Conventional chrome tanning in leather production produces spent liquors containing significant amounts of chromium and other polluting substances, both organic and inorganic. From the total chromium used for tanning only 60% to 70% is utilized, while the rest 30 to 40% remains in the spent tanning liquor, which is normally sent to a wastewater treatment plant. This inefficient use of chromium and its release to the environment has to be compensated by designing a good recovery and recycling scheme. The recovery of chromium from spent tanning and re-tanning baths provides a significant economic advantage in terms of both its reuse and the simplification of the processing of global wastewaters [1-2].
Several recovery techniques such as chemical precipitation, membrane processes, adsorption, redox adsorption, and ion exchange have been proposed for this purpose. Among these membrane process offer very interesting opportunities for the recovery and recycling of primary resources from spent liquors of unit operations such as soaking, unhairing, degreasing, pickling, dyeing, and chromium tannage. Studies showed that the application of nanofiltration (NF) and reverse osmosis (RO) in combination can provide better recovery of unreacted chromium from high concentrated spent tanning effluent. However, this technique is being challenged by the presence of considerable biological oxygen demand (BOD) and proteins, which can cause fouling and subsequent system failure, either temporarily or permanently [1-2].
In practice, there are two ways of chrome recycling methods which are widely practiced: these are direct and indirect recycling. The direct form entails spent float being recycled direct to the chrome tanning processing for re-use. While, the indirect form entails precipitating and separating the chrome from the float containing residual chrome, and then re-dissolving it in acid for re-use. The efficiency of both methods can be very high (more than 90%); it depends on the effectiveness of the float collection process and the recycling/reusing technique. Of the two approaches, chrome recovery is more widely used than chrome precipitation. However, it is mandatory to adopt and practice the new technologies which are more efficient in recovering this chemical, which is a big challenge to tanning industry [1-2].
This study was undertaken to investigate the removal of total chromium (Both Hexavalent and Trivalent) from synthetic and industrial effluents by chemical means in order to achieve the 100% removal efficiency. The study was conducted in three phases. In phase 1, the optimum pH, effective dosage and reaction time were optimized using Sodium Metabisulphite as reducing agent for the reduction of hexavalent chromium to trivalent chromium. As a result, pH of 2 and dosage of 80 mg/l was found to be optimized condition for the Cr(VI) reduction. Phase 2 studies were carried out to evaluate the potential of different precipitating agents in removal of Cr(III). In the Phase 2 studies optimum conditions for various precipitating agents were obtained. In phase 3 the experiments were carried out on industrial wastewater for the removal of hexavalent and trivalent chromium with the optimum operating conditions which were obtained from phase 1 and phase 2. The results from phase 3 on industrial wastewater showed the results in par with the synthetic sample except for ferric chloride. The combination of sodium hydroxide and calcium hydroxide is found to be best precipitating agent with dosage of 100 mg/l at pH 7. The volume of sludge produced by the same is very minimal compared to other precipitating agents.
As human needs increase and civilization changes, more and more finished products of different types are required. Accordingly, large number of industries born and grown in every country[1] Process waste streams from the mining operations, metal-plating facilities, power generation facilities, electronic device manufacturing units, and tanneries may contain heavy metals at concentrations exceeding the local discharge limits. These waste streams contain toxic heavy metals such as chromium, cadmium, lead, mercury, nickel, and copper. They are not easily removed without specialized or advanced treatment. Chromium is a common pollutant introduced into natural waters due to the discharge of a variety of industrial wastewaters. On the other hand, chromium based catalysts are also usually employed in various chemical processes, including selective oxidation of hydrocarbons. In India and all over the world, Chromium (Cr) is dominant in most of the effluent streams as compared to other heavy metal ions[2]. Caio F etal found that a reduction of more than 1.0 X 105 and 4.0 X 105 fold in total chromium and
hexavalent chromium concentrations, respectively, was observed by employing steel wool masses as low as 0.4420 g to 30 ml solutions of wastewater [3].The optimum pH for the precipitating chromium from tannery wastewater is 7.7-8.2 with a lime concentration (2g/100ml) and the effective settling rate was 120min. The bioremoval of Cr (VI) using actinomycetes is a suitable mean for reducing the tannery wastewater toxicity. The combination between the chemical precipitation and the biological removal of chromium from tanning wastewater make it meet the environment safely [4].The percent removal of metal ions increases to about 99 % with increasing the MgO dose to some limits. The optimum values of MgO doses were found to be 1.5-3.0 g/l. The pH value ranges are 9.5 to 10 with MgO precipitant and pH of 11.5 to 12 with CaO precipitant [5][6]. Combinations of ferric chloride and polymer at different ratio will also results in better removal efficiencies of the metals in the range 84 - 97% for total chromium, 69-90% for zinc and 6972% for total iron, also less sludge was produced [7]. There are many factors which affect the efficiency of precipitation (pH, nature and concentration of hazardous substances in water, precipitant dosage, temperature, water balance etc.,). In practice, the optimum precipitant and dosage for a particular application are determined by a “trial and errof’ approach using Jar test [8]. Moreover Ferrous sulphate requires pH =1for complete reduction as compared to sodium metabisulphite which requires a pH of about 2 [9].
