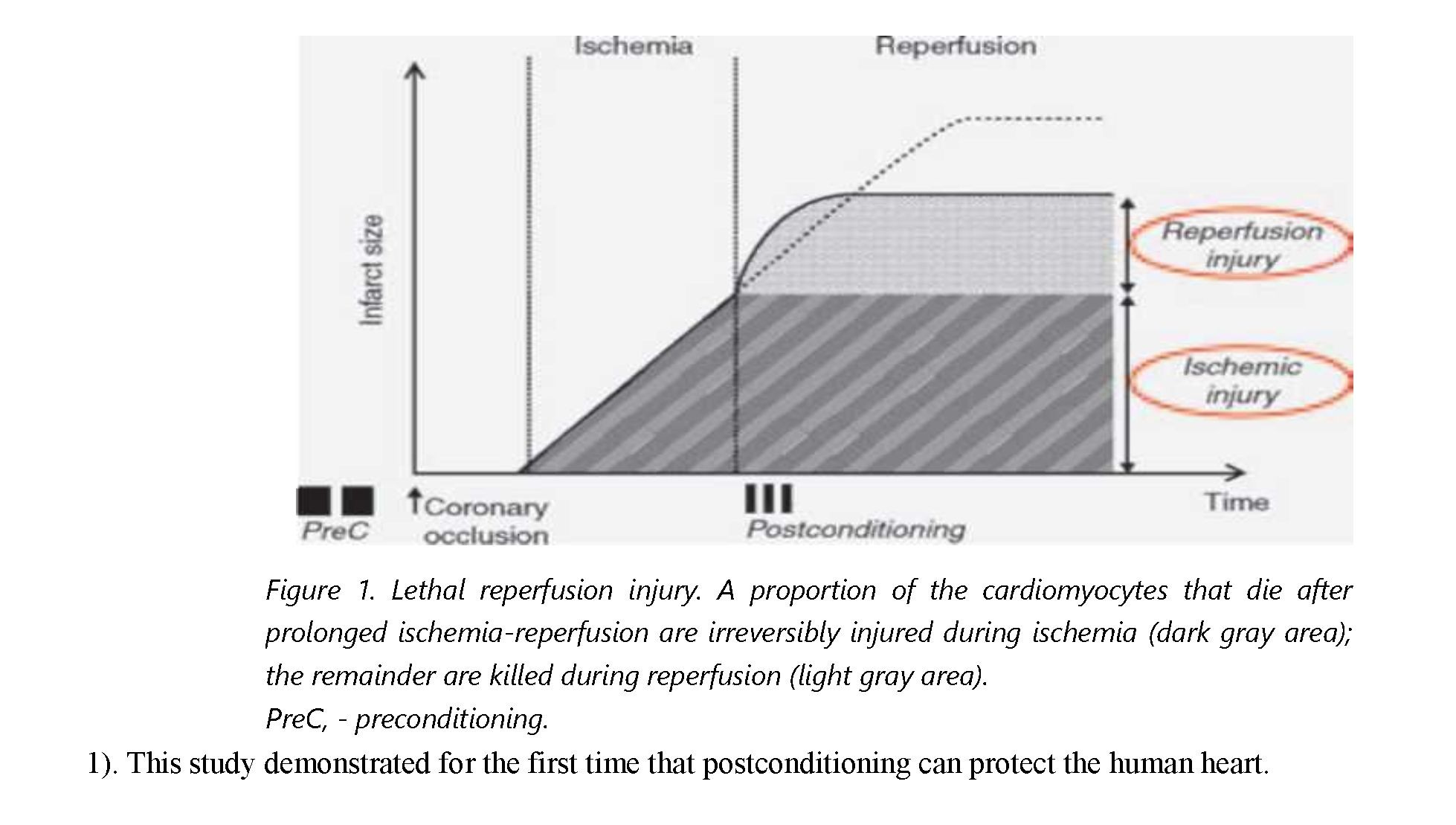
With their initial observation, Zhao et al [2] demonstrated that an unexpected intervention, performed after ischemia during the first minutes of reperfusion, was capable of reducing infarct size by approximately 40%. In other words, 40% of the overall myocardial damage (infarct size) occurs after ischemia - that is, during reperfusion. This observation is apparently paradoxical: reperfusion can both protect and kill. Reperfusion protects simply because it prevents additional lesions that would have occurred if ischemia (and subsequent reperfusion) had persisted. It kills because reflow is associated with dramatic ionic and metabolic disturbances that overwhelm the endogenous defenses of jeopardized cardiomyocytes. Thus the final irreversible damage to cardiac tissue observed after a prolonged ischemiareperfusion represents the superposition, in nearly equal amounts, of irreversible damage occurring before re-opening of the occluded coronary artery (ischemic damage) on irreversible damage occurring just at the time of reflow (reperfusion damage) (Figure 2). According to studies performed in experimental preparations, the window of time during which protection by postconditioning could be triggered would be very narrow (longest duration less than 1 min). Whether a comparable time course applies to the
51
human heart remains to be determined, but it would represent valuable information in the targeting of therapeutic strategies in patients with current acute myocardial infarction.
