Summary
In saline soils, imbalance of nutrients exerts profound effects on growth and nutrition of plants. Sunflower plants were affected by NaCI (0, 10, 20, 40, 80 and 160 mM) plus 2 or 10 mM CaSCM in Hoagland nutrient solution. Salinity, IncreasedNa+ and Mg2+and decreased K+, Ca2+, K+ /Na+, Ca2+/Na+ and Ca2+/Mg2+ content in plant parts. Reduction in intercellular space is less than cytoplasm in root cortical cells. Supplemental calcium in presence of salinity reduced Na+ and Mg2+ and increased Ca2+, K+/Na+, Ca2+/Na+ and Ca2+ZMg2+ content in plant organs and selectivity of K+ over Na+ by roots. Also it improves calcium transport from stem to mature leaves and showon a direct relationship between Na+/K+ ratio in rooting medium and cation ratio in plant parts. So, 10 mM of supplemental calcium is advisable for growth improvement of sunflower in presence of 40 mMNaCI (EC= 5 dS/m) in growth medium.
INTRODUCTION
Soil salinity and an imbalance of nutrient elements exert profound effects on plant growth and development. A simultaneous presence of salts and nutrient elements in soil can influence ion uptake by plant roots (Yeo, 2007). The cation-anion balance in saline soil and level of fertility (nutrient availability) may influence salinity tolerance in plants. Since both "high salinity and low fertility" are important growth limiting factors, their moderation is likely to affect crop growth response. Calcium must remain available in ionic form in the soil for absorption by roots (Pilbeam and Morley, 2007). Higher plant tissues contain Ca2+ in the range of 5-30 mg g 1 dry weight (Mengel and Kirkby, 2001).Under NaCI salinity, the rate of Na+ accumulation in roots and its transport to shoots not only increases but it also results in an inhibition of Ca2+Uptake and its movement across the root cells (Tester and Davenport, 2003). The rate of Ca2+ uptake by plant roots is generally low because Ca+2Can be absorbed efficiently by the root tips and adjacent undifferentiated zone in which the endodermal cell wall is still not suberized (Davenport, 2007/ Root growth is severely restricted in Ca2+-deficient soils (Gregory, 2006; Fageria, 2009; Beeckman, 2010). In saline soil, plants with poor root systems due to inadequate calcium availability in the growth medium have been observed to transfer large quantities of sodium to the shoot. Calcium availability in the growth medium seems to have an important role in salt tolerance in plants in saline soils (Collins et al., 2008). It is a non-toxic nutrient even at high content in growth medium and is very effective in detoxifying high concentrations of other elements in plant tissues (Marschner, 1998;Conn and Gillihaml 2010).In saline medium, calcium can mitigate the displacement of K+ by Na+ (Niazi et al., 2005), prevent Na+ influx into the cell wall, minimize K+ leakage caused by excess Na+ in the cell wall and maintain K+ influx (Tester and Davenport, 2003).These functions may be seriously impaired by reduced Ca2+ availability under saline conditions because of ionic interactions, precipitation and enhanced ionic strength. These combined effects are responsible for reduced crop yield under saline conditions (Grattan and Grieve, 1999). Therefore, maintenance of an adequate supply of Ca+2 in the growth medium is an important factor in controlling the severity of Na+ toxicity in saline soils. An elevated Ca2+ concentration in the external medium containing high salt content enhances Ca2+ status of tissue, preventing cytosolic K+ leakage and suppressing salt-induced reduction of plant growth, thereby allowing root elongation and root production (Cramer et al., 1994).Not much is known about the uptake, transport, distribution and accumulation of cations in sunflower. Particularly, there are no reports on the critical content of Ca2+in different parts in sunflower plants in saline conditions. Present work aims to investigate the distribution pattern of four major cations (Na+, K+, Ca2+ and Mg2+) in saline soil in relation to growth of sunflower plants ( cv. Morden) and to determine the variations in the effect of NaCI salinity on the accumulation of cations in different plant parts in vegetative stage. Effect of supplemental calcium under varying NaCI salinity levels, on growth, uptake, transport and accumulation of cations by roots and their distribution in stem and leaves, has also been investigated. A possible role of elevated calcium on the alteration of K+/Na+, Ca2VMg2+ and Ca2+/ Na+ ratios under NaCI salinity has been highlighted.
MATERIALS AND METHODS
Raising of Plants and Treatments
Experiments were performed on sunflower [Helianthus annuus L. cv.Morden) plants raised on purified silica sand in plastic pots with capillary function. Silica sand was sieved through a 2-mm mesh, treated with IN HCI and washed repeatedly with distilled water to remove residual elements. Washing was continued till an EC of less than 0.1 dSm-1and pH of 5.5 were achieved in the residual water. A modified Hoagland's nutrient solution was used for irrigation of sunflower plants in all experiments (Taiz and Zieger, 2006). The macronutrients were added at half-strength and micronutrients at full strength. For supplying sufficient quantity of iron, iron-EDTA complex was used in nutrient solution for plant growth. Electrical conductivity (EC) and pH of the Hoagland nutrient solution were set in 5.5 ± 0.5 and 0.7 - 0.9 dSm 1 range, respectively. Solutions were renewed after every 2days.