Materials and methodology
- Preparation of Synthetic hexavalent chromium sample The potassium dichromate (K2Cr2O7) is used as source of hexavalent chromium. A stock solution of 1000mg/l of Cr(VI) is prepared by dissolving 2.8287g of potassium dichromate in 1000ml distilled water. The solution is diluted as required to obtain standard solutions containing 10- 15mg/l of Cr(VI). pH adjustments were carried out using Sulphuric acid and sodium hydroxide.
- Analysis of Hexavalent Chromium:
The concentration of the hexavalent chromium ions in the sample is determined spectrophotometrically by developing a red- violet colour with 1,5 di phenyl carbazide in acidic condition solution as a complexing agent. The absorbance of the red - violet coloured solution is read at 530 nm- 540 nm after 20 mins [3].
Atomic absorption spectrophotometer A203 Version -04 was used to measure the total chromium concentration. In AAS fuel used was acetylene C2H2 and the oxidant used was air or Nitrous oxide for strong flames. Calibration of the AAS was done according to the equipment manual using certified standards and the analysis of calibrated standards was attained to ensure the accuracy of results.
The experiments were carried out under different pH condition and different dosage of Sodium MetabiSulphite and precipitating agents providing different contact time. The experimental set up is as shown in the fig 1.
Results and discussions. A. Optimum condition for chromium (VI) Reduction: Sodium Metabisulphite is used as reducing agent for hexavalent chromium reduction. Jar test method has been used to determine the effects of each parameter (Six next step samples were mixed for 25 mins with the speed of 20-30 rpm. The precipitate formed was allowed to settle completely. Supernatant was withdrawn from the beaker and analysed for hexavalent chromium and total chromium. was diluted to obtain concentration of Cr(VI) in the range 10 mg/lt.
Table 5.Summary of results of various precipitating reagents in removal Cr(III) in Synthetic Sample.
From the results of synthetic samples the optimum operating conditions for different precipitating agents were recorded. The best operating conditions are shown in table 1. The experimental results for chromium removal 83
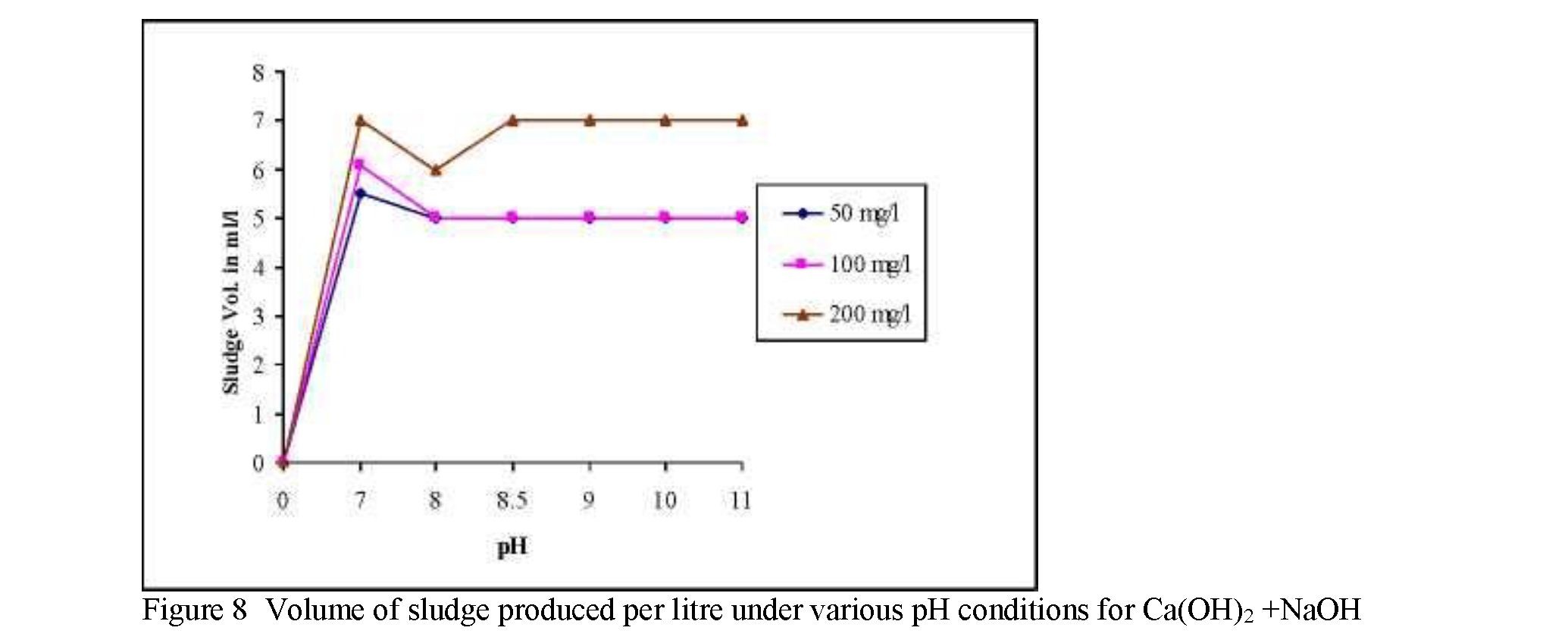
using combination of NaOH and Ca(OH)2 are shown in Fig 2-4. The effect of pH on the chromium precipitation using combination of NaOH and Ca(OH)- is as shown in Fig 2. Also the volume of sludge produced and variation in pH after the treatment are shown in the Fig 3 and 4. Fig 5 shows the efficiency of sodium hydroxide,
Table l.Summary of results of various precipitating reagents in removal Cr(III) in Synthetic Sample. (Concentration of Cr(III) in synthetic sample after reduction process = 10mg/lt) beakers have been used for each stage) and 500ml of synthetic sample was added to the each beaker. Sodium metabisulphite is added to each beaker to determine the optimum dosage, pH and contact time. The pH of the sample is maintained using sulphuric acid with mixing speed of 50 rpm. The sample was analyzed to know the hexavalent chromium concentration after reduction process.As a result, pH of 2 and dosage of 80 mg/l was found to be optimized condition for the Cr(VI) reduction at contact time of 5 mins. D. Optimum condition for chromium (III) Precipitation: After the complete