Postconditioning in clinical practice
The initial study by Staat et al [4] was designed as a proof-of-concept study aimed at determining whether ischemic postconditioning was able to protect the human heart in a clinical setting that would be as close as possible to that of experimental preparations. Although the findings of the trial were very encouraging, key questions remain to be addressed, including: (1) does postconditioning afford a persistent limitation of infarct size?, (2) does postconditioning improve recovery of myocardial contractile function, left ventricular remodeling, and survival?, and (3) is postconditioning feasible in all patients with current acute myocardial infarction?
The protective effect of postconditioning has been assessed as the release of cardiac enzymes over the first 3 days of reperfusion. Complementary studies are required to determine whether myocardial salvage by angioplasty is maintained over time: infarct size should be measured several months after the acute event, by magnetic resonance imaging or single photon emission computed tomography. Investigations are currently in progress to determine whether ischemic postconditioning improves clinical outcomes. It remains to be determined whether postconditioning might reduce mortality.
If one admits that postconditioning may improve patients' prognosis and if the approach becomes widely used in daily practice, two options appear to be available: first, to use it as it is - that is, as an angioplasty intervention - or, secondly, try to extend its availability to all patients by means of a pharmacological mimetic.
Postconditioning by angioplasty is technically simple and safe. Additional studies may help to determine what would be the shortest, safest, most efficient postconditioning procedure. One may question whether there might exist a wider window of time (which is supposedly limited to 1 min in animal species) for triggering postconditioning in patients. It would be useful to determine whether comorbidities (eg, diabetes, lipid disorders, age, hypertension) might attenuate the degree of protection.
A large number of patients achieve reperfusion via thrombolysis; these patients cannot benefit from angioplasty postconditioning. There is then an obvious need for a pharmacological mimetic of ischemic post-conditioning. In view of the narrow window of time defined in experimental studies, one has to consider the possibility that such a postconditioning drug should be administered before reperfusion, in order to obtain significant drug plasma concentration at the time of coronary reflow. It could then be administered in all patients with current acute myocardial infarction, either as an adjunct to 52