Sunflower seeds were obtained from National Seeds Corporation. Seeds of uniform weight were selected for germination. The uniform-sized seeds were selected and surface-sterilized with 0.2% mercuric chloride for 5 min, thoroughly washed with deionized water and soaked for 12h in glass distilled water. Seeds were germinated in plastic pots filled with fine silica sand and they were uniformly irrigated with Hoagland nutrient solution once daily. Emergence of radicle was taken as a sign of germination. The formation and extension of hypocotyls was observed after 48h.When the cotyledons had fully expanded, three uniform seedlings were retained for growth in each pot. Potted plants were maintained in controlled environment and subjected to 16h photoperiod diurnally at 25±1°C and 80± 5% relative humidity. Throughout the experiment, plants were irrigated daily and uniformly with half-strength Hoagland nutrient solution in presence of 10, 20, 40, 80 or 160 mMNaCI with or without supplemental calcium. Control plants were not subjected to any NaCI treatment. Plants were harvested after 30 days of growth. Calcium Sulphatewas used as source of calcium in the growth medium. Keeping in view the possibility that sulphate concentrations above 5 mM in growth medium could prove toxic, the growth of sunflower plants was compared in presence of two kinds of sodium salts in half-strength Hoagland nutrient solution. In one set (Tl), sulphate concentration was 0.5 mM and calcium was 2 mM. In the second set (T2), sulphate and calcium concentrations were 10 mM each (Table 1).
Tablet. Ionic composition of nutrients (in mM) in the modified Hoagland nutrient solution used for raising sunflower plants
|
Treatments |
NO3- |
SO42- |
H2PO4- |
ci- |
K+ |
Ca2+ |
Mg2+ |
Na+ |
|
Control |
8.0 |
0.5 |
1.0 |
0.05 |
3.0 |
2.0 |
0.5 |
0.05 |
|
Tl[1] |
8.0 |
0.5 |
1.0 |
40 |
3.0 |
2.0 |
0.5 |
40 |
|
T2* |
8.0 |
10 |
1.0 |
40 |
3.0 |
10 |
0.5 |
40 |
Standard solution for estimation of sodium, potassium, calcium and magnesium (5-100 ppm), were prepared using NaCI, KCI, Ca (N03)2 and Mg (N03)2, respectively. Cation contents of plant digests were calculated on dry weight basis and expressed in mg g 1DW (Benton Jones, 2001; Reuters, 2008), using the following formula:
C tion tma/k ) - ^ ation (rnS^L) " (method blank) x 25ml ation (mg/kg) -
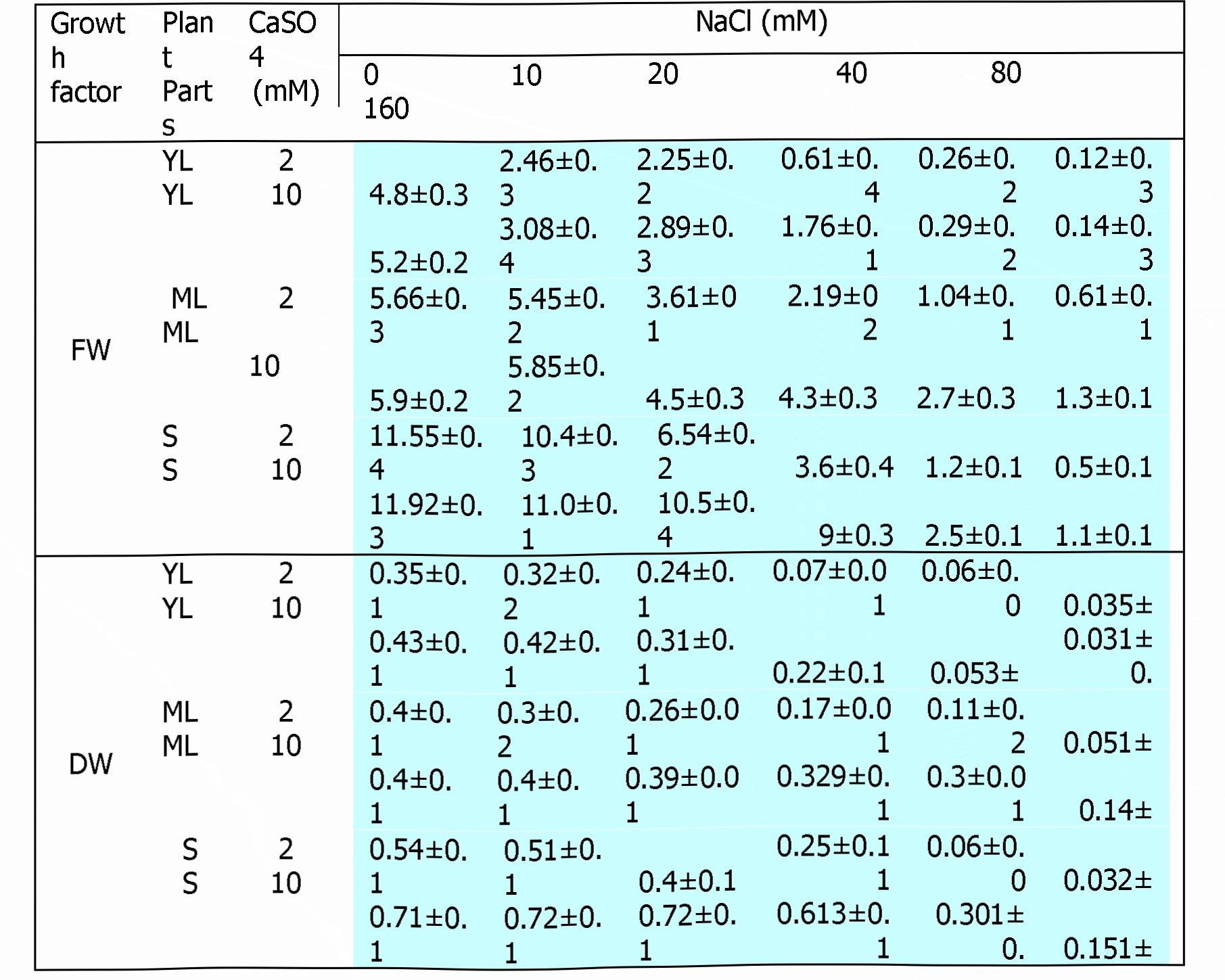
Fresh and dry weights of plant parts were significantly and linearly reduced with NaCI salinity at various levels (Table 2).
