Relationships between amplitude of penicillin- induced generalized epileptiform signals in different zones of the brain cortex (occipito- frontal bilateral leads, as well as occipital, temporal and frontal bipolar leads along with paleocerebellar lead) were investigated in WAG/rij rats using multiple linear regression method of analysis. Results were expressed in the form of polycyclic multigrafs (multidimensional presentation) with the identification of significant (p<0,1) negative or positive influences of certain direction between sources of signals. The prevalence of the involvement of left hemispheral cortex into creation of positive type of forward, and back- directed influences was identified, while greater ability to be involved in the creation of negative type of relationships was established for the occipital zone of cortex during generalized seizure activity development. Typical absence spike-wave activity in WAG/rij rats was characterized by the prevalence of positive relationships, which were present between almost all inspected zones of cortex. Besides, minimization of mutually negative relationships was also characteristic for WAG/rij ECoG activity.
Key words: epileptic activity, absence epilepsy, seizures, multiple linear regression, electroencephalography.
Introduction. The aim of the work was to identify the relationships which were created between brain structures in the period of converting of spike- wave absence activity, which is typical for WAG/rij rats to generalized one. Such a transition was provoked by solution of sodium salt of benzilpenicillin administration. As a structures for investigations the cortical ones of both hemispheres as well as paleocerebellar cortex (flocculo- nodular lobule) were chosen. Usage of these structures was explained by well known role of cortical neuronal population in spike-wave rhythm generation [1, 2, 3] as well as a ‘antiepileptic’ properties of
paleocerebellum. Besides, the problem of interhemispheric functional relationships was also in scope of the investigation.
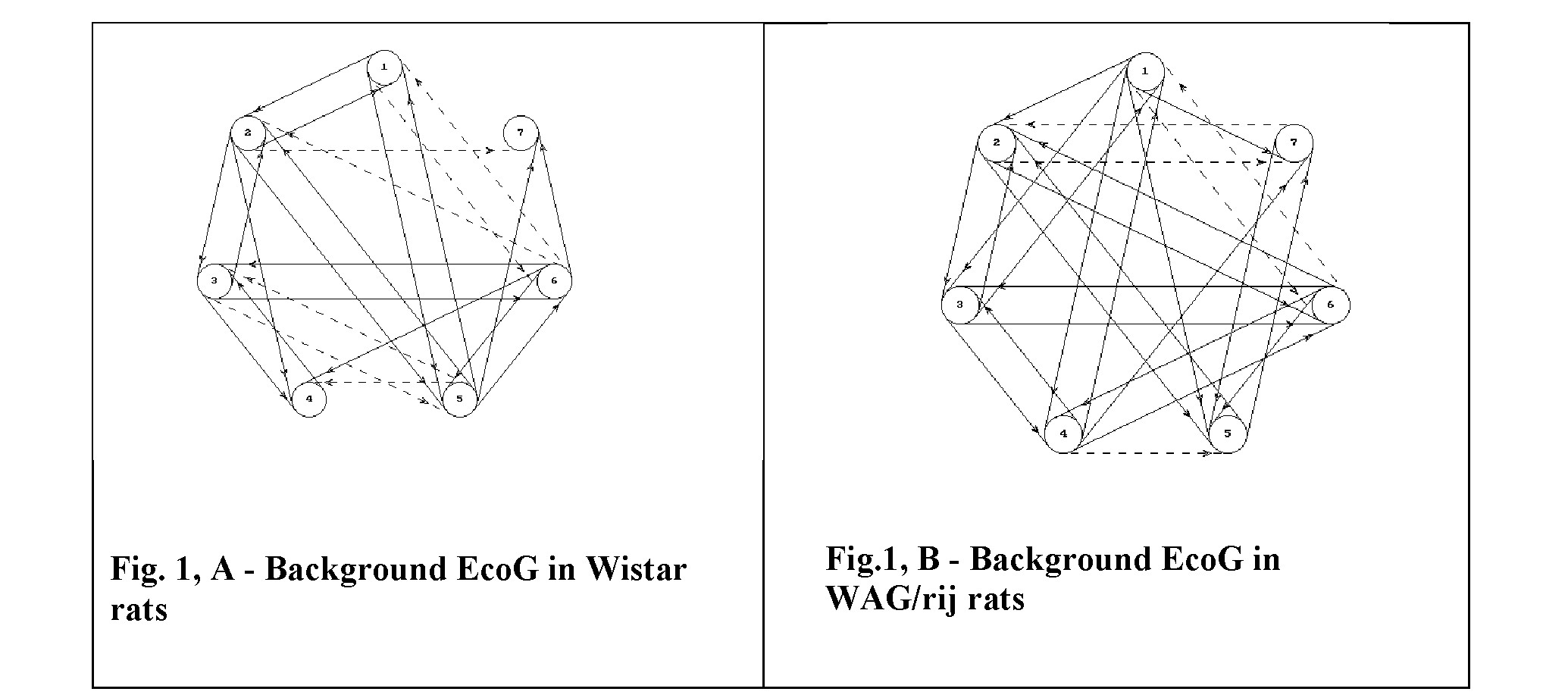
Material and methods. 6-months old WAG/rij rats (3 animals) under Nembutal anaesthesia (40,0 mg/kg) electrodes were implanted into frontal (AP=3,2; L=1,8) [3], occipital (AP=-7,8; L=2,5), and temporal (AP=- 7,8; L=7,0) zones of cortex of both hemispheres. Experimental observations started in 7 days from the moment of operation. Bipolar registration was performed with time constant of 0,3- 1,0 sec. The next leads were used for registration in left hemisphere: fronto- temporal (marked on Fig 1 with N1), temporo- occipital (N2) and fronto- occipital (N3). Similar system of leads was explored for the cortex of right hemisphere, which are marked with N4, N5 and N6 on Fig. 1 correspondently). With N7 the paleocerebellar lead is marked. With the aim of initiating of the generalized seizure activity animals were treated i.p. with solution of sodium salt of benzilpenicillin (300.000 IU/100 g) [3]. 4 Wistar rats administered with the analogous dosage of penicillin solution were used as a control.