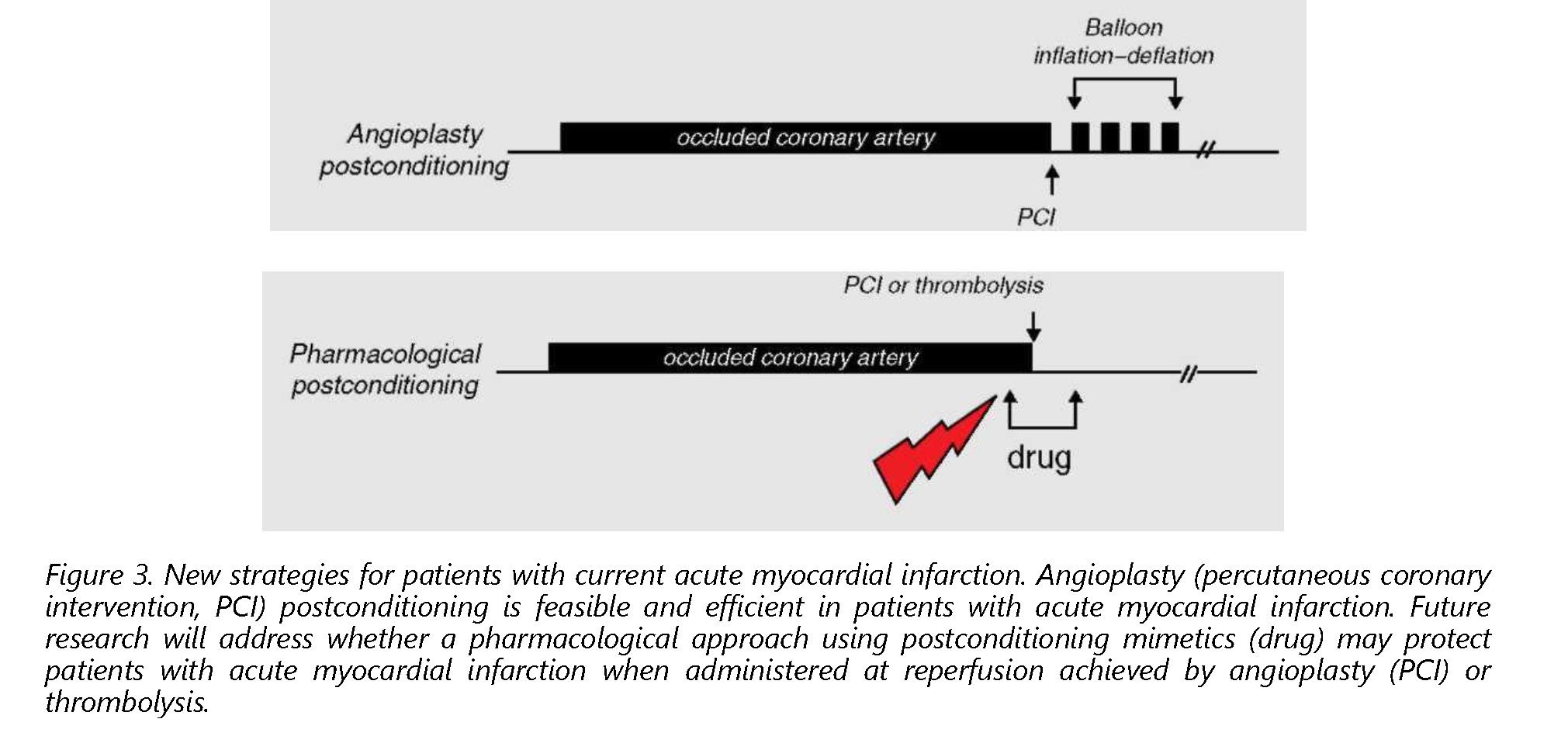
thrombolysis, or even just before coronary angioplasty (in place of ischemic post-conditioning) (Figure 3).
A considerable amount of experimental research has been performed since the discovery of pre- and, more recently, postconditioning. Several molecular targets have been identified as key players in cardioprotection by pre- or postconditioning, including signaling pathways (eg, protein kinase C, phosphatidyl inositol 3-kinase, Akt, endothelial nitric oxide synthase, extracellular-signal regulated kinases 1/2, glycogen synthetase kinase 3b), and, more recently, processes at the level of mitochondria. Growing evidence suggests that a change in mitochondrial permeability is involved in lethal reperfusion injury [5]. At the onset of reperfusion after a prolonged ischemic insult, abrupt accumulation of Ca2+ in the matrix, and overproduction of reactive oxygen species, render the inner mitochondrial membrane permeable through the opening of a nonspecific mega-channel (called the ''permeability transition pore''). The opening of this pore, and the spread of permeability changes among neighboring mitochondria throughout the cardiomyocyte may at some point result in irreversible damage. In experimental preparations, pharmacological inhibitors of the mitochondrial permeability transition (including cyclosporin A) do reduce infarct size to an extent similar to that in ischemic postconditioning [6,7]. Current trials are under way to determine whether inhibition of the transition in mitochondrial permeability may limit infarct size in patients with current acute myocardial infarction.
Postconditioning represents a new major hope of improving the prognosis of patients with acute myocardial infarction.
REFERENCES
- Gibbons RJ, Valeti US, Araoz PA, Jaffe AS. The quantification of infarct size. J Am Coll Cardiol. 2004;44:1533-1542.
- Zhao ZQ, CorveraJS, Halkos ME, etal. Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol. 2003;285:H579-H588.
- Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-1136.
- Staat P, Rioufol G, Piot C, et al. Postconditioning the human heart. Circulation. 2005;112:2143-2148.
- Duchen MR, McGuinness O, Brown LA, Crompton M. On the involvement of a cyclosporin A sensitive mitochondrial pore in myocardial reperfusion injury. Cardiovasc Res. 1993;27:1790-1794. 53
- Argaud L, Gateau-Roesch O, Muntean D, et al. Specific inhibition of the mitochondrial permeability transition prevents lethal reperfusion injury. J Mol Cell Cardiol. 2005;38:367-374.
- Argaud L, Gateau-Roesch O, Raisky O, Loufouat J, Robert D, Ovize M. Postconditioning inhibits mitochondrial permeability transition. Circulation. 2005;111:194-197.