Table 2. Fresh weight (FW), dry weight (DW) of young leaves (YL), mature leaves (ML) and stem (S) from 30 d old sunflower plants as affected by NaCI (0, 10, 20, 40, 80 and 160 mM) plus 2 or 10 mM CaSCU in the half-strength Hoagland nutrient solution. Values represent means ± SE.
Supplementation of high dosage of calcium (10 mM) to the growth medium significantly improved fresh weight of mature leaves and stem at all NaCI salinity levels. Fresh weight of young leaves also increased due to supplemental calcium at 10, 20 and 38
40 mMNaCI. Maximum increase in fresh weight due to supplemental calcium was observed at 40 mMNaCI in leaves and stem. However, at 80 and 160 mMNaCI, reduction in fresh weight due to NaCI salinity in young leaves was almost similar with 2 and 10 mM calcium. Similar trend was observed in dry weight improvement due to 10 mM supplemental calcium. At 10, 20 and 40 mMNaCI, dry weight of all plant parts was higher in presence of 10 mM calcium as compared to 2 mM calcium. At 80 and 160 mMNaCI, supplemental calcium (10 mM) improved dry weight of stem and mature leaves. Maximum improvement in dry weight due to supplemental calcium was observed at 40 mMNaCI.
Water Status
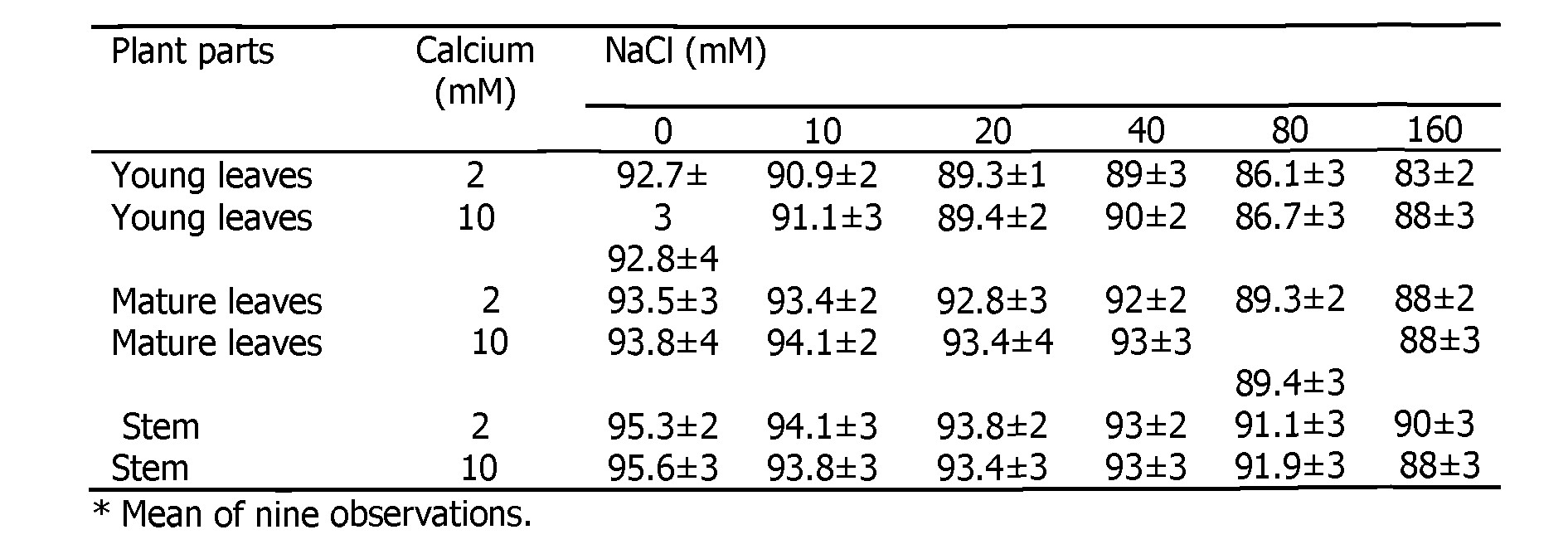
There was no significant difference in the water content of young leaves, mature leaves and stem in non-saline growth conditions and in presence of 2 or 10 mM CaSCM (Table 3).
Table 3. Effect of supplemental calcium (CaSCM) in the growth medium on the water content (% in fresh weight basis) in the parts of 30 d old plants grown in saline medium.
Addition of NaCI to rooting medium exerted a significant adverse effect on the water content of stem and leaves. Addition of 10 mM CaSCM to growth medium did not enhance water content in stems and leaves in salinized sunflower. Overall, extra calcium was not significantly effective in improving water content in plant parts in salt-stressed sunflower plants. Some recovery of water content due to supplemental calcium was evident in leaves of NaCI-Stressed plants. Water content did not, however, improve in stem at any level of NaCI due to 10 mM supplemental calcium.
Sodium Content
Under non-saline conditions, sodium content in plant parts is similar. Sunflower plants seemed to tolerate sodium up to 20 mM but accumulation began thereafter at higher NaCI concentrations in the medium (Fig. 1).
 39
39
Under NaCI salinity in presence of 2 mM calcium, sodium content increased linearly in leaves and stem. In high salinity, roots accumulated most of the sodium. Supplemental calcium in presence of 10-160 mMNaCI significantly reduced sodium content in stem and leaves. In presence oflO mM supplemental calcium, sodium content in young leaves remained negligible and unchanged at 10, 20 and 40 mMNaCI. Overall, reduction of sodium content due to supplemental calcium in young leaves was more than that in mature leaves and stem at all NaCI levels. Roots showed sensitivity to supplemental calcium (10 mM) by accumulating slightly lesser amount of sodium with 80- 160 mMNaCI in the growth medium. But this decline in sodium accumulation did not seem to be statistically significant.