EcoG registration was performed with frequency of discretization of 256 samples/sec in free moving rats. Mathematical models were created using self-prepared software which was
made on the basis of multiple linear regression and correlation. Average amplitude of EcoG signal determined for 10 sec period of registration was used. Altogether 30 such periods were used for final calculation. Finally, oriented functional influences were identified of positive (marked on Figures with solid lines) and negative character (interrupted lines). The direction of influence is marked with arrow. Only those influences were depicted which were statistically significant at P<0,1 [3].
Results and discussion. Basal activity in brain cortex of WAG/rij rats was characterized by spike- waves development with a frequency of discharges from 7 to 10 per sec and amplitude from 100 to 450 mcV. The average duration of complexes was 5 seconds (from 1 to 10 seconds). The frequency of appearance of such complexes wasw from 15 to 20 per hour. Specific behavior of animals during such bursts precipitation was confined to vibriss tremor, stereotype subtle nodding of head. Besides, animals did not respond to tactile stimuli of skin and vibrisses.
The multigraf, which represents the interaction between brain structures, which were under investigation was characterized by the presence of a number of connections and by the prevalence of positive mutual influences between structures and hemispheres as well (Fig. 1,A). All structures of left hemisphere were encompassed by mutual positive connections while between structures of right hemisphere few connections were present.
The comparison of this multigraf with the EcoG registered in Wistar rats showed that in Wistar rats structures of each hemisphere are far from being encompassed in the whole entity: between structures one-side less numerous influences were identified. The intensive mutual negative influences were present between hemispheres. The absence of influences from cerebellar cortex on hemispheres also was characteristic for Wistar rats EcoG (Fig. 1,B).
The penicillin action (270-400 sec from the moment of administration) was connected with the increasing of the frequency of spike-wave bursts up to 3-6 per minute. The amplitude of discharges was equal to 300,0- 750,0 mcV and duration of bursts- 2- 7 sec. The frequency of spike-wave discharges was from 7 to 10 sec. The amplitude of discharges in paleocerebellar cortex was from 120,0 to 300,0 mcV and their frequency- 7-8 per sec.