reduction of hexavalent chromium the precipitating agents viz., Sodium Hydroxide (NaOH), Combination of Sodium Hydroxide and Calcium Hydroxide (Ca(OH)2+NaOH) and ferric Chlorides (FeCl3) were added to each sample separately. In order to mix the solution, sample were taken to jar apparatus and samples were mixed for 5 Mins with the speed of 100 rpm as first step. In the combination of NaOH and Ca(OH)2 and Ferric chloride in removing chromium. Both combination of NaOH and Ca(OH)2 and Ferric chloride shows the 100 % chromium removal efficiency. The variation in pH and Volume of sludge produced has also been recorded (Fig 6 and 7). Compare to all NaOH produced large volume of sludge since because sludge produced by NaOH is gelatinous and light in nature but whereas sludge produced by Ferric chloride in voluminous with dense in nature. The combination of NaOH and Ca(OH)2 has produced minimum volume of sludge compared to other two. Hence the combination of NaOH and Ca(OH)2 is considers as best precipitating agent among three in precipitation of chromium.
A number of comparative experiments were conducted in third phase on industrial wastewater. The typical industrial wastewater characteristics showed higher concentration of hexavalent chromium concentration as 3222.6 mg/lt. Firstly
REFERENCES
- Рипан Р., Четяну И. Неорганическая химия- Мир,2000, Т.2,- 650 с.
- Химия: справ изд. / Шретер В., Лаутеншлегер К.Х. - М. Химия,-2005, -420с.
- В.В. Некрасов. Основы общей химии.- Химия-2004,Т.2,-352 с.
- Ахметов Н.С. Общая и неорганическая химия. 2-е изд.- М: Высшая школа, 2008 ,-С. 640-642
- Глинка Н.Я. Общая химия. - Л., Химия, 2006,- 640 с.
- T. T. Shen, “Industrial pollution prevention,” 2nd Edition, Springer, pp. 40, 1999.
- M. M. Altaf, F. Masood, and A. Malik, “Impact of long-term application of treated tannery effluents on the emergence of resistance traits in rhizobium sp. isolated from trifolium alexandrinum,” Turkish Journal of Biology, Vol. 32, pp. 1-8, 2008.
- V. J. Sundar, J. R. Rao, and C. Muralidharan, “Cleaner chrome tanning—emerging options,” Journal of cleaner production, Vol. 10, pp. 69-74, 2002.
- B. Wionczyk, W. Apostoluk, and W. A. Charewicz, “Solvent extraction of chromium (III) from spent tanning liquors with Aliquat 336,” Journal of Hydrometallurgy, Vol. 82, No. 1-2, pp. 83-92, 2006.
- M. Marchese, A. M. Gagneten, M. J. Parma, and P. J. Pave, “Accumulation and elimination of chromium by freshwater species exposed to spiked sediments,” Archives of Environ Contamination and Toxicology, Springer, Vol. 55, No. 1, pp. 603-609, 2008.
- S. Avudainayagam, M. Megharaj, G. Owens, R. S. Kookana, D. Chittleborough, and R. Naidu, “Chemistry of chromium in soils with emphasis on tannery waste sites,” Review of Environmental Contamination And Toxicology, Springer, Newyork, Vol. 178, pp. 53-91, 2003.
- J. C. Akan, E. A Moses, and V. O. Ogugbuaja, “Assessment of tannery industrial effluent from Kano metropolis, Nigeria,” Asian Network for Scientific Information, Journal of Applied Science, Vol. 7, No. 19, pp. 2788-2893, 2007.