Potassium Content
Under non-saline conditions, potassium content in stem was almost similar to that in young and mature leaves. At all NaCI salinity levels, potassium content in plant parts was less than that in control plants (Fig. 2).



Fig.2 :Potassium content in young leaves (A), mature leaves (B), stem (C) and root (D) in 30 d old plants as affected by 2 mM (A) or 10 mM ( ) CaSO4 content and NaCI concentration (0, 10, 20, 40, 80 and 160 mM) in the half- strength Hoagland nutrient solution. Values represent means ± SE (n=3).
It declined with increasing NaCI content in the growth medium although young leaves seemed insensitive to low NaCI (10-40 mM). In mature leaves and stem, potassium content decreased linearly but its rate of reduction in stem was higher than in mature leaves. Application of 10 mM supplemental calcium to the growth medium (as compared to 2 mM calcium)could not further improve the potassium content in young and mature leaves and in stem, and it remained similar as that observed with 2 mM calcium at all NaCI concentrations tried. In roots, it improved slightly at 10 mMNaCI due to supplemental calcium.
Calcium Content
Calcium content in aerial plant parts was considerably more than that of other cations in sunflower plants grown under non-saline conditions. In control plants, calcium content in stem was more than that in mature and young leaves and there was no significant difference in calcium content in leaves (Fig.3).
OVJl U LILJI I. VCIIUL-J I t,|JI LJ\_I IL I I ILUI IO Jl- I I — .JJ.
Under all NaCI salinity levels together with low level of calcium (2 mM) in the growth medium, calcium content in stem and leaves was significantly less than that in plants under non-saline conditions. Maximum reduction in calcium content in aerial plant parts was observed in stem. It decreased from 6.28 mg g 1 DW in stem in control plants to 0.83 mg g 1 DW in presence of 160 mMNaCI. Minimum reduction in calcium content due to NaCI salinity was observed in leaves. Increasing calcium content from 2 to 10 mM in the saline growth medium leads to increase in calcium content in roots, stem and mature leaves but not in young leaves.
Magnesium Content
Under non-saline conditions, magnesium content in stem and root was similar and it was more than that in leaves. Magnesium content increased drastically in response 41
to NaCI salinity up to 80 mM in young leaves and 40 mM in mature leaves. It remained unchanged in stem at 10, 20 and 40 mMNaCI and decreased slightly at 80 and 160 mMNaCI (Fig. 4).

Fig.4:Magnesium content in young leaves (A), mature leaves (B), stem (C) and root (D) in 30 d old plants as affected by 2 mM (A) or 10 mM ( ) CaSCM content and NaCI concentration (0, 10, 20, 40, 80 and 160 mM) in the half- strength Hoagland nutrient solution. Values represent means ± SE (n=3).
In contrast to 2 mM calcium, 10 mM calcium treatment decreased magnesium content in leaves. After reduction, it was similar to that in the leaves at non-saline conditions. 10 mM supplemental calcium decreased magnesium content in stem as compared to 2 mM calcium as well. Reduction in magnesium content due to supplemental calcium in leaves was more than that in stem at all NaCI salinity levels.
Cations Ratios
Under non-saline conditions, K+/Na+ ratio in mature leaves was more than that in young leaves (Table 4).
Table 4. K+/Na+, Ca2+/Na+, Ca2+/Mg++ ratios in young leaves (YL), mature leaves (ML), stem (S) and root (R) from 30 d old plants as affected by NaCI concentration (10, 20, 40, 80 and 160 mM) plus 2 or 10 mM CaSCb supplementation in half- strength Hoagland nutrient solution. Values represent means ± SE.
|
Cation ratios |
Plant parts |
CaSO4 _ (mM) |
0 |
10 |
NaCI (mM) |
160 |
|||
|
20 |
40 |
80 |
|||||||
|
YL |
2 |
10.5 |
2.99 |
2.49 |
1.77 |
0.67 |
0.32 |
||
|
YL |
10 |
19.21 |
18.2 |
14.6 |
14.3 |
1.36 |
0.29 |
||
|
ML |
2 |
12.57 |
4.2 |
2.14 |
1.54 |
0.62 |
0.32 |
||
|
K+/Na+ |
ML |
10 |
23.13 |
17.14 |
6.8 |
2.88 |
0.96 |
0.39 |
|
|
S |
2 |
6.47 |
1.65 |
0.52 |
0.52 |
0.16 |
0.16 |
||
|
S |
10 |
8.6 |
5.86 |
0.95 |
0.48 |
0.23 |
0.18 |
||
|
R R |
2 10 |
7.68 |
1.55 |
0.83 |
0.21 |
0.06 |
0.04 |
||
|
10.7 |
2.91 |
1.4 |
0.35 |
0.11 |
0.05 |
|
|
YL 2 |
15.8 |
10 |
6.2 |
5.3 |
2.06 |
0.83 |
|
YL 10 |
67 |
52 |
52 |
65 |
4.7 |
1.49 |
|
ML 2 |
41 |
9.18 |
7.64 |
5.34 |
1.5 |
0.5 |
|
ML 10 Ca2+ZNa+ c ., |
95 |
51.5 |
32.93 |
16.72 |
3.6 |
1.49 |
|
S Z |
7.04 |
3.22 |
2.1 |
1.56 |
1.02 |
0.44 |
|
S 10 |
19.84 |
16.6 |
4.62 |
2.91 |
1.55 |
1.05 |
|
R 2 |
25.14 |
6.75 |
5.04 |
1.62 |
0.53 |
0.36 |
|
R 10 |
35.09 |
12.73 |
9.08 |
2.7 |
1.04 |
0.73 |
|
YL 2 YL 10 |
22.21 |
4.67 |
4.70 |
4.39 |
4.27 |
4.01 |
|
23.42 |
14.09 |
11.07 |
10.25 |
7.19 |
6.50 |
|
|
ML 2 ML 10 |
22.33 |
6.03 |
6.03 |
6.11 |
6.71 |
7.96 |
|
Ca247Mg2+ |
23.33 |
17.05 |
15.35 |
15.03 |
12.88 |
11.22 |
|
S 2 |
19.61 |
5.09 |
4.70 |