The multigraph presentation of data during this period was in favor of the prevalence of mutual positive connections between structures, which were under investigation. Particularly, all structures of the left hemisphere were encompassed by mutual positive influences, and that was not characteristic for the structures of right hemisphere (Fig. 2,A). Besides, between
3structures of right hemisphere mutually negative interconnections were present. Under these condition, between cerebellum, on the one sides, and both fronto- temporal of the left hemisphere and fronto-occipital zones of right hemisphere, on the other side, the mutul negative connections were established. Altogether between hemispheres four oriented positive lines of influences were observed in the absence of the negative one.
Such character of interrelationships permits to characterize them as encompassing of all structures of left hemisphere by epileptic activity in some form of “epileptic system” with the consequent expansion of such system to opposite hemisphere. Such an expansion prohibits the formation of entity between cortical structures on the right side of the brain. All that is happened along with the maintenance of negative mutual influences between cortical sites of both hemispheres and paleocerebellar cortex. Hence, these negative relations might be characterized as inhibitory influences of cerebellum upon certain zones of cortex.
Next stage of penicillin- induced epileptogenesis (450- 820 sec from the moment of penicillin administration) was characterized by substitution of spike- wave bursts on synchronous epileptic spikes. First spikes in 2 out of 3 rats appeared in cortex of right hemisphere. During this period the marked reduction of positive influences between structures interhemispherically was observed. Namely, if general number of positive connections at the stage of penicillin- induced spike wave bursts intensification (Fig1,A) was equal to 9, and there were any negative ones, period of appearance of spikes and declining of spike-wave activity was characterized by reduced number of positive (up to 4) along with appearance of 4 negative connections (Fig. 2,B). During this period the sign of interaction of cerebellar cortex with fronto-temporal zone of left hemisphere and fronto- occipital zone of right hemisphere was changed from positive to negative ones. Simultaneously the negative relations with fronto- occipital zone of left hemisphere was established. Altogether all this dynamic might be characterized as a process of substitution of interhemispheric relationships from positive to negative one.
Hence, transformation of spike-wave rhythm into spike type of activity is characterized by changes of system relationships between investigated structures in the form of general reduction of mutual positive relationships (both inside- and between hemispheres), and simultaneous increasing of negative relationships which prevalently developed between hemispheres and between neocortex and paleocerebellar cortex. It should be stressed that this dynamic was followed by the development of severe seizure reaction and lethal outcome was observed in all WAG/rij rats in 120 min from the moment of penicillin administration, while Wistar rats (7 animals) survived 6 h from the moment of analogous epileptogen administration.
4
Thus, observed prevalence of positive relationships between cortical structures in course of spike- wave activity generation might be in favor for the synchronous involvement of all inspected cortical areas into shifting of amplitudes of all registered signals/discharges. This feature, which was pertinent for WAG/rij rats, was quite in difference when compared with generalized seizures in Wistar rats. Thus, for the generalized seizure activity the stable character of positive relations was seen only for interhemisphere interaction, while the rest of the cortical zones were involved into such form of interaction on less stable basis.
Conclusion: penicillin- induced seizures in animals with absence type of epilepsy represents “aggressive” form of epileptogenesis, which might contribute to the problem of pharmacological resistance of epilepsy.
LITERATURE
- Cause and effect relations in disease; lessons from epileptic syndromes in animals/ L.S.Godlevsky, van E.L.J.M.Luijtelaar, A.A.Shandra A.M.L. Coenen // Medical Hypothesis (London).- 2002.- Vol., 58, №3.- P. 237- 243.
- The cortico-thalamic theory for generalized spike- wave discharges/ H.K.M.Meeren, E.L.J.M. van Luijtelaar, F.H.Lopes de Silva et al. // Uspehi Fiziologicheskih Nauk, (In Russian).- 2004.- Vol. N35.-P. 3-19.
- Functional relationships between brain and cerebellar cortex during absence and clonic seizures/ V.N.Zaporozhan, L.S.Godlevsky, G.N.Vostrov et al. // Functional Neurology, Rehabilitation, and Ergonomics.— 2011.—Vol. 1, Issue 1.— P. 39-52.