5.12 |
5.10 |
6.01 |
|
21.84 |
16.94 |
15.99 |
10.96 |
9.24 |
8.24 |
|
|
R 2 |
11.77 |
6.82 |
6.74 |
5.39 |
5.01 |
4.51 |
|
27.77 |
20.18 |
19.35 |
19.06 |
23.15 |
22.27 |
It was least in stem. K+/Na+ ratio gradually and linearly decreased in leaves and stem with increasing concentration of NaCI in the growth medium. Under all NaCI salinity levels, this ratio was higher in leaves than in stem. Application of 10 mM calcium to saline growth medium enhanced K+/Na+ in stem and leaves. Maximum increase was observed at 10 mMNaCI due to calcium supplementation (10 mM) in leaves and stem. 10 mM calcium could not improve K+/Na+ ratio at 80 and 160 mMNaCI in young leaves, at 40-160 mM in mature leaves and at 20- 160 mM in stem.Ca2+/Na+ ratio in mature leaves under non-saline conditions was more than that in young leaves and was least in stem (Table 4). Under NaCI salinity, this ratio decreased in stem, leaves and roots. The reduction in mature leaves was more than that in young leaves, stem and roots. Supplemental calcium improved Ca2VNa+ ratio in all plant parts. In all concentrations of NaCI, this ratio improved in leaves more than that in stem. Maximum improvement was observed in young leaves. Under NaCI salinity, Ca2VMg2+ ratio decreased markedly in stem and leaves. Due to 10 mM supplemental calcium, this ratio increased in stem, leaves and roots at all NaCI salinity levels. Maximum improvement was observed at low salinity in leaves and stem (Table 4).
DISCUSSION
Three types of salinity and nutrient interactions have been reported to operate in plants. In the first case, their effects are independent at their optimum levels since the relative biomass yield is similar in thetwo conditions. The second interaction exhibits decreased salt tolerance at Suboptimal nutrient levels while the third shows increased salt tolerance in presence of adequate nutrients (Brady, 2007; Lucero, 2009). In this work, the best growth response to 10 mM supplemental calcium was observed in presence of 40 mMNaCI and it showed a direct relationship between the ratio of sodium and calcium ions in the rooting medium and cation ratio in leaves and stem. It can be suggested that an otherwise optimal availability of calcium in the growth medium in non-saline conditions proves to be deficient under saline condition for sunflower growth. It seems that under low salinity (10 and 20 mMNaCI), nutrient deficiency or imbalance limits the growth of sunflower plants more than toxic effects of salinity, and a positive interaction or increased salt tolerance response occurs due to supplemental calcium. Under 43
moderate salinity (40mM NaCI), nutrient deficiency and salinity stress may equally limit plant growth and a positive interaction occurs, thereby improving the plant growth significantly. Under high concentration of NaCI (80 and 160 mM), salinity limits the growth of sunflower plants more than nutrient deficiency does. Plant performance tends to exhibit a positive interaction or an "increased salt tolerance" response if a nutrient element is limiting growth under low salinity levels, and higher salinity treatment proves toxic, thereby severely limiting growth. Delgado and Sanchez-Raya (2007) reported that addition of calcium to the growth medium significantly improves seed germination and survival Ofsunflower seedlings in presence of 50 mMNaCI. Similarly, a positive correlation has been observed between dry weight and calcium content in stem and leaves at 10- 40 mMNaCI in the present work. It seems that supplemental calcium (10 mM) in presence of 10-40 mMNaCI helps in overcoming salinity-induced growth inhibition. Similar results have been reported by Arshi et al. (2005) with regard to growth improvement of Senna plants grown due to supplemental calcium under saline conditions.
In addition of salinity levels, the growth ameliorative effect of supplemental calcium under saline conditions depends on the type of plant species, growth conditions, source of calcium (counter anion), concentration of calcium applied to the growth medium and pH of the medium (Mengel and Kirkby, 2001). Addition of calcium to seawater does not significantly improve growth inhibition of whole plant and callus in six species of Brassica (Schmidt et al., 1993). Supplemental calcium in presence of salinity improves the growth of Brassica napus but not that of Brassica rapa, showing genotypical differences in plant growth responses to calcium supplementation (Ashraf and Naqvi, 1992; Kwon et al., 2009). It is to be noted that Ashraf and Naqvi (1992) used Ca (NCh)? as a source of calcium which has NOy instead of CL In their experiments, the effect of Ca2+ may be a minor factor in overcoming plant growth inhibition by salinity as compared to other factors, such as 'osmotic effect', which may not allow sufficient Ca2+ or K+ uptake to improve plant growth.
Calcium content in roots declines significantly under all NaCI salinity levels in sunflower. In barley roots exposed to NaCI salinity, an inhibition of Ca+2Uptake, is observed mainly by an "osmotic effect" of sodium salts. In order to protect sunflower roots against the adverse effects of sodium in the growth medium, calcium concentration has to be much higher than in a balanced nutrient solution. The enhanced Ca2+ and reduced Na+ accumulation and transport to shoots allows NaCI-treated plants to achieve higher levels of calcium and lower sodium. These findings are in agreement with those in Brassica (Kwon et al, 2009). Supplemental calcium treatment improves calcium transport from stem to mature leaves in sunflower in saline conditions. Maximum transport was observed in the presence of 40 mMNaCI. Fageria(2009) reported that even with calcium deficiency in young leaves, older or lower leaves may contain sufficient amounts of calcium. Calcium is translocated preferentially towards the young leaves in sunflower under non-saline conditions as well as in presence of 10 mMNaCI. This phenomenon reduces calcium requirement in shoot buds. A preferential movement of calcium is induced by the auxin (indole-3- acetic acid), which is synthesized in shoot apices (Mengel and Kirkby, 2001). It seems that there is a high rate of calcium export from mature leaves to young leaves in these conditions (NaCI up to 10 mM). But at salinity levels higher than 10 mMNaCI, calcium content in young leaves is less than that in mature leaves. So, it can be suggested that under NaCI>10 mM, stem exports calcium to mature leaves but it does not reach in enough quantity to young leaves. At high salinity (80 and 160 mMNaCI), calcium concentration in the external solution has to be several times higher (more than 10 mM) in order to counteract adverse effect of high sodium and magnesium in sunflower, especially in growing regions.
Another factor determining the calcium requirement in young leaves for optimum growth in sunflower is the concentration of other cations in external solution and in plant parts. Plants can grow satisfactorily in low levels of calcium in nutrient medium provided that other divalent ions are also maintained at low concentrations (Mengel and Kirkby, 2001). Marschner (1998) reported that calcium requirement increases with increasing external concentrations of NaCI or MgCh. It can be concluded that in this situation supplemental calcium is required to detoxify extra magnesium and sodium transportation and their accumulation in young leaves in sunflower plants grown in saline conditions. Increasing the supply of one cation in the nutrient medium generally lowers the concentration of other cations in plant parts. Calcium is readily replaced by other cations, such as magnesium, from its binding sites at the exterior surface of the plasma membrane, so with increasing sodium and magnesium content in plant parts, particularly in young leaves, sunflower plants require more calcium to balance calcium/sodium and calcium/magnesium ratios for optimum growth. This is because in presence of NaCI in the growth medium, magnesium content increases in young leaves as compared to nonsaline conditions in sunflower. Magnesium content has also been reported to increase due to NaCI salinity (50 mM) in reed plant leaves, and magnesium cannot substitute for calcium functions (Merhaut, 2007). Thus, it can be suggested that higher levels of calcium in young leaves are required to detoxify extra content of magnesium and to maintain optimum calcium/magnesium ratio in young leaves.
The positive effects of supplementary calcium on sunflower growth (present work) might also be due to improvement in potassium selectivity and differential transportation rates. Delgado and Sanchez-Raya (1999) reported that potassium supply increases plant leaf surface with moderate salt concentration (50 mMNaCI) in sunflower. Supplemental calcium (10 mM) slightly improves water content in sunflower only in low salinity (10 and 20 mMNaCI) as also reported bySohan et al. (1999). Calcium supplements of 10 mMhave been reported to exert no significant ameliorative effect on the negative effects of NaCI salinity (at 100 and 150 mM) on plant-water relations in sunflower plants (Sohan et al., 1999). It seems that increase in leaf potassium/sodium ratio due to supplemental calcium, as observed in the present work, improves relative water content in sunflower plant parts at low salinity. Apparently sunflower leaves are fully expanded in low salinity, indicating maintenance of turgor pressure by either increasing their organic or inorganic ion content, thereby regulating osmotic potential.
Under non-saline conditions, long distance transport of potassium takes place readily and it is very mobile in sunflower plants. Its main transport direction is towards the meristematic organs so that potassium content in young and mature leaves are similar in sunflower under non-saline conditions. Mengel and Kirkby (2001) reported that potassium from older plant parts is often redistributed to younger parts. Sunflower plants in which the rate of growth is depressed due to NaCI salinity, take up lower amount of potassium as compared to control plants. Supplemental calcium dose not overcome the adverse effect of NaCI salinity on potassium content in young leaves but it improves potassium distribution ratio in leaves as compared to stem at 10 mMNaCI. Arshi et al. (2005) reported that CaCh treatment enhances K+ content in Senna plants, while the combined treatments mitigate the adverse effects caused by NaCI. Calcium seems to alleviate NaCI-induced inhibition of plant growth via the maintenance of net K+ to Na+ selectivity. It is likely that in sunflower, supplemental calcium in growth medium improves potassium uptake by roots more than potassium transport to leaves. Song and Fujiyama (1996) reported that under saline conditions, supplemental Ca2+ increases potassium accumulation in roots and its transport to shoots but decreases Na+ accumulation in roots and transport to shoots in Oryza sativa. Enhanced K+ uptake and reduction in Na+ 45
transport to shoot by supplemental calcium seems to be associated with a positive growth response in stem and mature leaves in sunflower plants at 10-160 mMNaCI, as also observed in reed plants (Takahashi et al, 2007). A role for Ca2+ in inducing enhanced K+ selectivity over Na+ in saline conditions has been reported earlier in the roots of some plant species (Demidchik et al., 2002).
There exists a non-uniform distribution of Na+ and K+ within the sunflower shoot, major content of sodium being retained in old leaves and basal stem internodes, and potassium being available for growth and expansion of young leaves. This situation is observed particularly at 40 mMNaCI in sunflower. Takahashi et al., (2007) reported that the upper part of the Nanpi reed plants (PhragmitesaustraIistrinius) contains low levels of sodium and high levels of potassium. Similar results have been obtained in sunflower (present work). Plants growing under saline conditions suffer from ionic imbalance, nutrient deficiency and specific ion toxicity (Ashraf and Harris, 2004). For avoiding sodium toxicity in stem and leaves, most plant species accumulate major proportion of Na+ in their roots and exclude it from shoots (Ashraf and Harris, 2004). In the present work, salt tolerance appears to be related to accumulation of major proportion of Na+in roots under high salinity. Also, under high salinity, the rate of potassium translocation to young leaves is more than that for sodium translocation. This is a strategy of sunflower plants to protect young leaves from sodium toxicity. These results suggest that salt tolerance mechanisms Ofsunflower plants include an ability to take up K+ and prevent an influx of Na+ and an improved ability to exclude Na+ from shoots. Ashraf and O'Leary (1995) reported that salt tolerance in sunflower is related to exclusion of Na+ from the leaf lamina. Thus, regulation of Na+ uptake and its translocation from root to shoot seems to be a crucial adaptation of plants to sodium salinity (Munns, 1993). In the present work, application of 10 mM calcium reduces Na+ accumulation in stem, mature leaves and young leaves of salt-stressed sunflower plants. The major quantity of sodium remains in sunflower roots in presence of high levels of NaCI. These results are in agreement with the findings of Arshi et al. (2005) who reported that application of 10 mM calcium to saline growth medium reduces Na+ accumulation in the leaves of Senna plants. This is particularly more important in vine crops which are most prone to Na+ injury than most annual crops. In citrus, calcium has been found to be effective in reducing transport of Na+ from roots to leaves, thereby reducing foliar injury (Banuls et al., 1991).
A general beneficial effect of external Ca+2 on plant salt tolerance is often attributed to maintenance of plasma membrane integrity and improving K+/Na+ and selectivity of potassium uptake system (Niazi et al., 2005). Calcium can influence K+/Na+ selectivity at the plasma membrane by affecting ion channels and stimulation of the Na+ /H+ ATPase activity (Rus et al., 2001). Transport of potassium ions improves due to sulfur application (10 mM CaSCU) in sunflower (present work) indicating an increase in K+/Na+ ratio in leaves over root in sunflower due to high level of CaSCXZaman et al. (2002) reported that potassium has a synergistic relationship with sulphur. Garg and Gupta (1997) reported that calcium application enhances the growth of NaCI-Stressed plants by reducing Na+ transport to shoot and increasing K+/Na+ ratio. A high K+/Na+ ratio is more important for many species than simply maintaining a low content of Na+ (Mokhtar et al., 2006). Higher K+/Na+ ratio improves leaf water potential (Saeed Akram et al., 2009). This effect seems to be important in salt tolerance in sunflower plants.
Ca+2/Na+ ratio in mature leaves is much higher than that in young leaves under non-saline conditions, but in saline conditions this ratio is similar in all leaves. Under NaCI salinity, sunflower plants seem to translocate more calcium and less sodium from mature leaves to young leaves and plants try to accumulate sodium in mature leaves as 46
compared to young leaves. Maximum improvement in Ca+2/Na+ ratio in mature leaves and stem are observed at 10 mMNaCI treatment due to supplemental calcium. It means that at this level of NaCI, sodium content is low and calcium is high in mature leaves and stems. It is possible that IOmM supplemental calcium is not required at this level of NaCI to control sodium toxicity in sunflower. Genotypic differences toCa+2/Na+ ratio have also been reported (Grattan and Grieve, 1999). On the basis of increase in magnesium content in stem and leaves due to salinity, it could be suggested that detrimental effects of NaCI on sunflower could also result from the presence of a low level of Ca+2/Mg+2 ratio rather than from the accumulation of toxic levels of Na+ in young leaves in low salinity. At high salinity, both of these factors seem to reduce growth in sunflower plant parts. Garg and Gupta (1997) reported that at equal conductivity (10 dS m-1), MgCh is more detrimental than NaCI for the growth and yield of wheat. Growth rate in barley and water use efficiency in corn are reduced with a decrease in external Ca+2/Mg+2ratios. Magnesium salts have also been reported to reduce root growth of Eucalyptus more than sodium salts, and reduction in root growth is associated with low calcium concentrations in roots (Garg and Gupta, 1997). A simultaneous increase in the concentration of Na+ ions in the nutrient solution results in a considerable increase in the level of Mg+2 in sunflower leaves and stem and an associated decrease in Ca+2 levels (present work). A significant competition for Ca+2 and Mg+2 transport to sunflower shoots under NaCI salinity is evident. As Ca+2 competes with Mg+2 and the binding sites on the root plasma membrane appear to have less affinity for the highly hydrated Mg+2 than for Ca+2, therefore, high concentrations of substrate Ca+2 usually result in increased calcium content in leaves along with marked reduction in magnesium content. For this reason, supplemental calcium is required to improve Ca+2/Mg+2ratios in sunflower plant parts.
Rererences
- Arshi, A., Abdin, M.Z., Iqbal, M. 2005. Ameliorative effects of CaCh on growth, ionic relations, andproline content of Senna under salinity stress. J. Plant Nutr. 28,101-125.
- Ashraf M., O'Leary, J.W. 1995. Distribution of cations is leaves of salt- tolerant and salt-sensitive lines of sunflower under saline conditions. J. Plant Nutr. 18, 2379-2388.
- Ashraf, M., Naqvi, M.I. 1992. Growth and ion uptake of four Brassica species as affected by Na+/Ca2+ ratio in saline sand culture. Z. Pflanzenemahr. Bodenkd.155, 101-108.
- Ashraf, M., Harris, P.J.C.2004. Potential biochemical indicators of salinity tolerance in plants. Plant Sci. 166, 3-16.
- Banuls, J.E.,Legaz, F., Primo-Millo, E. 1991. Salinity- calcium interactions on growth and ionic concentration Ofcitrus plants. Plant Soil.133, 39-46.
- Beeckman, D.2010. Root Development. Blackwell Publishing, Ltd, London.
- Benton Jones, J.2001. Laboratory guide for conducting soil tests and plant analysis. Boca Raton, USA: CRC Press.
- Brady, N.2007. The Nature and Properties of Soils. CRC Press, Florida-Collins R.P., Harris, P.J.C., Bateman, M.J.,Henderson, J. 2008. Effect of calcium and potassium nutrition on yield, ion content and salt tolerance of Brassica Campestris (rapa). J. Plant Nutr.31,1461-1481.
- Conn, S., GiIIihamzM.2010. Comparative physiology of elemental distribution in plants. Ann. Bot. 105,1081-1102.
- Cramer, G.R., AIbericozGJvSchmidt, C.1994. Salt tolerance is not associated with the sodium accumulation of two maize hybrids. Aust. J. Plant Physiol.21, 675-692.
- Davenport, RJ.2007. Ion uptake by plant roots. In:Yeo, A. and Flowers, T.:Plant SoIuteTransport. Blackwell Publishing, London.193-213.
- Delgado, I.C.,Sanchez-Raya, AJ. 1999. Physiological response of sunflower seedlings to salinity and potassium supply. Com. Soil Sci. Plant Anal.30, 773-783.
- Delgado, I.C., Sanchez-Raya, AJ. 2007. Effects of sodium chloride and mineral nutrients on initial stages of development of sunflower life. Commun. Soil Sci. Plant Anal.38, 2013-2027.
- Demidchik, V., Davenport, R. J., Tester, M.2002. Nonselective cation channels. Annu. Rev. Plant Biol. 53, 67-107.
- Fageria, N.K. 2009. The Use of Nutrients in Crop Plants. Boca Raton, USA: CRC Press.
- Garg, B.K., Gupta, I.C. 1997. Saline wastelands environment and plant growth. Scientific publications. Jodhpur, India.
- Grattan S.R., Grieve, C.M. 1999. Salinity-mineral nutrient relations in horticultural crops.Sci. Hort.78, 127-157.
- Gregory, P. 2006. Plant roots: growth activity and interactions with soils. Blackwell Publishing, Ltd, London.
- KirkhamzM-B. 2005. Principle of Soil and Plant Water Relations. Elsevier Academic Press, Boston.
- Kwon, T. R., Siddiqui, Z. S., Harris, PJ.C.2009. Effects of supplemental calcium on ion accumulation, transport and plant growth of salt sensitive Brassica rapa. J. Plant Nutr.32, 644-667.
- Lucero, D.P.2009. Soil Fertility. Blackwell Publishing Ltd, London.
- Marschner, H.1998. Mineral Nutrition of Higher Plants. London, England: Academic Press, London.
- Mengel, K., Kirkby, E.A. 2001. Principles of Plant Nutrition. Kluwer academic publisher, Dordrecht.
- MerhautzD. J.2007. Magnesium. In: Barker A.V. and Pilbeam DJ.: Handbook of Plant Nutrition. CRC Press, Florida, pp:
- MokhtarzR., Mohammad, V.A., Arbi, G., Mohammad, N.2006. Effect of NaCI on the growth and the ionic balance of two populations of Lotus creticus (L.). Lotus Newsletter. 36, 34-53.
- Munns, R. 1993. Physiological processes limiting plant growth in saline soils: some dogmas and hypothesis. Plant Cell Environ.16,15-24.
- Niazi, B.H., Athar, M., Salim, M.,Rozema. J. 2005. Growth and ionic relations of fodderbeet and seabeet under saline habitat. Intl. J. Environ. Sci. Tech.2, 113-120.
- Pilbeam, DJ., Morley, P.S. 2007. Calcium. In: Barker, A.V. and DJ.
- Pilbeam: Handbook of Plant Nutrition. CRC Press, Florida, pp: 287.
- ReuterszDJ. 2008. Soil and PIantAnaIysis. Blackwell Publishing Ltd, London.
- Rus, A., Yokoi, S., Sharkhuu, A., Reddy, M., Lee, B. H., Matsumoto, T.K., Koiwa, H., Zhu, J. K., Bressan, R.A., Hasegawa, P.M. 2001. AtHKTl is a salt tolerance determinant that controls Na+ entry into plant roots. Proc. Natl. Acad. Sci. 98, 14150- 14155.
- Saeed Akram, M., Ashraf, M., Aisha Akram, N. 2009). Effectiveness of potassium sulfate in mitigating salt-induced adverse effects on different physico- biochemical attributes in sunflower. Flora.10,1-13.
- Schmidt, C., He, T., Cramer, G. R.1993. Supplemental calcium does not improve growth of salt-stressed Brassica. Plant. Soil. 155, 415-418.
- Sohan, D., Jasoni, R., Zajicek, J. 1999. Plant water relations of NaCI and calcium- treated sunflower plants. Environ. Exp. Bot.42,105-111.
- Song, J., Fujiyama, H.1996. Importance of sodium content and water status for growth in non-salinized rice and tomato plants. Soil Sci. Plant Nutr. 44,197-208.
- Taiz, L., Zeiger, E.2006. Plant Physiology. Sinauer Associates, Inc., Massachusetts.
- Takahashi, R., Takayoshi, N., Ichizen, N., Takano, T.2007. Salt-tolerant reed plants contain lower Na+ and higher K+ than salt-sensitive reed plants. Acta Physiol.Plant.29, 431-438.
- Tester,M.,Davenport, R. 2003. Na+ tolerance and Na+ transport in higher plants. Ann. Bot.91, 503-527.
- Yeo, A. 2007. Salinity. In:Yeo A. and Flowers, T.: plant solute transport. Blackwell Publishing. Ltd., London, pp. 340-356.
- Zaman, B., Ali, A., Salim, M., Niazi, B.H. 2002. Role of sulphur for potassium/sodium ratio in sunflower under saline conditions. Helia. 37, 69-78.